Abstract
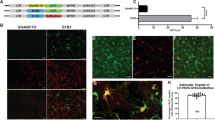
We have examined the potential of cationic liposomes as a tool for approaches to gene therapy in the CNS. Our previous work has shown that cationic liposomes formulated from 3β-[N-(N′,N′-dimethylaminoethane)carbamoyl] cholesterol (DC-Chol) and dioleoyl-L-α-phosphatidylethanolamine (DOPE) could achieve high transfection levels in a neuronal cell line (McQuillin et al. Neuroreport 1997; 8: 1481–1484). We therefore wished to assess transfection efficiencies in organotypic cultures from the brain with a reporter plasmid expressing E. coli β-galactosidase in order to mimic an in vivo model. Explant cultures were generated according to the method of Stoppini et al (J Neurosci Meth 1991; 37: 173–182) with slight modifications. Brain slices were maintained on transparent porous membranes and were observed to maintain their intrinsic con- nectivity and cytoarchitecture to a large degree over periods of up to 6 weeks in culture. CNS tissue was obtained from rats at birth or 5 days after birth. After transfection β-galactosidase expression was detected in cells of both neuronal and non-neuronal morphology. Control cultures were exposed to liposome alone and a plasmid that had the β-galactosidase gene insert removed. Only low levels of endogenous β-galactosidase reactivity were seen in these control cultures. DC-Chol/DOPE-mediated transfection was confirmed using a RT-PCR protocol capable of differentiating between untranscribed plasmid DNA and RNA generated from the transfected vector. These results suggest that cationic liposomes, particularly DC-Chol/DOPE liposomes, will be useful as delivery agents for gene transfer to CNS cells in vitro and possibly in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Karpati G et al. The principles of gene therapy for the nervous system Trends Neurosci 1996 19: 49–54
Suhr ST, Gage FH . Gene therapy for neurologic disease Arch Neurol 1993 50: 1252–1268
Blomer U et al. Applications of gene therapy to the CNS Hum Mole Genet 1996 5: 1397–1404
Lasalle GL et al. An adenovirus vector for gene transfer into neurons and glia in the brain Science 1993 259: 988–990
Wood MJA et al. Specific patterns of defective HSV-1 gene transfer in the adult central nervous system – implications for gene targeting Exp Neurol 1994 130: 127–140
Wood MJA et al. Inflammatory effects of gene transfer into the CNS with defective HSV-1 vectors Gene Therapy 1994 1: 283–291
Byrnes AP et al. Adenovirus gene transfer causes inflammation in the brain Neuroscience 1995 66: 1015–1024
Naldini L et al. In vivo gene delivery and stable transduction of nondividing cells by a lentiviral vector (see comments) Science 1996 272: 263–267
Gao X, Huang L . Cationic liposome-mediated gene transfer Gene Therapy 1995 2: 710–722
Felgner PL et al. Lipofection – a highly efficient, lipid-mediated DNA-transfection procedure Proc Natl Acad Sci USA 1987 84: 7413–7417
Farhood H, Serbina N, Huang L . The role of dioleoyl phosphatidylethanolamine in cationic liposome mediated gene transfer Biochim Biophys Acta 1995 1235: 289–295
Caplen NJ et al. In vitro liposome-mediated DNA transfection of epithelial cell lines using the cationic liposome DC-Chol/DOPE Gene Therapy 1995 2: 603–613
Labat-Moleur F et al. An electron microscopy study into the mechanism of gene transfer with lipopolyamines Gene Therapy 1996 3: 1010–1017
Zabner J et al. Cellular and molecular barriers to gene transfer by a cationic lipid J Biol Chem 1995 270: 18997–19007
Felgner JH et al. Enhanced gene delivery and mechanism studies with a novel series of cationic lipid formulations J Biol Chem 1994 269: 2550–2561
Sahenk Z et al. Gene delivery to spinal motor neurons Brain Res 1993 606: 126–129
Iwamoto Y et al. Liposome-mediated BDNF CDNA transfer in intact and injured rat brain Neuroreport 1996 7: 609–612
Roessler BJ, Davidson BL . Direct plasmid-mediated transfection of adult murine brain cells in vivo using cationic liposomes Neurosci Lett 1994 167: 5–10
Zhou X, Huang L . DNA transfection mediated by cationic liposomes containing lipopolylysine: characterization and mechanism of action Biochim Biophys Acta 1994 1189: 195–203
McQuillin A et al. Optimisation of liposome mediated transfection of a neuronal cell line Neuroreport 1997 8: 1481–1484
Caplen NJ et al. Liposome-mediated CFTR gene transfer to the nasal epithelium of patients with cystic fibrosis Nature Med 1995 1: 39–46
Nabel GJ, Nabel EG, Yang ZY . Direct gene-transfer with DNA liposome complexes in melanoma – expression, biological activity, and lack of toxicity in humans Proc Natl Acad Sci USA 1993 90: 11307–11311
Nabel G, Chang A, Nabel E . Clinical protocol: immunotherapy of malignancy by in vivo gene transfer into tumors Hum Gene Ther 1992 3: 399–410
Stewart MJ et al. Gene transfer in vivo with DNA liposome complexes – safety and acute toxicity in mice Hum Gene Ther 1992 3: 267–275
Stoppini L, Buchs PA, Muller D . A simple method for organotypic cultures of nervous tissue J Neurosci Meth 1991 37: 173–182
Ostergaard K, Finsen B, Zimmer J . Organotypic slice cultures of the rat striatum – an immunocytochemical, histochemical and in situ hybridization study of somatostatin, neuropeptide-Y,nicotinamide adenine-dinucleotide phosphate-diaphorase, and enkephalin Exp Brain Res 1995 103: 70–84
Forno LS . Neuropathology of Parkinson’s disease J Neuropathol Exp Neurol 1996 55: 259–272
Ostergaard K, Schou JP, Zimmer J . Rat ventral mesencephalon grown as organotypic slice cultures and cocultured with striatum, hippocampus, and cerebellum Exp Brain Res 1990 82: 547–565
Bankiewicz K, Mandel RJ, Sofroniew MV . Trophism, transplantation, and animal models of Parkinson’s disease Exp Neurol 1993 124: 140–149
Alton EWFW et al. Non-invasive liposome-mediated gene delivery can correct the ion transport defect in cystic fibrosis mutant mice Nat Genet 1993 5: 135–142
Chomczynski P, Sacchi N . Single-step method of RNA isolation by acid guanidinium thiocyanate phenol chloroform extraction Anal Biochem 1987 162: 156–159
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Murray, K., McQuillin, A., Stewart, L. et al. Cationic liposome-mediated DNA transfection in organotypic explant cultures of the ventral mesencephalon. Gene Ther 6, 190–197 (1999). https://doi.org/10.1038/sj.gt.3300743
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3300743
Keywords
This article is cited by
-
Recent Advances in Self-assembled Nano-therapeutics
Chinese Journal of Polymer Science (2018)
-
Characterisation of LMD virus-like nanoparticles self-assembled from cationic liposomes, adenovirus core peptide μ (mu) and plasmid DNA
Gene Therapy (2002)