Abstract
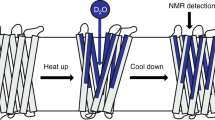
Recently, there has been great interest in determining the three-dimensional structures of membrane proteins, particularly bacteriorhodopsin, for which a variety of possible folding arrangements have been suggested1–4. In this paper we present nuclear magnetic resonance (NMR) spectra5–8 of deuterated bacteriorhodopsin, and use the data to help interpret the various suggested bacteriorhodopsin folding patterns. The results strongly indicate that (1) a membrane surface (±1 residue) may be defined by NMR in bacteriorhodopsin; (2) all amino acids inside the surface are essentially crystalline; (3) all amino acids outside the surface (surface residues) in bacteriorhodopsin are highly mobile on the time scale of the 2H NMR experiments; (4) NMR data may be used to help evaluate the various structural models that have been proposed; (5) aggregation of purple membrane sheets may lead to an immobilization of the surface residues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Engelman, D. M., Henderson, R., McLachlan, A. D. & Wallace, B. A. Proc. natn. Acad. Sci. U.S.A. 77, 2023–2027 (1980).
Ovchinnikov, Y. A. FEBS Lett. 148, 179–191 (1982).
Huang, K.-S., Radhakrishnan, R., Bayley, H. & Khorana, H. G. J. biol. Chem. 257, 13616–13623 (1982).
Engelman, D. M., Goldman, A. & Steitz, T. A. Meth. Enzym. 88, 81–88 (1982).
Kang, S. Y., Gutowsky, H. S. & Oldfield, E. Biochemistry 18, 3268–3271 (1971).
Barnes, R. G. in Advances in Nuclear Quadrupole Resonance (ed. Smith, J. A. S.) Vol 1, 335–355 (Pergamon, New York, 1974).
Oldfield, E., Meadows, M., Rice, D. & Jacobs, R. Biochemistry 17, 2727–2740 (1978).
Oldfield, E. et al. Biochem. Soc. Symp. 46, 155–181 (1980).
Wertz, D. H. & Scheraga, H. A. Macromolecules 11, 9–15 (1978).
Oldfield, E. & Meadows, M. J. mag. Res. 31, 327–335 (1978).
Davis, J. H., Jeffrey, K. R., Bloom, M., Valic, M. I. & Higgs, T. P. Chem. phys. Lett. 42, 390–394 (1976).
Rothgeb, T. M. & Oldfield, E. J. biol. Chem. 256, 1432–1446 (1981).
Berlinquet, L. Can. J. Chem. 33, 1119–1129 (1955).
Sato, M., Okawa, K. & Akabori, S. Bull. Chem. Soc. Japan 30, 937–938 (1957).
Kinsey, R. A., Kintanar, A. & Oldfield, E. J. biol. Chem. 256, 9028–9036 (1981).
Kinsey, R. A. et al. J. biol. Chem. 256, 4146–4149 (1981).
Becher, R. M. & Cassim, J. Y. Prep. Biochem. 5, 161–178 (1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Keniry, M., Gutowsky, H. & Oldfield, E. Surface dynamics of the integral membrane protein bacteriorhodopsin. Nature 307, 383–386 (1984). https://doi.org/10.1038/307383a0
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/307383a0
This article is cited by
-
NMR studies of retinal proteins
Journal of Bioenergetics and Biomembranes (1992)
-
Sequence-specific resonance assignment and secondary structure of (1–71) bacterioopsin
Journal of Biomolecular NMR (1992)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.