Abstract
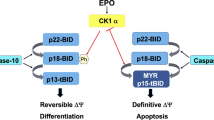
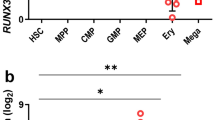
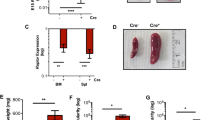
Programmed cell death plays an important role in erythropoiesis under physiological and pathological conditions. In this study, we show that the Notch/RBPjκ signaling pathway induces erythroid apoptosis in different hematopoietic tissues, including yolk sac and bone marrow as well as in murine erythroleukemia cells. In RBPjκ−/− yolk sacs, erythroid cells have a decreased rate of cell death that results in increased number of Ter119+ cells. A similar effect is observed when Notch activity is abrogated by incubation with the γ-secretase inhibitors, DAPT or L685,458. We demonstrate that incubation with Jagged1-expressing cells has a proapoptotic effect in erythroid cells from adult bone marrow that is prevented by blocking Notch activity. Finally, we show that the sole expression of the activated Notch1 protein is sufficient to induce apoptosis in hexametilene-bisacetamide-differentiating murine erythroleukemia cells. Together these results demonstrate that Notch regulates erythroid homeostasis by inducing apoptosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Bray SJ . Notch signalling: a simple pathway becomes complex. Nat Rev Mol Cell Biol 2006; 7: 678–689.
Lai EC . Notch signaling: control of cell communication and cell fate. Development 2004; 131: 965–973.
Kumano K, Chiba S, Kunisato A, Sata M, Saito T, Nakagami-Yamaguchi E et al. Notch1 but not Notch2 is essential for generating hematopoietic stem cells from endothelial cells. Immunity 2003; 18: 699–711.
Gering M, Patient R . Hedgehog signaling is required for adult blood stem cell formation in zebrafish embryos. Dev Cell 2005; 8: 389–400.
Burns CE, Traver D, Mayhall E, Shepard JL, Zon LI . Hematopoietic stem cell fate is established by the Notch–Runx pathway. Genes Dev 2005; 19: 2331–2342.
Robert-Moreno A, Espinosa L, de la Pompa JL, Bigas A . RBPjkappa-dependent Notch function regulates Gata2 and is essential for the formation of intra-embryonic hematopoietic cells. Development 2005; 132: 1117–1126.
Palis J, Robertson S, Kennedy M, Wall C, Keller G . Development of erythroid and myeloid progenitors in the yolk sac and embryo proper of the mouse. Development 1999; 126: 5073–5084.
Palis J, Segel GB . Developmental biology of erythropoiesis. Blood Rev 1998; 12: 106–114.
Mulcahy L . The erythropoietin receptor. Semin Oncol 2001; 28: 19–23.
Silva M, Grillot D, Benito A, Richard C, Nunez G, Fernandez-Luna JL . Erythropoietin can promote erythroid progenitor survival by repressing apoptosis through Bcl-XL and Bcl-2. Blood 1996; 88: 1576–1582.
Motoyama N, Wang F, Roth KA, Sawa H, Nakayama K, Nakayama K et al. Massive cell death of immature hematopoietic cells and neurons in Bcl-x-deficient mice. Science 1995; 267: 1506–1510.
Ganguli G, Back J, Sengupta S, Wasylyk B . The p53 tumour suppressor inhibits glucocorticoid-induced proliferation of erythroid progenitors. EMBO Rep 2002; 3: 569–574.
Peller S, Frenkel J, Lapidot T, Kahn J, Rahimi-Levene N, Yona R et al. The onset of p53-dependent apoptosis plays a role in terminal differentiation of human normoblasts. Oncogene 2003; 22: 4648–4655.
Weiss MJ, Orkin SH . Transcription factor GATA-1 permits survival and maturation of erythroid precursors by preventing apoptosis. Proc Natl Acad Sci USA 1995; 92: 9623–9627.
Ishiko E, Matsumura I, Ezoe S, Gale K, Ishiko J, Satoh Y et al. Notch signals inhibit the development of erythroid/megakaryocytic cells by suppressing GATA-1 activity through the induction of HES1. J Biol Chem 2005; 280: 4929–4939.
Zweidler-McKay PA, He Y, Xu L, Rodriguez CG, Karnell FG, Carpenter AC et al. Notch signaling is a potent inducer of growth arrest and apoptosis in a wide range of B-cell malignancies. Blood 2005; 106: 3898–3906.
Romer S, Saunders U, Jack HM, Jehn BM . Notch1 enhances B-cell receptor-induced apoptosis in mature activated B cells without affecting cell cycle progression and surface IgM expression. Cell Death Differ 2003; 10: 833–844.
Jang MS, Miao H, Carlesso N, Shelly L, Zlobin A, Darack N et al. Notch-1 regulates cell death independently of differentiation in murine erythroleukemia cells through multiple apoptosis and cell cycle pathways. J Cell Physiol 2004; 199: 418–433.
Shelly LL, Fuchs C, Miele L . Notch-1 inhibits apoptosis in murine erythroleukemia cells and is necessary for differentiation induced by hybrid polar compounds. J Cell Biochem 1999; 73: 164–175.
Oka C, Nakano T, Wakeham A, de la Pompa JL, Mori C, Sakai T et al. Disruption of the mouse RBP-J kappa gene results in early embryonic death. Development 1995; 121: 3291–3301.
Friend C, Preisler HD, Scher W . Studies on the control of differentiation of murine virus-induced erythroleukemic cells. Curr Top Dev Biol 1974; 8: 81–101.
Schroeter EH, Kisslinger JA, Kopan R . Notch-1 signalling requires ligand-induced proteolytic release of intracellular domain. Nature 1998; 393: 382–386.
Li L, Milner LA, Deng Y, Iwata M, Banta A, Graf L et al. The human homolog of rat jagged1 expressed by marrow stroma inhibits differentiation of 32d cells through interaction with notch1. Immunity 1998; 8: 43–55.
Lam LT, Ronchini C, Norton J, Capobianco AJ, Bresnick EH . Suppression of erythroid but not megakaryocytic differentiation of human K562 erythroleukemic cells by notch-1. J Biol Chem 2000; 275: 19676–19684.
Xue Y, Gao X, Lindsell CE, Norton CR, Chang B, Hicks C et al. Embryonic lethality and vascular defects in mice lacking the Notch ligand Jagged1. Hum Mol Genet 1999; 8: 723–730.
Krebs LT, Xue Y, Norton CR, Shutter JR, Maguire M, Sundberg JP et al. Notch signaling is essential for vascular morphogenesis in mice. Genes Dev 2000; 14: 1343–1352.
Krebs LT, Shutter JR, Tanigaki K, Honjo T, Stark KL, Gridley T . Haploinsufficient lethality and formation of arteriovenous malformations in Notch pathway mutants. Genes Dev 2004; 18: 2469–2473.
Loken MR, Shah VO, Dattilio KL, Civin CI . Flow cytometric analysis of human bone marrow: I. Normal erythroid development. Blood 1987; 69: 255–263.
Ihle JN, Quelle FW, Miura O . Signal transduction through the receptor for erythropoietin. Semin Immunol 1993; 5: 375–389.
Neubauer H, Cumano A, Muller M, Wu H, Huffstadt U, Pfeffer K . Jak2 deficiency defines an essential developmental checkpoint in definitive hematopoiesis. Cell 1998; 93: 397–409.
Socolovsky M, Nam H, Fleming MD, Haase VH, Brugnara C, Lodish HF . Ineffective erythropoiesis in Stat5a(−/−)5b(−/−) mice due to decreased survival of early erythroblasts. Blood 2001; 98: 3261–3273.
Koury MJ, Bondurant MC . Control of red cell production: the roles of programmed cell death (apoptosis) and erythropoietin. Transfusion 1990; 30: 673–674.
Yu H, Bauer B, Lipke GK, Phillips RL, Van Zant G . Apoptosis and hematopoiesis in murine fetal liver. Blood 1993; 81: 373–384.
Dolznig H, Habermann B, Stangl K, Deiner EM, Moriggl R, Beug H et al. Apoptosis protection by the Epo target Bcl-X(L) allows factor-independent differentiation of primary erythroblasts. Curr Biol 2002; 12: 1076–1085.
Motoyama N, Kimura T, Takahashi T, Watanabe T, Nakano T . bcl-x Prevents apoptotic cell death of both primitive and definitive erythrocytes at the end of maturation. J Exp Med 1999; 189: 1691–1698.
Yang X, Klein R, Tian X, Cheng HT, Kopan R, Shen J . Notch activation induces apoptosis in neural progenitor cells through a p53-dependent pathway. Dev Biol 2004; 269: 81–94.
Huang Q, Raya A, DeJesus P, Chao SH, Quon KC, Caldwell JS et al. Identification of p53 regulators by genome-wide functional analysis. Proc Natl Acad Sci USA 2004; 101: 3456–3461.
Maetens M, Doumont G, De Clercq S, Francoz S, Froment P, Bellefroid E et al. Distinct Roles of Mdm2 and Mdm4 in Red Cell Production. Blood 2007; 109: 2630–2633.
Campbell PJ, Green AR . The myeloproliferative disorders. N Engl J Med 2006; 355: 2452–2466.
Miyazato A, Ueno S, Ohmine K, Ueda M, Yoshida K, Yamashita Y et al. Identification of myelodysplastic syndrome-specific genes by DNA microarray analysis with purified hematopoietic stem cell fraction. Blood 2001; 98: 422–427.
MacKenzie F, Duriez P, Wong F, Noseda M, Karsan A . Notch4 inhibits endothelial apoptosis via RBP-Jkappa-dependent and -independent pathways. J Biol Chem 2004; 279: 11657–11663.
Jehn BM, Bielke W, Pear WS, Osborne BA . Cutting edge: protective effects of notch-1 on TCR-induced apoptosis. J Immunol 1999; 162: 635–638.
Curry CL, Reed LL, Golde TE, Miele L, Nickoloff BJ, Foreman KE . Gamma secretase inhibitor blocks Notch activation and induces apoptosis in Kaposi's sarcoma tumor cells. Oncogene 2005; 24: 6333–6344.
Purow BW, Haque RM, Noel MW, Su Q, Burdick MJ, Lee J et al. Expression of Notch-1 and its ligands, Delta-like-1 and Jagged-1, is critical for glioma cell survival and proliferation. Cancer Res 2005; 65: 2353–2363.
Jundt F, Anagnostopoulos I, Forster R, Mathas S, Stein H, Dorken B . Activated Notch1 signaling promotes tumor cell proliferation and survival in Hodgkin and anaplastic large cell lymphoma. Blood 2002; 99: 3398–3403.
Miller DT, Cagan RL . Local induction of patterning and programmed cell death in the developing Drosophila retina. Development 1998; 125: 2327–2335.
Lundell MJ, Lee HK, Perez E, Chadwell L . The regulation of apoptosis by Numb/Notch signaling in the serotonin lineage of Drosophila. Development 2003; 130: 4109–4121.
Ohishi K, Varnum-Finney B, Flowers D, Anasetti C, Myerson D, Bernstein ID . Monocytes express high amounts of Notch and undergo cytokine specific apoptosis following interaction with the Notch ligand, Delta-1. Blood 2000; 95: 2847–2854.
Qi R, An H, Yu Y, Zhang M, Liu S, Xu H et al. Notch1 signaling inhibits growth of human hepatocellular carcinoma through induction of cell cycle arrest and apoptosis. Cancer Res 2003; 63: 8323–8329.
Acknowledgements
We thank Liheng Li for Jagged1 plasmid, Raphael Kopan for N1ΔE plasmid and M Caudy for the hes1 plasmid. Thanks to I Fabregat and J Gil for extremely helpful suggestions and reagents. Many thanks to L Riera and J Inglés-Esteve for critical comments of the manuscript. We thank E Castaño from Serveis Cientifico-Tècnics, UB-Bellvitge, and J Petriz for flow cytometry technical support. LE is an investigator from the Carlos III program (ISCIII/02/3027). ARM is a recipient of a CIRIT predoctoral fellowship (2002-SI00791). This work was supported by the grants from Ministerio de Educación y Ciencia (SAF2004-03/98) and (SAF205-23978-E) and (SAF2003-07214).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Leukemia website (http://www.nature.com/leu)
Rights and permissions
About this article
Cite this article
Robert-Moreno, À., Espinosa, L., Sanchez, M. et al. The notch pathway positively regulates programmed cell death during erythroid differentiation. Leukemia 21, 1496–1503 (2007). https://doi.org/10.1038/sj.leu.2404705
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2404705
Keywords
This article is cited by
-
Notch and retinoic acid signals regulate macrophage formation from endocardium downstream of Nkx2-5
Nature Communications (2023)
-
A systems biology pipeline identifies regulatory networks for stem cell engineering
Nature Biotechnology (2019)
-
Notch-Hes-1 axis controls TLR7-mediated autophagic death of macrophage via induction of P62 in mice with lupus
Cell Death & Disease (2016)
-
Influence of Jagged1 on apoptosis-related gene expression: a microarray database analysis
Genes & Genomics (2015)
-
Notch1 regulates progenitor cell proliferation and differentiation during mouse yolk sac hematopoiesis
Cell Death & Differentiation (2014)