Abstract
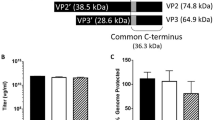
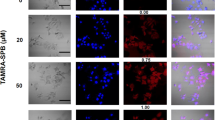
We recently showed that retroviral vectors can be targeted through protease substrate interactions. Infectivity is blocked by a polypeptide fused to the viral envelope glycoprotein (SU) and is restored when a protease cleaves the connecting linker, releasing the inhibitory polypeptide from the viral surface. Protease specificity is achieved by engineering the sequence of the linker. Here, using two different matrix-metalloproteinase (MMP)-activatable vectors, we demonstrated highly efficient and selective transduction of MMP-rich target cells in a heterogeneous cell population. In vivo, the MMP-targeted vectors showed strong selectivity for MMP-rich tumor xenografts. Protease-activatable vectors offer new possibilities for in vivo targeting of gene delivery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Cosset FL, Russel SJ . Targeting retrovirus entry Gene Therapy 1996 3: 946–956
Kasahara N, Dozy AM, Kan YW . Tissue-specific targeting of retroviral vectors through ligand-receptor interactions Science 1994 266: 1373–1376
Somia NV, Zoppe M, Verma IM . Generation of targeted retroviral vectors by using single-chain variable fragment: an approach to in vivo gene delivery Proc Natl Acad Sci USA 1995 92: 7570–7574
Ager S et al. Retroviral display of antibody fragments: interdomain spacing strongly influences vector infectivity Hum Gene Ther 1996 7: 2157–2164
Hall FL et al. Targeting retroviral vectors to vascular lesions by genetic engineering of the MoMLV gp70 envelope protein Hum Gene Ther 1997 8: 2183–2192
Chu TH, Dornburg R . Toward highly efficient cell-type-specific gene transfer with retroviral vectors displaying single-chain antibodies J Virol 1997 71: 720–725
Pinter A et al. Localization of the labile disulfide bond between SU and TM of the murine leukemia virus envelope protein complex to a highly conserved CWLC motif in SU that resembless the active-site sequence of thiol-disulfide exchange enzymes J Virol 1997 71: 8073–8077
Fass D, Harrison SC, Kim PS . Retrovirus envelope domain at 1.7Å resolution Nat Struct Biol 1996 3: 465–469
Fass D et al. Structure of a murine leukemia virus receptor-binding glycoprotein at 2.0 angstrom resolution Science 1997 277: 1662–1666
Cosset F-L et al. Retroviral retargeting by envelopes expressing an N-terminal binding domain J Virol 1995 69: 6314–6322
Nilson BHK, Morling FJ, Cosset F-L, Russell SJ . Targeting of retroviral vectors through protease–substrate interactions Gene Therapy 1996 3: 280–286
Peng KW et al. A gene delivery system activatable by disease-associated matrix metalloproteinases Hum Gene Ther 1997 8: 729–738
Fielding AK et al. Inverse targeting of retroviral vectors: selective gene transfer in a mixed population of hematopoietic and nonhematopoietic cells Blood 1998 91: 1802–1809
Peng KW, Morling FJ, Cosset F-L, Russell SJ . A retroviral gene delivery system activatable by plasmin Tumor Targeting 1998 3: 112–120
Chadwock MP, Morling FJ, Cosset F-L, Russell SJ . Modification of retroviral tropism by display of IGF-I J Mol Biol 1999 285: 485–494
Morling FJ, Peng KW, Cosset F-L, Russel SJ . Masking of retroviral envelope functions by oligomerizing polypeptide adaptors Virology 1997 234: 51–61
Buchholz CJ et al. In vivo selection of protease cleavage sites from retrovirus display libraries Nat Biotech 1998 16: 951–954
Okada A et al. Membrane-type matrix metalloproteinase (MT-MMP) gene is expressed in stromal cells of human colon, breast, and head and neck carcinomas Proc Natl Acad Sci USA 1995 92: 2730–2734
Murphy G et al. Regulation of matrix metalloproteinase activity Ann NY Acad Sci 1994 732: 31–41
Ray JM, Stetler-Stevenson WG . The role of matrix metalloproteases and their inhibitors in tumor invasion, metasasis and angiogenesis Eur Resp J 1994 7: 2062–2072
Edwards DR, Murphy G . Proteases-invasion and more Nature 1998 394: 527–528
Duffy MJ . Proteases as prognostic markers in cancer Clin Cancer Res 1996 2: 613–618
Ray JM, Stetler-Stevenson WG . Gelatinase A activity directly modulates melanoma cell adhesion and spreading EMBO J 1995 14: 908–917
Hooper NM, Karran EH, Turner AJ . Membrane protein secretases Biochem J 1997 321: 265–279
Derian CK, Eckardt AJ, Andrade-Gordon P . Differential regulation of human keratinocyte growth and differentiation by a novel family of protease-activated receptors Cell Growth Differ 1997 8: 743–749
Springett GM et al. Infection efficiency of T-lymphocytes with amphotropic retroviral vectors in cell cycle dependent J Virol 1989 63: 3865–3869
Naldini L et al. Efficient transfer, integration and sustained long-term expression of the transgene in adult rat brains injected with a lentiviral vector Proc Natl Acad Sci USA 1996 93: 11382–11388
Indraccolo S et al. Psuedotyping of Moloney leukemia virus-based retroviral vectors with simian immunodeficiency virus envelope leads to targeted infection of human CD4+ lymphoid cells Gene Therapy 1998 5: 209–217
Poeschla EM, Wong-Staal F, Looney DJ . Efficient transduction of non-dividing human cells by feline immunodeficiency virus lentiviral vectors Nature Med 1998 4: 354–357
Karpusas M et al. 2Å crystal structure of an extracellular fragment of human CD40 ligand Structure 1995 3: 1031–1039
Takeuchi Y et al. Type C retrovirus inactivation by human complement is determined by both the viral genome and producer cell J Virol 1994 68: 8001–8007
Cosset F-L et al. High-titer packaging cells producing recombinant retroviruses resistant to human serum J Virol 1995 69: 7430–7436
Morling FJ, Russel SJ . Enhanced transduction efficiency of retroviral vectors coprecipitated with calcium phosphate Gene Therapy 1995 2: 504–508
Acknowledgements
We thank Drs K Siemering and G Murphy, respectively, for their kind gifts of the GFP expression plasmid, mGFPS65T and the MT1-MMP expressing HT1080 cell line. This work was supported by the UK Medical Research Council Center (KWP and SJR), Mayo and Siebens Foundations (KWP, RGV and SJR), Singapore Nanyang Technological University/National Institute of Education (KWP), Centre National de la Recherche Scientifique (FLC) and Imperial Cancer Research Fund (RGV).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Peng, KW., Vile, R., Cosset, FL. et al. Selective transduction of protease-rich tumors by matrix-metalloproteinase-targeted retroviral vectors. Gene Ther 6, 1552–1557 (1999). https://doi.org/10.1038/sj.gt.3300982
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3300982
Keywords
This article is cited by
-
Viral vectors: from virology to transgene expression
British Journal of Pharmacology (2009)
-
Reprogrammed viruses as cancer therapeutics: targeted, armed and shielded
Nature Reviews Microbiology (2008)
-
Delivery of Viral Vectors to Tumor Cells: Extracellular Transport, Systemic Distribution, and Strategies for Improvement
Annals of Biomedical Engineering (2006)
-
Ligand-directed targeting of genes to the site of disease
Nature Medicine (2003)
-
Targeting the cytotoxicity of fusogenic membrane glycoproteins in gliomas through protease–substrate interaction
Gene Therapy (2003)