Abstract
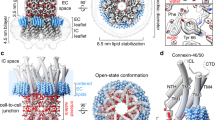
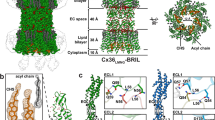
The protein oligomer forming the gap junction channel has been analysed in two Ca2+-sensitive states by electron microscopy of membranes in frozen aqueous solutions. Switching between states occurs by a small cooperative rearrangement involving tilting of the subunits, which may be responsible for the effect of Ca2+ on channel permeability in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Rose, B. & Loewenstein, W. R. Nature 254, 250–252 (1975).
Spray, D. C., Stern, J. H., Harris, A. L. & Bennett, M. V. L. Proc. natn. Acad. Sci. U.S.A. 79, 441–445 (1981).
Makowski, L., Caspar, D. L. D., Phillips, W. C. & Goodenough, D. A. J. Cell Biol. 74, 629–645 (1977).
Unwin, P. N. T. & Zampighi, G. Nature 283, 545–549 (1980).
Hertzberg, E. G., Lawrence, T. S. & Gilula, N. B. A. Rev. Physiol. 43, 479–491 (1981).
Loewenstein, W. R. Physiol. Rev. 61, 829–913 (1981).
Unwin, P. N. T. & Ennis, P. D. J. Cell Biol. 97, 1459–1466 (1983).
Taylor, K. A. & Glaeser, R. M. Science 186, 1036–1037 (1974).
Dubochet, J., Lepault, J., Freeman, R., Berriman, J. A. & Homo, J.-C. J. Microsc. 128, 219–237 (1982).
Henderson, R. & Unwin, P. N. T. Nature 257, 28–32 (1975).
Agard, D. A. J. molec. Biol. 167, 849–852 (1983).
Caspar, D. L. D., Goodenough, D. A., Makowski, L. & Phillips, W. C. J. Cell Biol. 74, 605–628 (1977).
Hertzberg, E. L. & Gilula, N. B. J. biol. Chem. 254, 2138–2147 (1979).
Nicholson, B. J., Hunkapiller, M. W., Grim, L. B., Hood, L. E. & Revel, J.-P. Proc. natn. Acad. Sci. U.S.A. 78, 7594–7598 (1981).
Flagg-Newton, J., Simpson, I. & Loewenstein, W. R. Science 205, 404–407 (1979).
Schwarzmann, G. et al. Science 213, 551–553 (1981).
Durham, A. C. H. & Klug, A. Nature new Biol. 229, 6–10 (1971).
Robinson, I. K. & Harrison, S. C. Nature 297, 563–568 (1982).
Thon, F. Z. Naturforsch. 219, 476–478 (1966).
Jaffe, J. & Glaeser, R. M. Proc. 40th an. EMSA Meet., 72–73 (San Francisco, (1982).
Taylor, K. A., Milligan, R. A., Raeburn, C. & Unwin, P. N. T. Ultramicroscopy (submitted).
Unwin, P. N. T. & Henderson, R. J. molec. Biol. 95, 425–440 (1975).
Baker, T. S., Caspar, D. L. D., Hollingshead, C. J. & Goodenough, D. A. J. Cell Biol. 96, 204–216 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Unwin, P., Ennis, P. Two configurations of a channel-forming membrane protein. Nature 307, 609–613 (1984). https://doi.org/10.1038/307609a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/307609a0
This article is cited by
-
Accessing gap-junction channel structure-function relationships through molecular modeling and simulations
BMC Cell Biology (2017)
-
Atomic structure of the innexin-6 gap junction channel determined by cryo-EM
Nature Communications (2016)
-
An electrostatic mechanism for Ca2+-mediated regulation of gap junction channels
Nature Communications (2016)
-
Krabbe disease: involvement of connexin43 in the apoptotic effects of sphingolipid psychosine on mouse oligodendrocyte precursors
Apoptosis (2016)
-
Structure of the connexin 26 gap junction channel at 3.5 Å resolution
Nature (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.