Abstract
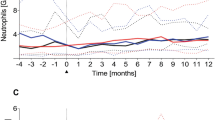
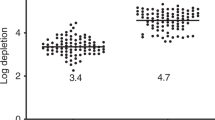
The present study examines the nature of humoral and cellular immune reconstitution in 28 patients with advanced breast cancer following high-dose chemotherapy with stem cell rescue. Patients underwent testing of T, B, NK and dendritic cell function at serial time points until 1 year post transplant or until the time of disease progression. Abnormalities in T cell phenotype and function were observed following high-dose chemotherapy that persisted for at least 6–12 months. The vast majority of patients experienced an inversion of the CD4/CD8 ratio and demonstrated an anergic response to candida antigen. Mean T cell proliferation in response to PHA and to co-culture with allogeneic monocytes was significantly compromised. In contrast, mean IgG and IgA levels were normal 6 months post transplant and NK cell yields and function were transiently elevated following high-dose chemotherapy. Dendritic cells generated from peripheral blood progenitors displayed a characteristic phenotype and were potent inducers of allogeneic T cell proliferation in the post-transplant period. The study demonstrates that patients undergoing autologous transplantation for breast cancer experience a prolonged period of T cell dysfunction. In contrast, B, NK, and DC recover more rapidly. These findings carry significant implications for the design of post-transplant immunotherapy. Bone Marrow Transplantation (2000) 26, 169–176.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Lazarus HM . Hematopoietic progenitor cell transplantation in breast cancer: current status and future directions Cancer Invest 1998 16: 1–25
Antman KH, Rowlings PA, Vaughan WP et al. High-dose chemotherapy with autologous hematopoietic stem-cell support for breast cancer in North America J Clin Oncol 1997 15: 1870–1879
Guillaume T, Rubinstein DB, Symann M . Immune reconstitution and immunotherapy after autologous hematopoietic stem cell transplantation Blood 1998 92: 1471–1490
Atkinson K . Reconstruction of the haemopoietic and immune systems after marrow transplantation Bone Marrow Transplant 1990 5: 209–226
Lum LG . The kinetics of immune reconstitution after human marrow transplantation Blood 1987 69: 369–380
Witherspoon RP . Suppression and recovery of immunologic function after bone marrow transplantation J Natl Cancer Inst 1986 76: 1321–1324
Ochs L, Shu XO, Miller J et al. Late infections after allogeneic bone marrow transplantation: comparison of incidence in related and unrelated donor transplant recipients Blood 1995 86: 3979–3986
Small TN . Immunologic reconstitution following stem cell transplantation Curr Opin Hematol 1996 3: 461–465
Aucouturier P, Barra A, Intrator L et al. Long lasting IgG subclass and antibacterial polysaccharide antibody deficiency after allogeneic bone marrow transplantation Blood 1987 70: 779–785
Sugita K, Soiffer RJ, Murray C et al. The phenotype and reconstitution of immunoregulatory T cell subsets after T cell depleted allogeneic and autologous bone marrow transplantation Transplantation 1994 57: 1465
Koehne G, Zeller W, Stockschlaeder M et al. Phenotype of lymphocyte subsets after autologous peripheral blood stem cell transplantation Bone Marrow Transplant 1997 19: 149–156
Foot ABM, Potter MN, Donaldson C et al. Immune reconstitution after BMT in children Bone Marrow Transplant 1993 11: 7–13
Witherspoon RP, Storb R . Stem cell reconstitution after bone marrow transplantation into adult patients does not resemble T cell development in early life Bone Marrow Transplant 1995 16: 413–425
Olsen GA, Gockerman JP, Bast RC et al. Altered immunologic reconstitution after standard-dose chemotherapy or high dose chemotherapy with autologous bone marrow support Transplantation 1988 46: 57–60
Bengtsson M, Totterman TH, Smedymyr B et al. Regeneration of functional and activated NK and T subset cells in the marrow and blood after autologous bone marrow transplantation: a prospective phenotypic study with 2/3-color FACS analysis Leukemia 1989 3: 68–75
Miller RA, Daley J, Ghalie R et al. Clonal analysis of T cell deficiencies in autotransplant recipients Blood 1991 77: 1845–1850
Sugita K, Nojima Y, Tachibana K et al. Prolonged impairment of very late activating antigen-mediated T cell proliferation via the CD3 pathway after T cell depleted allogeneic bone marrow transplantation J Clin Invest 1994 94: 481–488
Cayeux S, Meuer S, Pezzutto A et al. T cell ontogeny after autologous bone marrow transplantation: failure to synthesize interleukin 2 (IL-2) and lack of CD2- and CD3-mediated proliferation by both CD4+ and CD8+ cells even in the presence of exogenous IL-2 Blood 1989 74: 2270–2277
Welte K, Keever CA, Levick J et al. Interleukin-2 production and response to interleukin-2 by peripheral blood mononuclear cells from patients after bone marrow transplantation: II. Patients receiving soybean lectin-separated and T cell depleted bone marrow Blood 1987 70: 1595–1603
Kiesel S, Pezzutto A, Moldenhauer G et al. B cell proliferative and differentiative responses after autologous peripheral blood stem cell or bone marrow transplantation Blood 1988 72: 672–678
Brenner MK, Wimperis JZ, Reittie JE et al. Recovery of immunoglobulin isotypes following T cell depleted allogeneic bone marrow transplantation Br J Haematol 1986 64: 125–132
Steinman RM . The dendritic system and its role in immunogenicity Ann Rev Immunol 1991 9: 271–296
Young JW, Steinman RM . The hematopoietic development of dendritic cells. A distinct pathway for myeloid differentiation Stem Cells 1996 14: 376–387
Young JW, Kouliva L, Soergel SA et al. The B7/BB1 antigen provides one of several costimulatory signals for the activation of CD4+ T lymphocytes by human blood dendritic cells in vitro J Clin Invest 1992 90: 229–237
Inaba K, Witmer-Pack M, Inaba M et al. The tissue distribution of the B7–2 costimulator in mice: abundant expression on dendritic cells in situ and during maturation in vitro J Exp Med 1994 180: 1849–1860
Caux C, Vanbervliet B, Massacrier C et al. B70/B7–2 is identical to CD86 and is the major functional ligand for CD28 expressed on human dendritic cells J Exp Med 1994 180: 1841–1847
Zitovogel L, Mayordomo JI, Tjandrawan T et al. Therapy of murine tumors with tumors peptide-pulsed dendritic cells: dependence on T cells, B7 costimulation, and T helper cell 1-associated cytokines J Exp Med 1996 183: 87–97
Gong JL, Chen D, Chen D et al. Induction of antigen specific antitumor immunity with adenoviral transduced dendritic cells Gene Therapy 1997 4: 1023–1028
Hsu FJ, Benike C, Fagnoni F et al. Vaccination of patients with B-cell lymphoma using autologous antigen-pulsed dendritic cells Nature Med 1996 2: 52–58
Gong J, Avigan D, Chen D et al. Activation of antitumor cytotoxic T lymphocytes by fusions of human dendritic cells and breast carcinoma cells Proc Natl Acad Sci USA 2000 97: 2715–2718
Celluzzi CM, Mayordomo C, Storkus WJ et al. Peptide-pulsed dendritic cells induce antigen specific CTL-mediated protective tumor immunity J Exp Med 1996 183: 283–287
Mayordomo JI, Zorina T, Storkus WJ et al. Bone marrow derived dendritic cells pulsed with synthetic tumor peptides elicit protective and therapeutic antitumor immunity Nature Med 1995 1: 1297–1302
Paglia P, Chiodoni C, Rodolfo M et al. Murine dendritic cells loaded in vitro with soluble protein prime cytotoxic T lymphocytes against tumor antigen in vivo J Exp Med 1996 183: 317–322
Song W, Kong HL, Carpentar H et al. Dendritic cells genetically modified with an adenovirus vector encoding the cDNA for a model antigen induce protective and therapeutic antitumor immunity J Exp Med 1997 186: 1247–1256
Nestle FO, Alijagic S, Gilliet M et al. Vaccination of melanoma patients with peptide-or tumor lysate-pulsed dendritic cells Nature Med 1998 4: 328–332
Antman K, Ayash L, Elias A et al. A phase II study of high-dose cyclophosphamide, thiotepa and carboplatin with autologous marrow support in women with measurable advanced breast cancer responding to standard-dose therapy J Clin Oncol 1992 10: 102–110
Sandmaier BM, Oparin DV, Holmberg LA et al. Evidence of a cellular immune response against sialyl-Tn in breast and ovarian cancer patients after high dose chemotherapy, stem cell rescue and immunization with theraptope STn-KLH cancer vaccine J Immunother 1999 22: 54–66
Acknowledgements
I would like to thank Courtney Ives for assistance in preparation of the manuscript. The research was supported in part by a grant from the Immunex Corporation.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Avigan, D., Wu, Z., Joyce, R. et al. Immune reconstitution following high-dose chemotherapy with stem cell rescue in patients with advanced breast cancer. Bone Marrow Transplant 26, 169–176 (2000). https://doi.org/10.1038/sj.bmt.1702474
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1702474
Keywords
This article is cited by
-
Development of T cell-mediated immunity after autologous stem cell transplantation: prolonged impairment of antigen-stimulated production of γ-interferon
Bone Marrow Transplantation (2007)
-
Immunity for tumors and microbes after autotransplantation: if you build it, they will (not) come
Bone Marrow Transplantation (2006)
-
T-cell reconstitution and expansion after hematopoietic stem cell transplantation: ‘T’ it up!
Bone Marrow Transplantation (2005)
-
Immune restoration following hematopoietic stem cell transplantation: an evolving target
Bone Marrow Transplantation (2005)
-
Resolution of late-onset asthma following high-dose chemotherapy
Bone Marrow Transplantation (2003)