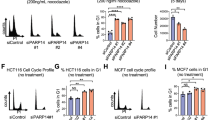
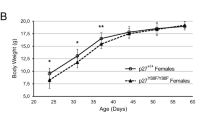
Abstract
The proliferation of mammalian cells is under strict control, and the cyclin-dependent-kinase inhibitory protein p27Kip1 is an essential participant in this regulation both in vitro and in vivo 1. Although mutations in p27Kip1 are rarely found in human tumours, reduced expression of the protein correlates well with poor survival among patients with breast or colorectal carcinomas2, suggesting that disruption of the p27Kip1 regulatory mechanisms contributes to neoplasia. The abundance of p27Kip1 in the cell is determined either at or after translation3, for example as a result of phosphorylation by cyclinE/Cdk2 complexes4,5, degradation by the ubiquitin/proteasome pathway6, sequestration by unknown Myc-inducible proteins7, binding to cyclinD/Cdk4 complexes8, or inactivation by the viral E1A oncoprotein9. We have found that a mouse 38K protein (p38) encoded by the Jab1 gene10 interacts specifically with p27Kip1 and show here that overexpression of p38 in mammalian cells causes the translocation of p27Kip1 from the nucleus to the cytoplasm, decreasing the amount of p27Kip1 in the cell by accelerating its degradation. Ectopic expression of p38 in mouse fibroblasts partially overcomes p27Kip1-mediated arrest in the G1 phase of the cell cycle and markedly reduces their dependence on serum. Our findings indicate that p38 functions as a negative regulator of p27Kip1 by promoting its degradation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sherr, C. J. Cancer cell cycles. Science 274, 1672–1677 (1996).
Steeg, P. S. & Abrams, J. S. Cancer prognostics: Past, present and p27. Nature Med. 3, 152–154 (1997).
Hengst, L. & Reed, S. I. Translational control of p27Kip1 accumulation during the cell cycle. Science 271, 1861–1864 (1996).
Sheaff, R. J., Groudine, M., Gordon, M., Roberts, J. M. & Clurman, B. E. Cyclin E-CDK2 is a regulator of p27Kip1. Genes Dev. 11, 1464–1478 (1997).
Vlach, J., Hennecke, S. & Amati, B. Phosphorylation-dependent degradation of the cyclin-dependent kinase inhibitor p27Kip1. EMBO J. 16, 5334–5344 (1997).
Pagano, M. et al. Role of the ubiquitin-proteasome pathway in regulating abundance of the cyclin-dependent kinase inhibitor p27. Science 269, 682–685 (1995).
Vlach, J., Hennecke, S., Alevizopoulos, K., Conti, D. & Amati, B. Growth arrest by the cyclin-dependent kinase inhibitor p27Kip1 is abrogated by c-Myc. EMBO J. 15, 6595–6604 (1996).
Polyak, K. et al. p27Kip1, a cyclin-Cdk inhibitor, links transforming growth factor-β and contact inhibition to cell cycle arrest. Genes Dev. 8, 9–22 (1994).
Mal, A. et al. Inactivation of p27Kip1 by the viral E1A oncoprotein in TGFβ-treated cells. Nature 380, 262–265 (1996).
Claret, F. X. et al. Anew group of conserved coactivators that increase the specificity of AP-1 transcription factors. Nature 383, 453–457 (1996).
Durfee, T. et al. The retinoblastoma protein associates with the protein phosphatase type 1 catalytic subunit. Genes Dev. 7, 555–569 (1993).
Polyak, K. et al. Cloning of p27Kip1, a cyclin-dependent kinase inhibitor and a potential mediator of extracellular antimitogenic signals. Cells 78, 59–66 (1994).
Hofmann, K. & Bucher, P. The PCI domain: a common theme in three multiprotein complexes. Trends Biochem. Sci. 23, 204–205 (1998).
Asano, K. et al. Structure of cDNAs encoding human eukaryotic initiation factor 3 subunits. Possible roles in RNA binding and macromolecular assembly. J. Biol. Chem. 272, 27042–27052 (1997).
Matsuoka, S. et al. p57Kip2, a structurally distinct member of the p21Cip1 Cdk inhibitor family, is a candidate tumor suppressor gene. Genes Dev. 9, 650–662 (1995).
Ogawa, H., Inouye, S., Tsuji, F. I., Yasuda, K. & Umesono, K. Localization, trafficking, and temperature-dependence of the Aequorea green fluorescent protein in cultured vertebrate cells. Proc. Natl Acad. Sci. USA 92, 11899–11903 (1995).
Rock, K. L. et al. Inhibitors of the proteasome block the degradation of most cell proteins and the generation of peptides presented on MHC class I molecules. Cell 78, 761–771 (1994).
Nishi, K. et al. Leptomycin B targets a regulatory cascade of crm1, a fission yeast nuclear protein, involved in control of higher order chromosome structure and gene expression. J. Biol. Chem. 269, 6320–6324 (1994).
Fukuda, M. et al. CRM1 is responsible for intracellular transport mediated by the nuclear export signal. Nature 390, 308–311 (1997).
Fukuda, M., Gotoh, I., Adachi, M., Gotoh, Y. & Nishida, E. Anovel regulatory mechanism in the mitogen-activated protein (MAP) kinase cascade. Role of nuclear export signal of MAP kinase kinase.. J. Biol. Chem. 272, 32642–32648 (1997).
Coats, S., Flanagan, W. M., Nourse, J. & Roberts, J. M. Requirement of p27Kip1 for restriction point control of the fibroblast cell cycle. Science 272, 877–880 (1996).
Seeger, M. et al. Anovel protein complex involved in signal transduction possessing similarities to 26S proteasome subunits. FASEB J. 12, 469–478 (1998).
Wei, N. et al. The COP9 complex is conserved between plants and mammals and is related to the 26S proteasome regulatory complex. Curr. Biol. 8, 919–922 (1998).
Glickman, M. H. et al. Asubcomplex of the proteasome regulatory particle required for ubiquitin-conjugate degradation and related to the COP9-signalosome and eIF3. Cell 94, 615–623 (1998).
Harper, J. W., Adami, G., Wei, N., Keyomarsi, K. & Elledge, S. J. The 21 kd Cdk interacting protein Cip1 is a potent inhibitor of G1 cyclin-dependent kinases. Cell 75, 805–816 (1993).
Hirai, H., Roussel, M. F., Kato, J.-y., Ashmun, R. A. & Sherr, C. J. Novel INK4 proteins, p19 and p18, are specific inhibitors of the cyclin D-dependent kinases CDK4 and CDK6. Mol. Cell. Biol. 15, 2672–2681 (1995).
Akamatsu, E., Tanaka, T. & Kato, J.-y. Transcription factor E2F and cyclin E/cdk2 complex cooperate to induce chromosomal DNA replication in Xenopus oocytes. J. Biol. Chem. 273, 16494–16500 (1998).
Kato, J.-y., Matsushime, H., Hiebert, S. W., Ewen, M. E. & Sherr, C. J. Direct binding of cyclin D to the retinoblastoma gene product (pRb) and pRb phosphorylation by the cyclin D-dependent kinase, CDK4. Genes Dev. 7, 331–342 (1993).
Tanaka, M. & Herr, W. Differential transcriptional activation by Oct-1 and Oct-2: interdependent activation domains induce Oct-2 phosphorylation. Cell 60, 375–386 (1990).
Chen, C. & Okayama, H. High-efficiency transformation of mammalian cells by plasmid DNA. Mol. Cell. Biol. 7, 2745–2752 (1987).
Acknowledgements
We thank C. J. Sherr, J. Fujisawa, D. O. Morgan, F. Claret, K. Umesono, M. Yoshida and N. Kato for materials, and N. Sanechika for construction of p27 mutants. This work was supported by grants-in-Aid for Scientific Research and for Cancer Research from the Ministry of Education, Science, and Culture of Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tomoda, K., Kubota, Y. & Kato, Jy. Degradation of the cyclin-dependent-kinase inhibitor p27Kip1 is instigated by Jab1. Nature 398, 160–165 (1999). https://doi.org/10.1038/18230
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/18230
This article is cited by
-
Regulatory mechanisms and therapeutic potential of JAB1 in neurological development and disorders
Molecular Medicine (2023)
-
Prognostic value of CSN5 in patients with digestive system cancers: a systematic review and meta-analysis
BMC Cancer (2022)
-
Prognostic and therapeutic significance of COP9 signalosome subunit CSN5 in prostate cancer
Oncogene (2022)
-
Neurogenesis and Proliferation of Neural Stem/Progenitor Cells Conferred by Artesunate via FOXO3a/p27Kip1 Axis in Mouse Stroke Model
Molecular Neurobiology (2022)
-
Ubiquitin signaling in cell cycle control and tumorigenesis
Cell Death & Differentiation (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.