Abstract
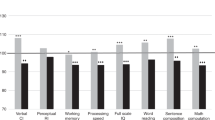
Little is known about the incidence of and risk factor for late effects of infant leukemia. We evaluated 19 children with acute lymphoblastic leukemia and 15 with acute myeloid leukemia who were diagnosed at age 12 months or younger and have survived for more than 5 years after the diagnosis (median length of follow-up, 13 years; range, 5.7–29 years). Ten patients received chemotherapy alone (group A), 17 received chemotherapy and CNS-directed radiation therapy (CRT) (group B), and seven received chemotherapy, CRT and bone marrow transplantation (group C). The most frequently observed late sequelae included problems in growth (66% of survivors), learning (50%), hypothyroidism (15%), and pubertal development (12%). Cataract, cardiac and hearing abnormalities occurred in 6% of patients. Only eight patients (24%) survive without late effects. In comparison to patients in group A, patients in groups B and C had a higher incidence of having at least one late complication (P = 0.009), a greater decrease in height Z score at 5 years after diagnosis (P = 0.023), and a higher incidence of academic difficulties (P = 0.004). The estimated odds of academic difficulties increased by 18% (P = 0.032) for each month younger in age at the time of CRT. These results indicate that late sequelae are common in long-term survivors of infant leukemia and are often related to CRT and the patient's age at the time of CRT.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Pui CH . Childhood leukemia New Engl J Med 1995 332: 1618–1630
Pui CH, Kane JR, Crist WM . Biology and treatment of infant leukemia Leukemia 1995 9: 762–769
Felix CA, Lange BJ . Leukemia in infants Oncologist 1999 4: 225–240
Pui CH, Ribeiro RC, Campana D, Raimondi SC, Hancock ML, Behm FG, Sandlund JT, Rivera GK, Evans WE, Crist WM, Krance R . Prognostic factors in the acute lymphoid and myeloid leukemia of infants Leukemia 1996 10: 952–956
Pui CH, Behm FG, Downing JR, Hancock ML, Shurtleff SA, Ribeiro RC, Head DR, Mahmoud HH, Sandlund JT, Furman WL, Roberts WM, Crist WM, Raimondi SC . 11q23/MLL rearrangement confers a poor prognosis in infants with acute lymphoblastic leukemia J Clin Oncol 1994 12: 909–915
Reaman GH, Sposto R, Sensel MG, Lange BJ, Feusner JH, Heerema NA, Leonard M, Holmes EJ, Sather HN, Pendergrass TW, Johnstone HS, O'Brien RT, Steinherz PG, Zeltzer PM, Gaynon PS, Trigg ME, Uckun FM . Treatment outcome and prognostic factors for infants with acute lymphoblastic leukemia treated on two consecutive trials of the Children's Cancer Group J Clin Oncol 1999 17: 445–455
Dordelmann M, Reiter A, Borkhardt A, Ludwig WD, Gotz N, Viehmann S, Gadner H, Riehm H, Schrappe M . Prednisone response is the strongest predictor of treatment outcome in infant acute lymphoblastic leukemia Blood 1999 94: 1209–1217
Pui CH, Raimondi SC, Srivastava DK, Tong X, Behm FG, Razzouk B, Rubnitz JE, Sandlund JT, Evans WE, Ribeiro R . Prognostic factors in infants with acute myeloid leukemia Leukemia 2000 14: 684–687
McLeod HL, Relling MV, Crom WR, Silverstein K, Groom S, Rodman JH, Rivera GK, Crist WM, Evans WE . Disposition of antineoplastic agents in the very young child Br J Cancer 1992 66: (Suppl.18) S23–29
Pui CH, Kalwinsky DK, Schell MJ, Mason CA, Mirro J Jr, Dahl GV . Acute nonlymphoblastic leukemia in infants: clinical presentation and outcome J Clin Oncol 1988 6: 1008–1013
Aur RJ, Hustu HO, Verzosa MS, Wood A, Simone JV . Comparison of two methods of preventing central nervous system leukemia Blood 1973 42: 349–357
Aur RJ, Simone JV, Verzosa MS, Hustu HO, Barker LF, Pinkel DP, Rivera G, Dahl GV, Wood A, Stagner S, Mason C . Childhood acute lymphocytic leukemia: study VIII Cancer 1978 42: 2123–2134
Pui CH, Aur RJ, Bowman WP, Dahl GV, Dodge RK, George SL, Ochs J, Kalwinsky DK, Abromowitch M, Hustu HO . Failure of late intensification therapy to improve a poor result in childhood lymphoblastic leukemia Cancer Res 1984 44: 3593–3598
Pui CH, Simone JV, Hancock ML, Evans WE, Williams DL, Bowman WP, Dahl GV, Dodge RK, Ochs J, Abromowitch M, Rivera GK . Impact of three methods of treatment intensification on acute lymphoblastic leukemia in children: long-term results of St Jude total therapy study X Leukemia 1992 6: 150–157
Rivera GK, Raimondi SC, Hancock ML, Behm FG, Pui CH, Abromowitch M, Mirro J Jr, Ochs JS, Look AT, Williams DL . Improved outcome in childhood acute lymphoblastic leukaemia with reinforced early treatment and rotational combination chemotherapy Lancet 1991 337: 61–66
Evans WE, Relling MV, Rodman JH, Crom WR, Boyett JM, Pui CH . Conventional compared with individualized chemotherapy for childhood acute lymphoblastic leukemia New Engl J Med 1998 338: 499–505
Pui CH, Mahmoud HH, Rivera GK, Hancock ML, Sandlund JT, Behm FG, Head DR, Relling MV, Ribeiro RC, Rubnitz JE, Kun LE, Evans WE . Early intensification of intrathecal chemotherapy virtually eliminates central nervous system relapse in children with acute lymphoblastic leukemia Blood 1998 92: 411–415
Ribeiro RC, Rivera GK, Hudson M, Mulhern RK, Hancock ML, Kun L, Mahmoud H, Sandlund JT, Crist WM, Pui CH . An intensive re-treatment protocol for children with an isolated CNS relapse of acute lymphoblastic leukemia J Clin Oncol 1995 13: 333–338
Relling MV, Mahmoud HH, Pui CH, Sandlund JT, Rivera GK, Ribeiro RC, Crist WM, Evans WE . Etoposide achieves potentially cytotoxic concentrations in CSF of children with acute lymphoblastic leukemia J Clin Oncol 1996 14: 399–404
Dahl GV, Kalwinsky DK, Murphy S, Look AT, Amadori S, Kumar M, Novak R, George SL, Mason C, Mauer AM, Simone JV . Cytokinetically based induction chemotherapy and splenectomy for childhood acute nonlymphocytic leukemia Blood 1982 60: 856–863
Dahl GV, Kalwinsky DK, Mirro J Jr, Look AT, Pui CH, Murphy SB, Mason C, Ruggiero M, Schell M, Johnson FL, Thomas ED . Allogeneic bone marrow transplantation in a program of intensive sequential chemotherapy for children and young adults with acute nonlymphocytic leukemia in first remission J Clin Oncol 1990 8: 295–303
Kalwinsky D, Mirro J Jr, Schell M, Behm F, Mason C, Dahl GV . Early intensification of chemotherapy for childhood acute nonlymphoblastic leukemia: improved remission induction with a five-drug regimen including etoposide J Clin Oncol 1988 6: 1134–1143
Hurwitz CA, Krance R, Schell MJ, Santana VM, Brenner MK, Ribeirio R, Roberts WM, Mahmoud H, Belt J, Crom W . Current strategies for treatment of acute myeloid leukemia at St Jude Children's Research Hospital Leukemia 1992 6: (Suppl.2) 39–43
Marshall WA, Tanner JM . Variations in pattern of pubertal changes in girls Arch Dis Child 1969 44: 291–303
Marshall WA, Tanner JM . Variations in the pattern of pubertal changes in boys Arch Dis Child 1970 45: 13–23
Tanner JM, Davies PS . Clinical longitudinal standards for height and height velocity for North American children J Pediatr 1985 107: 317–329
Mulhern RK, Kovnar E, Langston J, Carter M, Fairclough D, Leigh L, Kun LE . Long-term survivors of leukemia treated in infancy: factors associated with neuropsychologic status J Clin Oncol 1992 10: 1095–1102
Mulhern RK, Fairclough D, Ochs J . A prospective comparison of neuropsychologic performance of children surviving leukemia who received 18-Gy, 24-Gy, or no cranial irradiation J Clin Oncol 1991 9: 1348–1356
Kruskal WH, Wallis WA . Use of ranks in one-criterion variance analysis J Am Stat Assoc 1952 47: 583–621
Mehta CR, Patel NR . A network algorithm for performing Fisher's exact test in r×c contingency tables J Am Stat Assoc 1983 78: 427–434
Tritchler D . An algorithm for exact logistic regression J Am Stat Assoc 1984 79: 709–711
Schell MJ, Ochs JJ, Schriock EA, Carter M . A method of predicting adult height and obesity in long-term survivors of childhood acute lymphoblastic leukemia J Clin Oncol 1992 10: 128–133
Craig F, Leiper AD, Stanhope R, Brain C, Meller ST, Nussey SS . Sexually dimorphic and radiation dose dependent effect of cranial irradiation on body mass index Arch Dis Child 1999 81: 500–504
Reaman G, Zeltzer P, Bleyer W, Amendola B, Level C, Sather H, Hammond D . Acute lymphoblastic leukemia in infants less than one year of age: a cumulative experience of the Children's Cancer Study Group J Clin Oncol 1985 3: 1513–1521
Silverman LB, McLean TW, Gelber RD, Donnelly MJ, Gilliland DG, Tarbell NJ, Sallan SE . Intensified therapy for infants with acute lymphoblastic leukemia: results from the Dana-Farber Cancer Institute Consortium Cancer 1997 80: 2285–2295
Ferster A, Bertrand Y, Benoit Y, Boilletot A, Behar C, Margueritte G, Thyss A, Robert A, Mazingue F, Souillet G . Improved survival for acute lymphoblastic leukaemia in infancy: the experience of EORTC-Childhood Leukaemia Cooperative Group Br J Haematol 1994 86: 284–290
Waber DP, Tarbell NJ, Fairclough D, Atmore K, Castro R, Isquith P, Lussier F, Romero I, Carpenter PJ, Schiller M . Cognitive sequelae of treatment in childhood acute lymphoblastic leukemia: cranial radiation requires an accomplice J Clin Oncol 1995 13: 2490–2496
Kaleita TA, Reaman GH, MacLean WE, Sather HN, Whitt JK . Neurodevelopmental outcome of infants with acute lymphoblastic leukemia: a Children's Cancer Group report Cancer 1999 85: 1859–1865
Acknowledgements
We thank James M Boyett, PhD, for his helpful comments, Patricia Harrison for exact logistic regression analysis, the staff of the Department of Behavioral Medicine for psychological evaluations, Julia C Jones, PhD, for scientific editing, Lisa Edwards, Sherri Patterson, and Margie Zacher for data management, the physicians and staff who provide medical care, and the patients and families who completed the questionnaires and participated in this study. This work was supported in part by grants CA-20180 and CA-21765 from the National Institutes of Health, by a Center of Excellence grant from the State of Tennessee, and by the American Lebanese Syrian Associated Charities (ALSAC).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Leung, W., Hudson, M., Zhu, Y. et al. Late effects in survivors of infant leukemia. Leukemia 14, 1185–1190 (2000). https://doi.org/10.1038/sj.leu.2401818
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.leu.2401818
Keywords
This article is cited by
-
Late effects in survivors of infant acute lymphoblastic leukaemia—a study of the Australian and New Zealand Children’s Haematology/Oncology Group
Blood Cancer Journal (2023)
-
Acute Leukemia in Infants
Current Oncology Reports (2021)
-
Late effects in survivors of infantile acute leukemia: a study of the L.E.A program
Blood Cancer Journal (2017)
-
Incidence and severity of crucial late effects after allogeneic HSCT for malignancy under the age of 3 years: TBI is what really matters
Bone Marrow Transplantation (2016)
-
The evolution of clinical trials for infant acute lymphoblastic leukemia
Blood Cancer Journal (2014)