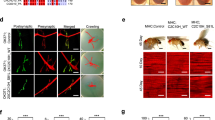
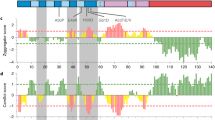
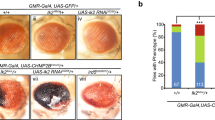
Abstract
Presenilins are membrane proteins with multiple transmembrane domains that are thought to contribute to the development of Alzheimer's disease by affecting the processing of β-amyloid precursor protein1. Presenilins also facilitate the activity of transmembrane receptors of the LIN-12/Notch family2,3,4,5. After ligand-induced processing, the intracellular domain of LIN-12/Notch can enter the nucleus and participate in the transcriptional control of downstream target genes6,7,8,9. Here we show that null mutations in the Drosophila Presenilin gene abolish Notch signal transduction and prevent its intracellular domain from entering the nucleus. Furthermore, we provide evidence that presenilin is required for the proteolytic release of the intracellular domain from the membrane following activation of Notch by ligand.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Selkoe, D. J. The cell biology of beta-amyloid precursor protein and presenilin in Alzheimer's disease. Trends Cell Biol. 8, 447–453 (1998).
Levitan, D. & Greenwald, I. Facilitation of lin-12-mediated signalling by sel-12, a Caenorhabditis elegans S182 Alzheimer's disease gene. Nature 377, 351–354 (1995).
Li, X. & Greenwald, I. HOP-1, a Caenorhabditis elegans presenilin, appears to be functionally redundant with SEL-12 presenilin and to facilitate LIN-12 and GLP-1 signalling. Proc. Natl Acad. Sci. USA 94, 12204–12209 (1997).
Shen, J. et al. Skeletal and CNS defects in Presenilin-1 deficient mice. Cell 89, 629–639 (1997).
Wong, P. C. et al. Presenilin 1 is required for Notch1 and Dll1 expression in the paraxial mesoderm. Nature 387, 288–292 (1997).
Struhl, G. & Adachi, A. Nuclear access and action of Notch in vivo. Cell 93, 649–660 (1998).
Schroeter, E. H., Kisslinger, J. A. & Kopan, R. Notch-1 signalling requires ligand-induced proteolytic release of intracellular domain. Nature 393, 381–386 (1998).
Lecourtois, M. & Schweisguth, F. Indirect evidence for Delta-dependent intracellular processing of Notch in Drosophila embryos. Curr. Biol. 8, 771–774 (1998).
Kidd, S., Lieber, T. & Young, M. W. Ligand-induced cleavage and regulation of nuclear entry of Notch in Drosophila melanogaster embryos. Genes Dev. 12, 3728–3740 (1998).
DeStrooper, B. et al. Deficiency of presenilin-1 inhibits the normal cleavage of amyloid precursor protein. Nature 391, 387–380 (1998).
Greenwald, I. LIN-12/Notch signaling: lessons from worms and flies. Genes Dev. 12, 1751–1762 (1998).
Blaumueller, C. M., Qui, H., Zagouras, P. & Artavanis-Tsakonas, S. Intracellular cleavage of Notch leads to a heterodimeric receptor on the plasma membrane. Cell 90, 281–291 (1997).
Logeat, F. et al. the Notch1 receptor is cleaved constitutively by a furin-like convertase. Proc. Natl Acad. Sci. USA 95, 8108–8112 (1998).
Levitan, D. & Greenwald, I. Effects of SEL-12 presenilin on LIN-12 localization and function in C. elegans. Development 125, 3599–3606 (1998).
Boulianne, G. L. et al. Cloning and characterization of the Drosophila presenilin homologue. Neuroreport 8, 1025–1029 (1997).
Hong, C. S. & Koo, E. H. Isolation and characterization of Drosophila presenilin homolog. Neuroreport 8, 665–668 (1997).
Marfany, G. et al. Identification of a Drosophila presenilin homologue: evidence of alternatively spliced forms. J. Neurogenet. 12, 41–45 (1993).
Struhl, G., Fitzgerald, K. & Greenwald, I. Intrinsic activity of the Lin-12 and Notch intracellular domains in vivo. Cell 74, 331–45 (1993).
Menne, T. V. & Klaembt, C. The formation of commissures in the Drosophila CNS depends on the midline cells and on the Notch gene. Development 120, 123–133 (1994).
Blair, S. S. Compartments and appendage development in Drosophila. Bioessays 17, 299–309 (1995).
Huppert, S. S., Jacobsen, T. L. & Muskavitch, M. Feedback regulation is central to Delta-Notch signalling required for Drosophila wing vein morphogenesis. Development 124, 3283–3291 (1997).
Ye, Y. & Fortini, M. E. Characterization of Drosophila Presenilin and its colocalization with Notch during development. Mech. Dev. 79, 199–211 (1998).
Li, X. & Greenwald, I. Additional evidence for an eight-transmembrane-domain topology for Caenorhabditis elegans and human presenilins. Proc. Natl Acad. Sci. USA 95, 7109–7114 (1998).
Chou, T. B. & Perrimon, N. Use of a yeast site-specific recombinase to produce female germline chimeras in Drosophila. Genetics 131, 643–653 (1992).
Golic, K. G. Site-specific recombination between homologous chromosomes in Drosophila. Science 252, 958–961 (1991).
Rulifson, E. J. & Blair, S. S. Notch regulates wingless expression and is not required for reception of the paracrine wingless signal during wing margin neurogenesis in Drosophila. Development 121, 2813–2824 (1995).
Greenwald, I. & Seydoux, G. Analysis of gain-of-function mutations of the lin-12 gene of Caenorhabditis elegans. Nature 346, 197–1999 (1990).
Heitzler, P. & Simpson, P. Altered epidermal growth factor-like sequences provide evidence for a role of Notch as a receptor in cell fate decisions. Development 117, 1113–1123 (1993).
Acknowledgements
We thank J. Jiang and C.-M. Chen for the isolation and preliminary characterization of the PS alleles as neurogenic mutations; A. Adachi, R.Perez and X.-J. Qin for technical assistance; S. T. Crews, T. Lieber and P. M. Macdonald for antisera; and R. Axel, D. Brower, S.-K. Chan, H.-M. Chung and M. Zecca for advice and discussion. G.S. is an investigator and I.G. is an associate investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Struhl, G., Greenwald, I. Presenilin is required for activity and nuclear access of Notch in Drosophila. Nature 398, 522–525 (1999). https://doi.org/10.1038/19091
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/19091
This article is cited by
-
The advent of Alzheimer treatments will change the trajectory of human aging
Nature Aging (2024)
-
A tailored tetravalent peptide displays dual functions to inhibit amyloid β production and aggregation
Communications Biology (2023)
-
Ubiquitylation is required for the incorporation of the Notch receptor into intraluminal vesicles to prevent prolonged and ligand-independent activation of the pathway
BMC Biology (2022)
-
Regulation of gene expression by the APP family in the adult cerebral cortex
Scientific Reports (2022)
-
Essential roles of plexin-B3+ oligodendrocyte precursor cells in the pathogenesis of Alzheimer’s disease
Communications Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.