Abstract
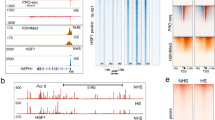
DNA sequences, important for the control of Drosophila heat shock gene expression, are packaged in chromatin in a nuclease hypersensitive configuration1. Recently, two protein-binding (exonuclease-resistant) sites which cover the TATA box sequence and an upstream control element were shown to occur in vivo amidst the 5′ terminal hypersensitive regions of several heat shock genes2. Protein-binding at the TATA box is independent of heat shock, but the binding at the upstream element is heat shock dependent, and it was proposed that a heat shock activator protein, HAP, positively regulates the genes2. Here, I report the detection of HAP activity in heat shocked cell extracts by reconstituting specific binding to hsp82 gene chromatin in vitro. Inhibition of the binding by free DNA from the 5′ region of heat shock genes implies a coordinate regulation of the gene family through HAP interaction with the upstream heat shock consensus sequence3. Furthermore, the special ease of induction of the hsp82 gene over other heat shock genes can be explained in molecular terms by the higher affinity of HAP for the hsp82 binding site, which contains a 28 base sequence with almost perfect dyad symmetry, GAAGCCTCTAGAAG|TTTCTAGAGACTTC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Wu, C. Nature 286, 854–860 (1980).
Wu, C. Nature 309, 229–234 (1984).
Pelham, H. R. B. Cell 30, 517–528 (1982).
Holmgren, R., Livak, K., Morimoto, R., Freund, R. & Meselson, M. Cell 18, 1359–1370 (1979).
Holmgren, R., Corces, V., Morimoto, R., Blackman, R. & Meselson, M. Proc. natn. Acad. Sci. U.S.A. 78, 3775–3778 (1981).
O'Connor, D. & Lis, J. T. Nucleic Acids Res. 9, 5075–5092 (1981).
Hackett, R. W. & Lis, J. T. Nucleic Acids Res. 11, 7011–7030 (1983).
Blackman, R. K. thesis, Harvard Univ. (1983).
Storti, R. V., Scott, M. P., Rich, A. & Pardue, M. L. Cell 22, 825–834 (1980).
Lindquist, S. Devl Biol. 77, 463–479 (1980).
Buzin, C. M. and Petersen, N. S. J. molec. Biol. 158, 181–201 (1982).
Spradling, A., Pardue, M. L. & Penman, S. J. molec. Biol. 109, 559–587 (1977).
Ashburner, M. Chromosoma 31, 356–376 (1970).
Emerson, B. M. & Felsenfeld, G. Proc. natn. Acad. Sci. U.S.A. 81, 95–99 (1984).
Jack, R. S., Gehring, W. J. & Brack, C. Cell 24, 321–331 (1981).
Compton, J. L. & Bonner, J. J. Cold Spring Harb. Symp. quant. Biol. 42, 835–838 (1978).
Compton, J. L. & McCarthy, B. J. Cell 14, 191–201 (1979).
Bonner, J. J. Devl Biol. 86, 409–418 (1981).
Craine, B. L. & Kornberg, T. Cell 25, 671–681 (1981).
Parker, C. S. & Topol, J. Cell 37, 273–283 (1984).
Dignam, A. D., Martin, P. L., Shastry, B. S. & Roeder, R. G. Meth. Enzym. 101, 582–598 (1983).
McKenzie, S. L., Henikoff, S. & Meselson, M. Proc. natn. Acad. Sci. U.S.A. 72, 1117–1121 (1975).
Lifton, R. P., Goldberg, M. L., Karp, R. W. & Hogness, D. S. Cold Spring Harb. Symp. quant. Biol. 42, 1047–1051 (1978).
Holmgren, R. thesis, Harvard Univ. (1980).
Craig, E. A. & McCarthy, B. J. Nucleic Acids Res. 8, 4441–4457 (1980).
Corces, V., Holmgren, R., Freund, R., Morimoto, R. & Meselson, M. Proc. natn. Acad. Sci. U.S.A. 77, 5390–5393 (1980).
Ingolia, T. D. & Craig, E. A. Nucleic Acids Res. 9, 1627–1642 (1981).
Southgate, R., Ayme, A. & Voellmy, R. J. molec. Biol. 165, 35–57 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wu, C. Activating protein factor binds in vitro to upstream control sequences in heat shock gene chromatin. Nature 311, 81–84 (1984). https://doi.org/10.1038/311081a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/311081a0
This article is cited by
-
The interaction of heat shock proteins with cellular membranes: a historical perspective
Cell Stress and Chaperones (2021)
-
Role of glutamine, as free or dipeptide form, on muscle recovery from resistance training: a review study
Nutrire (2018)
-
Heat shock factors: integrators of cell stress, development and lifespan
Nature Reviews Molecular Cell Biology (2010)
-
Delivery of HSF1(+) protein using HIV-1 TAT protein transduction domain
Molecular Biology Reports (2009)
-
Signaling through the antigen receptor of B lymphocytes activates a p53-independent pathway of c-Myc-induced apoptosis
Oncogene (1999)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.