Abstract
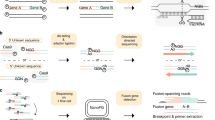
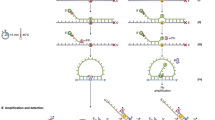
The identification of prognostically relevant fusion genes is required in the routine diagnostic process of most advanced clinical protocols for leukemia patients, either for risk stratification, target-specific treatments, and/or as markers for monitoring Minimal Residual Disease during treatment. However, there is emerging need to implement diagnostics and patient classification based on other biological features, such as expression levels of specific genes or genomic polymorphisms and/or mutations. This advancement would ideally be pursued in a diagnostic laboratory by an unique platform capable of different diagnostic purposes. We developed a rapid, accurate and reproducible assay to screen for the most common fusion gene transcripts in human leukemia, which combines a multiplex RT–PCR approach with the electronic hybridization and fluorescent detection on the Nanogen NanoChip Molecular Biology Workstation. This study demonstrates, as a proof-of-principle, that this microelectronic device, highly effective in detecting single base mutations, is also efficient in the analysis of gene expression, thus providing as a multi-purpose platform for relevant comprehensive diagnostics of hemato-oncology patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Mrozek K, Bloomfield CD . Chromosome aberrations, gene mutations and expression changes, and prognosis in adult acute myeloid leukemia. Hematology Am Soc Hematol Educ Program 2006; 1: 169–177.
Goldman JM, Melo JV . Chronic myeloid leukemia—advances in biology and new approaches to treatment. N Engl J Med 2003; 349: 1451–1464.
Pui C-H, Schrappe M, Ribeiro RC, Niemeyer CM . Childhood and adolescent lymphoid and myeloid leukemia. Hematology Am Soc Hematol Educ Program 2004; 1: 118–145.
Cazzaniga G GG, Rossi V, Biondi A . Minimal residual disease as a surrogate marker for risk assignment to ALL patients. Rev Clin Exp Hematol 2003; 7: 292–323.
Goulden N, Virgo P, Grimwade D . Minimal residual disease directed therapy for childhood acute myeloid leukaemia: the time is now. Br J Haematol 2006; 134: 273–282.
Kern W, Schoch C, Haferlach T, Schnittger S . Monitoring of minimal residual disease in acute myeloid leukemia. Crit Rev Oncol Hematol 2005; 56: 283–309.
van Dongen JJ, Macintyre EA, Gabert JA, Delabesse E, Rossi V, Saglio G et al. Standardized RT-PCR analysis of fusion gene transcripts from chromosome aberrations in acute leukemia for detection of minimal residual disease. Report of the BIOMED-1 Concerted Action: investigation of minimal residual disease in acute leukemia. Leukemia 1999; 13: 1901–1928.
Kern W, Kohlmann A, Schnittger S, Schoch C, Haferlach T . Role of gene expression profiling for diagnosing acute leukemias. Rev Clin Exp Hematol 2005; 9: E1.
Jabbour E, Kantarjian H, Jones D, Talpaz M, Bekele N, O'Brien S et al. Frequency and clinical significance of BCR-ABL mutations in patients with chronic myeloid leukemia treated with imatinib mesylate. Leukemia 2006; 20: 1767–1773.
Tartaglia M, Martinelli S, Cazzaniga G, Cordeddu V, Iavarone I, Spinelli M et al. Genetic evidence for lineage-related and differentiation stage-related contribution of somatic PTPN11 mutations to leukemogenesis in childhood acute leukemia. Blood 2004; 104: 307–313.
Boissel N, Leroy H, Brethon B, Philippe N, de Botton S, Auvrignon A et al. Incidence and prognostic impact of c-Kit, FLT3, and Ras gene mutations in core binding factor acute myeloid leukemia (CBF-AML). Leukemia 2006; 20: 965–970.
de Reynies A, Geromin D, Cayuela JM, Petel F, Dessen P, Sigaux F et al. Comparison of the latest commercial short and long oligonucleotide microarray technologies. BMC Genomics 2006; 7: 51.
Keen-Kim D, Grody WW, Richards CS . Microelectronic array system for molecular diagnostic genotyping: Nanogen NanoChip 400 and molecular biology workstation. Expert Rev Mol Diagn 2006; 6: 287–294.
Gilles PN, Wu DJ, Foster CB, Dillon PJ, Chanock SJ . Single nucleotide polymorphic discrimination by an electronic dot blot assay on semiconductor microchips. Nat Biotechnol 1999; 17: 365–370.
Ferrari M, Cremonesi L, Bonini P, Foglieni B, Stenirri S . Single-nucleotide polymorphism and mutation identification by the nanogen microelectronic chip technology. Methods Mol Med 2005; 114: 93–106.
Beillard E, Pallisgaard N, van der Velden VH, Bi W, Dee R, van der Schoot E et al. Evaluation of candidate control genes for diagnosis and residual disease detection in leukemic patients using ‘real-time’ quantitative reverse-transcriptase polymerase chain reaction (RQ-PCR)—a Europe against cancer program. Leukemia 2003; 17: 2474–2486.
Sosnowski RG, Tu E, Butler WF, O'Connell JP, Heller MJ . Rapid determination of single base mismatch mutations in DNA hybrids by direct electric field control. Proc Natl Acad Sci USA 1997; 94: 1119–1123.
Ferrari F, Foglieni B, Arosio P, Camaschella C, Daraio F, Levi S et al. Microelectronic DNA chip for hereditary hyperferritinemia cataract syndrome, a model for large-scale analysis of disorders of iron metabolism. Hum Mutat 2006; 27: 201–208.
Gabert J, Beillard E, Van Der Velden VH, Bi W, Grimwade D, Pallisgaard N et al. Standardization and quality control studies of ‘real-time’ quantitative reverse transcriptase polymerase chain reaction of fusion gene transcripts for residual disease detection in leukemia—a Europe Against Cancer Program. Leukemia 2003; 17: 2318–2357.
Pallisgaard N, Hokland P, Riishoj DC, Pedersen B, Jorgensen P . Multiplex reverse transcription-polymerase chain reaction for simultaneous screening of 29 translocations and chromosomal aberrations in acute leukemia. Blood 1998; 92: 574–588.
Weidenhammer EM, Kahl BF, Wang L, Duhon M, Jackson JA, Slater M et al. Multiplexed, targeted gene expression profiling and genetic analysis on electronic microarrays. Clin Chem 2002; 48: 1873–1882.
Huang Y, Shirajian J, Schroder A, Yao Z, Summers T, Hodko D et al. Multiple sample amplification and genotyping integrated on a single electronic microarray. Electrophoresis 2004; 25: 3106–3116.
Sosnowski R, Heller MJ, Tu E, Forster AH, Radtkey R . Active microelectronic array system for DNA hybridization, genotyping and pharmacogenomic applications. Psychiatr Genet 2002; 12: 181–192.
Nardi V, Azam M, Daley GQ . Mechanisms and implications of imatinib resistance mutations in BCR-ABL. Curr Opin Hematol 2004; 11: 35–43.
Levine RL, Gilliland DG . JAK-2 mutations and their relevance to myeloproliferative disease. Curr Opin Hematol 2007; 14: 43–47.
Mrózek K, Bloomfield CD . Chromosome aberrations, gene mutations and expression changes, and prognosis in adult acute myeloid leukemia. Hematology Am Soc Hematol Educ Program 2006; 1: 169–177.
Acknowledgements
We are very grateful to Donatella Guardione, Karin Monn, Bjorn Hihn and Wolfgang Trautwein (Nanogen Europe), for technical assistance and fruitful discussion. We thank Fondazione Tettamanti, Fondazione Cariplo, AIRC and MIUR for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Corradi, B., Fazio, G., Palmi, C. et al. Efficient detection of leukemia-related fusion transcripts by multiplex PCR applied on a microelectronic platform. Leukemia 22, 294–302 (2008). https://doi.org/10.1038/sj.leu.2404987
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404987