Summary:
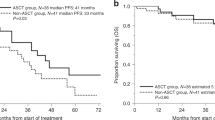
The role of autologous stem cell transplantation (AuSCT) in older multiple myeloma patients is unclear. Using data from the Autologous Blood and Marrow Transplant Registry, we compared the outcome of 110 patients ⩾the age of 60 (median 63; range 60–73) years, undergoing AuSCT with that of 382 patients <60 (median 52; range 30–59) years. The two groups were similar except that older patients had a higher β2-microglobulin level at diagnosis (P=0.016) and fewer had lytic lesions (P=0.007). Day 100 mortality was 6% (95% confidence interval 4–9) and 1-year treatment-related mortality (TRM) was 9% (6–13) in patients <60 years, compared with 5% (2–10) and 8% (4–14), respectively, in patients ⩾60 years. The relapse rate, progression-free survival (PFS) and overall survival (OS) in the two groups were also similar. Multivariate analysis of all patients identified only an interval from diagnosis to AuSCT >12 months and the use of two prior chemotherapy regimens within 6 months of AuSCT as adverse prognostic factors. Our results indicate that AuSCT can be safely performed in selected older patients: the best results were observed in patients undergoing AuSCT relatively early in their disease course.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Attal M, Harousseau JL, Stoppa AM et al. A prospective randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. N Engl J Med 1996; 335: 91–97.
Barlogie B, Jagannath S, Vesole DH et al. Superiority of tandem autologous transplantation over standard therapy for previously untreated multiple myeloma. Blood 1997; 89: 789–793.
Lenhoff S, Hjorth M, Holmberg E et al. Impact on survival of high dose therapy with autologous stem cell transplant support in patients younger than 60 years with newly diagnosed multiple myeloma: a population-based study. Nordic Myeloma Study Group. Blood 2000; 95: 7–11.
Harousseau JL, Attal M, Divine M et al. Autologous stem cell transplantation after first remission induction treatment in multiple myeloma: a report of the French registry on autologous transplantation in multiple myeloma. Blood 1995; 85: 3077–3085.
Leger CS, Bredeson C, Kewarns B et al. Autologous blood and marrow transplantation in patients 60 years and older. Biol Blood Marrow Transplant 2000; 6: 204–210.
De la Rubia J, Sadverdra S, Sauz GF et al. Transplant-related mortality in patients older than 60 years undergoing autologous hematopoietic stem cell transplantation. Bone Marrow Transplant 2001; 27: 21–25.
Palumbo A, Triolo S, Argentino C et al. Dose-intensive melphalan with stem cell support (MEL 100) is superior to standard treatment in elderly myeloma patient's blood. Blood 1999; 94: 1248–1253.
Siegel DS, Desikan KR, Mehta J et al. Age is not a prognostic variable with autotransplants for multiple myeloma. Blood 1999; 93: 51–54.
Badros A, Barlogie B, Siegel E et al. Autologous stem cell transplantation in elderly multiple myeloma patients over the age of 70 years. Br J Haematol 2001; 114: 600–607.
Sirohi B, Powles R, Treleaven J et al. The role of autologous transplantation in patients with multiple myeloma aged 65 years and over. Bone Marrow Transplant 2000; 25: 533–539.
Dumontet C, Ketterer N, Espinouse D et al. Reduced progression-free survival in elderly patients receiving intensification with autologous peripheral blood stem cell vein fusion for multiple myeloma. Bone Marrow Transplant 1998; 21: 1037–1041.
Blade J, Samson D, Reece D et al. Criteria for evaluating disease response and progression in patients with multiple myeloma treated by high-dose therapy and hematopoietic stem cell transplantation. Br J Haematol 1998; 102: 1115–1123.
Gooley A, Leisenring W, Crowley J et al. Estimation of failure probabilities in the presence of competing risks: new representations of old estimators. Stat Med 1999; 18: 695–706.
Klein JP, Moeschberger ML . Survival Analysis: Techniques for Censored and Truncated Data. Springer-Verlag: New York, NY, 1997.
Andersen PK, Klein JP, Zhang MH . Testing for center effects in multi-centre survival studies: a Monte Carlo comparison of fixed and random effects tests. Stat Med 1999; 18: 1489–1500.
Vesole DH, Tricot G, Jagannath S et al. Autotransplants in multiple myeloma: what have we learned? Blood 1996; 88: 838–847.
Moreau P, Facon T, Attal M et al. Comparison of 200 mg/m2 melphalan and 8 Gy total body irradiation plus 140 mg/m2 as conditioning regimens for peripheral blood stem cell transplantation in patients with newly diagnosed multiple myeloma. Final analysis of the IFM 95-02 randomized trial. Blood 2002; 99: 731–735.
Bjorkstrand B, Svensson G, Goldschmidt H et al. Autotransplants in multiple myeloma: a registry from the European Group for Blood and Marrow Transplantation (EBMT). Blood 1999; 94 (Suppl 1): 714a (abstract 3154).
Cunningham D, Powles R, Malpas J et al. A randomized trial of maintenance interferon following high-dose chemotherapy in multiple myeloma: long-term follow-up results. Br J Haematol 1999; 102: 495–502.
Berenson JR, Crowley JJ, Grogan TM et al. Maintenance therapy with alternate-day prednisone improves survival in multiple myeloma patients. Blood 2002; 99: 3163–3168.
Alexanian R, Weber D, Giralt S et al. Consolidation therapy of multiple myeloma with thalidomide-dexamethasone after intensive chemotherapy. Ann Oncol 2002; 13: 1116–1119.
Rufffini PA, Kwak LW . Immunotherapy of multiple myeloma. Sem Hematol 2001; 38: 260–267.
Hideshima T, Chuhan D, Podar K et al. Novel therapies targeting the myeloma cell and its bone marrow microenvironment. Semin Oncol 2001; 28: 607–612.
Maloney DG, Sahebi F, Stockerl-Goldstein KE et al. Combining an allogeneic graft-vs-myeloma effect with high-dose autologous stem cell rescue in the treatment of multiple myeloma. Presentation at the American Society of Hematology 43rd Annual Meeting and Exposition. Blood 2001; 98 (Part 1 of 2) 434a–435a (abstract 1822).
Acknowledgements
This work was supported by Public Health Service Grant U24-CA76518 from the National Cancer Institute, the National Institute of Allergy and Infectious Diseases, and the National Heart, Lung and Blood Institute, and grants from Abgenix, Inc.; AmCell Corporation; American Cancer Society; American Society of Clinical Oncology; Amgen, Inc.; Anonymous; Aventis Pharmaceuticals; Berlex Laboratories; BioTransplant, Incorporated; Blue Cross and Blue Shield Association; Lynde and Harry Bradley Foundation; Bristol-Myers Squibb Oncology; CelMed Biosciences; Center for Advanced Studies in Leukemia; Cerus Corporation; Chimeric Therapies; Chiron Therapeutics; Eleanor Naylor Dana Charitable Trust; Deborah J Dearholt Memorial Fund; Edwards Lifesciences RMI; Empire Blue Cross Blue Shield; Fujisawa Healthcare, Inc.; Gambro BCT, Inc.; Genentech, Inc.; GlaxoSmithKline, Inc.; Human Genome Sciences; ICN Pharmaceuticals, Inc.; IDEC Pharmaceuticals Corporation; IntraBiotics Pharmaceuticals; Kettering Family Foundation; Kirin Brewery Company; Robert J Kleberg, Jr and Helen C Kleberg Foundation; LifeTrac/Allianz; Eli Lilly and Company; The Liposome Company; Nada and Herbert P Mahler Charities; Market Certitude, LLC; Mayer Ventures; MedImmune, Inc.; Merck & Co., Inc.; Milliman & Robertson, Inc.; Milstein Family Foundation; The Greater Milwaukee Foundation/Elsa Schoeneich Research Fund; NeoRx; Nexell Therapeutics; Novartis Pharmaceuticals; Orphan Medical; Ortho Biotech, Inc.; John Oster Family Foundation; Osiris Therapeutics, Inc.; Pfizer US Pharmaceuticals; Pharmacia Corporation; Pharmametrics GmbH; Principal Life Insurance Company; Protein Design Labs, Inc.; Response Oncology, Inc.; RGK Foundation; Roche Laboratories, Inc.; SangStat; Schering AG; Schering Oncology/Biotech; Stackner Family Foundation; The Starr Foundation; SuperGen, Inc.; TheraTechnologies, Inc.; Unicare Life & Health Insurance; Wyeth/Genetics Institute and ZymoGenetics, Inc. The contents of this article are solely the responsibility of the authors and do not necessarily represent the official views of the National Cancer Institute.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Reece, D., Bredeson, C., Pérez, W. et al. Autologous stem cell transplantation in multiple myeloma patients <60 vs ⩾60 years of age. Bone Marrow Transplant 32, 1135–1143 (2003). https://doi.org/10.1038/sj.bmt.1704288
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1704288
Keywords
This article is cited by
-
The influence of high-efficiency particulate air filtration on mortality among multiple myeloma patients receiving autologous stem cell transplantation
Scientific Reports (2021)
-
Outcomes with autologous stem cell transplant vs. non-transplant therapy in patients 70 years and older with multiple myeloma
Bone Marrow Transplantation (2021)
-
Autologous stem cell transplantation in elderly patients with multiple myeloma in Korea: the KMM1807 study
International Journal of Hematology (2020)
-
High-dose therapy and autologous stem cell transplant in older adults with multiple myeloma
Bone Marrow Transplantation (2015)
-
Defining and treating high-risk multiple myeloma
Leukemia (2015)