Abstract
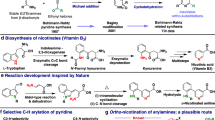
THE earlier confusion regarding the structure of vitamin A2 was removed by its synthesis1, which conclusively proved it to have the dehydrovitamin A1 formula suggested by Morton, Salah and Stubbs2. In this synthesis an extra double bond was introduced into the β-ionone ring of the methylester of vitamin A1 acid, by bromination with N-bromosuccinimide, followed by dehydrobromination with 4-phenyl morpholine. Reduction with lithium aluminium hydride gave a product which closely agreed with the spectroscopic properties of vitamin A2. Recently, Henbest, Jones and Owen3 carried out an elegant conversion of vitamin A1 to A2 by treating the aldehyde of vitamin A1 (retinene1) with N-bromosuccinimide, to obtain retinene2, which by reduction with lithium aluminium hydride was converted to vitamin A2.
Similar content being viewed by others
Article PDF
References
Jones, E. R. H., et al., J. Chem. Soc., 2657 (1952).
Morton, R. A., Salah, M. K., and Stubbs, A. L., Nature, 159, 744 (1947).
Henbest, H. B., et al., J. Chem. Soc., 4909 (1957); 2763 (1955).
Ball, S., Goodwin, T. W., and Morton, R. A., Biochem. J., 42, 516 (1948).
Wald, G. J. Gen. Physiol., 31, 489 (1947).
Attenburrow, J., et al., J. Chem. Soc., 1094 (1952).
Barua, R. K., and Morton, R. A., Biochem. J., 45, 308 (1948).
Cama, H. R., Dalvi, P. D., Morton, R. A., Salah, M. K., Steinberg, G. R., and Stubbs, A. L., Biochem. J., 52, 535 (1952).
Cama, H. R., Dalvi, P. D., Morton, R. A., and Salah, M. K., Biochem. J., 52, 540, 542 (1952).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
BARUA, R., NAIR, M. A New Synthesis of Vitamin A2 : Conversion of Vitamin A1 to A2. Nature 193, 165 (1962). https://doi.org/10.1038/193165a0
Issue Date:
DOI: https://doi.org/10.1038/193165a0
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.