Abstract
Obesity is highly prevalent among patients with schizophrenia and is associated with detrimental health consequences. Although excessive consumption of fast food and pharmacotherapy with such second-generation antipsychotic agents (SGAs) as clozapine and olanzapine has been implicated in the schizophrenia/obesity comorbidity, the pathophysiology of this link remains unclear. Here, we propose a mechanism based on brain reward function, a relevant etiologic factor in both schizophrenia and overeating. A comprehensive literature search on neurobiology of schizophrenia and of eating behavior was performed. The collected articles were critically reviewed and relevant data were extracted and summarized within four key areas: (1) energy homeostasis, (2) food reward and hedonics, (3) reward function in schizophrenia, and (4) metabolic effects of the SGAs. A mesolimbic hyperdopaminergic state may render motivational/incentive reward system insensitive to low salience/palatability food. This, together with poor cognitive control from hypofunctional prefrontal cortex and enhanced hedonic impact of food, owing to exaggerated opioidergic drive (clinically manifested as pain insensitivity), may underlie unhealthy eating habits in patients with schizophrenia. Treatment with SGAs purportedly improves dopamine-mediated reward aspects, but at the cost of increased appetite and worsened or at least not improved opiodergic capacity. These effects can further deteriorate eating patterns. Pathophysiological and therapeutic implications of these insights need further validation via prospective clinical trials and neuroimaging studies.
Similar content being viewed by others
INTRODUCTION
Obesity has reached pandemic proportions and it is rapidly surpassing smoking as a number one killer in the industrialized world (Sturm, 2002; Skidmore and Yarnell, 2004). Its annual cost to the American society is staggering, and is estimated to be around $117 billion owing to related illnesses and loss of productivity (National Institute of Diabetes and Digestive and Kidney Diseases, 2005).
In schizophrenia, obesity is twice as prevalent as in the general public, afflicting over half this patient population (Allison et al, 1999a; Dixon et al, 2000; American Diabetes Association, 2004; Marder et al, 2004; Wirshing, 2004). Besides negative psychosocial impacts (distorted self-esteem and societal stigmatization) and medications noncompliance, schizophrenics appear to be particularly susceptible to the detrimental medical sequelae of obesity such as the ‘Metabolic Syndrome’, which is a cluster of cardiovascular risk factors, including abdominal adiposity, insulin resistance, impaired, glucose tolerance, dyslipidemia, and hypertension (McKee et al, 1986; Mukherjee et al, 1996; Brown et al, 2000; Haupt and Newcomer, 2002; Ryan and Thakore, 2002; Ryan et al, 2003; Holt et al, 2004; Kohen, 2004; Marder et al, 2004; Lieberman et al, 2005).
Excessive body weight gain (BWG) could be attributed to a constellation of endocrine, molecular, genetic, demographic, and lifestyle-related factors. Provided that a common trait may be unhealthy eating, this review considers the dietary aspects of obesity that are pertinent to schizophrenia. First, we provide clinical context on the association between schizophrenia and consumption of the foodstuffs implicated in the development of BWG. Next are discussed the putative neuroanatomy and neurochemistry of mechanisms involved in the regulation of food intake and reward, as they relate to ‘reward deficiency’ state (Comings and Blum, 2000) and unhealthy eating patterns (Peet, 2004a) associated with schizophrenia.
Subsequent sections compare the data on the effects of food, addictive substances, and second-generation antipsychotic agents (SGAs) on homeostasis- and reward-related function, and this comparison is taken as a basis for the analysis of the role of the latter in worsening of BWG. The last subsection covers specific background for a testable hypothesis on alleviation of these emerging abnormalities via augmentation of the SGAs with opioid receptor antagonists such as naltrexone. As this review draws heavily upon preclinical research, it is important to highlight many uncertainties inherent in extrapolating from animal data cited here to schizophrenia (or any other neuropsychiatric syndrome) such as absence of well-defined pathognomonic neurobiological abnormalities and sole reliance on behavioral symptoms and subjective self-reports for the illness diagnosis (Lieberman, 2000). More objective phenotypic (eg, neuroimaging) or genotypic (eg, gene markers or single-nucleotide polymorphisms analyses) tests would provide further evidence on the pathophysiological mechanisms underlying metabolic disturbances in patients with schizophrenia.
SEARCH TERMS AND METHODOLOGY
English language literature search of reward function in schizophrenia, mechanisms of food hedonics and reward and metabolic effects of the SGAs was undertaken using PubMed. Keywords used included schizophrenia plus obesity, sugar, opioid, fat, food, palatable, fast food, eating, appetite, pain, hunger and reward. Each specific SGA was combined with opioid, reward, appetite, hunger, pain and obesity terms. Data on reward, homeostatic mechanisms and SGAs’ metabolic effects were also drawn from recent seminal reviews of these topics (Grace 2000; Koob and Le Moal, 2001; Saper et al, 2002; Berridge and Robinson, 2003; Wise, 2004; Kelley, 2004a, 2004b; Kalra and Kalra, 2004; Nestler, 2004; Newcomer, 2005; Volkow and Wise, 2005; Kalivas and Volkow, 2005; Hyman, 2005; Kalivas et al, 2005; Schwartz and Porte, 2005). Additional strategies included manual searches for relevant articles from the selected papers’ reference lists as well as utilization of PubMed's related articles function.
WEIGHT STATUS OF DRUG NAIVE/FREE SCHIZOPHRENIC PATIENTS
Obesity and associated pathologic conditions (eg, insulin resistance, impaired glucose tolerance, and dyslipidemia) tend to be more prevalent (Finney, 1989; Ryan et al, 2003, 2004), albeit not universally present (Allison et al, 1999a; Zhang et al, 2004) in patients with schizophrenia. The basic mechanisms behind these metabolic irregularities have not been fully elucidated, but it appears that antipsychotic medications could play an important role (Newcomer, 2005). Moreover, high rates of obesity and type II diabetes mellitus, observed in drug-naive/free patients (Mukherjee et al, 1996; Allison et al, 1999a; Thakore et al, 2002; Ryan et al, 2003, 2004) before (Haupt and Newcomer, 2002; Kohen, 2004) and after the advent of antipsychotics and in nonschizophrenic blood relatives (Dynes, 1969; Mukherjee et al, 1989; Cheta et al, 1990; Martins et al, 2001; Lamberti et al, 2004), were potentially attributed to genetic factors (Stone et al, 2004), illness neurobiology (Thakore, 2005) and to unhealthy lifestyle (Brown et al, 1999). The interpretability of the preneuroleptic era data (reviewed in Kohen, 2004) is, however, limited by flaws in epidemiological methodology including lack of evaluation of and adjustments for adiposity, lifestyle, and anthropometric measures together with inconsistent diagnostic criteria for schizophrenia and glucose/insulin abnormalities (Haupt and Newcomer, 2002; Newcomer, 2005).
Seeking to tackle serious methodological limitations of the early research, Thakore et al (2002) applied a well-validated technique of computed tomography (Mayo-Smith et al, 1989) to directly measure abdominal adiposity in a sample of drug-naive/free schizophrenic patients and age- and sex-matched healthy controls. They found that schizophrenic patients had higher body mass index (BMI) and over three times more abdominal fat (Thakore et al, 2002), accumulation of which poses even greater cardiovascular/diabetes risks than overall obesity (Wang et al, 2005). A similar magnitude of increases in abdominal adiposity was observed in a tandem study where drug naive, first episode schizophrenic subjects (N=19) were matched to healthy controls in terms of BMI, age, gender, exercise, smoking, and alcohol use (Ryan et al, 2004).
Although generally consistent with yet another observation by the same group (Ryan et al, 2003), these results have not been replicated in an ethnically distinct sample of drug-naive Chinese patients where increased obesity and adiposity was not detected using low field strength (0.2 Tesla) magnetic resonance imaging (Zhang et al, 2004). This apparent discrepancy in the studies’ outcomes underscores the importance of socioeconomic, cultural, and ethnic variables for the development of excessive weight and abdominal fat deposits. Replication of both groups (Thakore et al, 2002; Zhang et al, 2004) findings may be, however, warranted for obtaining more conclusive evidence because of several confounds including hypercortisolemia and older age (that could reflect a longer duration of untreated psychosis) in the participants of the Thakore et al studies (Thakore et al, 2002; Ryan et al, 2003, 2004) as well as use of a nonstandardized imaging protocol and of unmatched schizophrenics/controls samples for the determination of abdominal adiposity by Zhang et al (2004). Additionally, correction for geographic region-specific normative values will be required in future studies as average BMIs tend to be lower in Chinese vs Western population (Cheng, 2005).
DIETARY HABITS OF SCHIZOPHRENIC PATIENTS
Obesity and adiposity represent a complex pathophysiological condition that could result from increased caloric intake (Strassnig et al, 2003a, 2003b) and decreased energy expenditure owing to reduced physical activity (Strassnig et al, 2003a) or a combination of both (Peet, 2004a). Dietary intake, a relatively easily modifiable (Brar et al, 2005) behavioral phenomenon, could provide a rich framework for preventive/therapeutic clinical interventions.
A rather scarce (relatively to the scope of the problem) literature on schizophrenic eating habits describes exaggerated preference for unhealthy predominately fast food-type (Peet, 2004a, 2004c; Strassnig et al, 2005) palatable nutrition, poor in fiber (Brown et al, 1999; McCreadie et al, 1998), in fruits and in vegetables (McCreadie et al, 1998, 2005; McCreadie, 2003), but excessive in saturated fat (Brown et al, 1999; Strassnig et al, 2003a, 2005; Ryan et al, 2003, 2004), in carbohydrates (Strassnig et al, 2003b), and in high glycemic index (Foster-Powell et al, 2002) foodstuffs such as cereal (McCreadie, 2003). These patterns may be further worsened by treatment with SGAs such as clozapine, olanzapine, and to lesser degree quetiapine and risperidone, but not ziprasidone or aripiprazole (Allison et al, 1999b; American Diabetes Association, 2004; Kane et al, 2004; Newcomer 2005).
The pathophysiological significance of these findings remains unclear. Environmental factors play an obvious role as patients are frequently uninformed about dietary issues (Meyer, 2002) and are unable to access/afford healthy food choices (Holt et al, 2004). They also tend to exercise less (Brown et al, 1999; Strassnig et al, 2005), and therefore even slight excess in caloric intake could result in the accumulation of fat.
In this article, we propose a complementary view that in addition to lifestyle-related factors and similar to its comorbidity with substance use disorders (SUDs; Chambers et al, 2001), schizophrenia and unhealthy food preferences may be potentially explained by recursive partly shared neural systems. Direct evidence for such a neuropathological link is missing. There is, however, consistent (McIntosh and Lawrie, 2004) correlational evidence at a population level revealing a link (not causal-effect relations) between high dietary levels of refined sugar (Peet, 2004b) and of saturated fat (Christensen and Christensen, 1988; Gupta, 1992; Peet, 2004b) with the illness poor prognosis (Christensen and Christensen, 1988; Gupta, 1992), including increased length of hospitalizations and severe social deficit symptoms (Peet, 2004b).
These correlational findings were obtained from two types of international databases. The first contains dietary information, that is, national dietary report provided by the Food and Agriculture Organization of the United Nations (2002). The second source involves three large-scale multicenter surveys of various schizophrenia outcome measures, namely the International Pilot Study of Schizophrenia (World Health Organization, 1979; Gupta, 1992; Peet, 2004b), the Determinants of Outcomes of Severe Mental Disorders (Jablensky et al, 1992; Gupta, 1992; Peet, 2004b), and the World Health Organization International Follow-up Study (World Health Organization, 1979; Christensen and Christensen, 1988). Although the reasons behind transcultural differences in schizophrenia outcomes detected in these and other studies (Kulhara, 1994; Sartorius et al, 1996) have received less attention than the outcomes themselves, it may be relevant that, taken as a whole, schizophrenia prognosis tends to be substantially worse in the developed Western nations, in comparison to the developing countries, that have not yet adopted the Westernized diet (Peet, 2004a).
To gain insight into this entity, research must isolate the interaction of schizophrenia with voracious appetite for food containing high quantities of sugar and fat. One such possible overlap area is the neural pathways mediating reward (subjective experience of pleasure) and reinforcement (increased rate of recurrent actions), a relevant etiologic factor in both schizophrenia (Green et al, 1999; Chambers et al, 2001; Brady and Sinha, 2005) and eating behavior (Wang et al, 2001, 2002, 2003, 2004). The following sections discuss energy balance systems and how they intersect with the brain reward and reinforcement mechanisms.
NEUROBIOLOGY OF SYSTEMS MEDIATING CALORIC BALANCE
Eating is imperative for continued existence of all living organisms (Maslow, 1943), and even brief (days) starvation leads to detrimental physical and psychological changes (Phillips, 1994; Fessler, 2003). Hence, eating behavior is insured via powerful neural systems responsible for the maintenance of caloric balance and for the rewarding nature of food intake. These systems are schematically overviewed in Figures 1 and 2, respectively.
Schematic overview of the metabolic, endocrine, and neural pathways involved in the homeostatic control of eating behavior (ie, maintenance of energy balance). Peripheral adiposity- and nutrient-related signals converge on the level of the medial hypothalamus (other CNS sites, for example, nucleus of the solitary tract and the area postrema are also involved but are not represented here). Fasting or decrease in body energy stores and in circulating nutrients, signaled by gastric hormone, ghrelin, and/or by hypothalamic intracellular agent, AMP-activated protein kinase, promote anabolic processes (ie, appetite and conservation of energy) and restrain catabolic processes via hypothalamic orexigenic pathways. Food intake, conversely, increases body fat content and plasma levels of the nutrients’ metabolites (eg, glucose and free fatty acids; FFA) and triggers (among other metabolic events) release of catabolic hormones, insulin (from pancreas), and leptin (from adipose tissues). These hormones, respectively, suppress and activate hypothalamic orexigenic and anorexigenic networks, thus inhibiting further eating and enhancing energy expenditure. The neural orexigenic networks, originating in the arcuate nucleus (ARC) of the hypothalamus and projecting to the paraventricular nucleus (PVN), coexpress neuropeptide, gamma-aminobutyric acid, and agouti-related protein. The anorexigenic networks also project from ARC to PVN and are comprised of neurons coreleasing pro-opiomelanocortin, alpha-melanocyte-stimulating hormone, brain-derived neurotrophic factor, and cocaine- and amphetamine-regulated transcript (Gerozissis, 2004; Kalra and Kalra, 2004; Saper et al, 2002; Schwartz and Porte, 2005). Orexin and melanin-concentrating hormone (MCH) from neurons in the lateral hypothalamus (LH) stimulate the orexigenic networks; ventromedial nucleus (VMN) exerts an opposite effect potentially via histaminergic mechanisms (Mercer et al, 1994; Sakata et al, 1997). Histamine also has an important role in catabolic processes by mediating the effects of leptin (Masaki et al, 2004), which promotes its release (Itateyama et al, 2003). Other important anorexigenic factors include serotonin (5HT) via 5HT1b, 5HT2A and 5HT2C receptors (Makarenko et al, 2002; Muller et al, 2004) and corticotropin-releasing hormone (CRH; Kalra et al, 1999), whereas orexigenic processes are favored by endogenous opioid peptides (Horvath et al, 1992; Kalra et al, 1999; Morley, 1987) and glucocorticoids (Kalra et al, 1999). Lastly, dopamine in the dorsal striatum (basal ganglia) is involved in nonhedonic homeostatic drive and goal-directed motor behavior towards procurement of food (Cannon et al, 2004; Volkow et al, 2002c). For the clarity of presentation, the scheme was rendered out-of-scale and simplified to reduce the numbers of the displayed links and structures to those of direct relevance to the main themes of this review.
Schematic overview of the ‘motivational system’ for ingestive behavior (Swanson, 2000; Kelley, 2004a) formed from hierarchically organized clusters of hypothalamic and brainstem nuclei (behavioral control column) along with the associated corticolimbic regions. Hypothalamus plays the critical role in the maintenance of body energy balance (see Figure 1), whereas brainstem mediates orosensory sensations and motor function involved in the act of eating, which is ‘powered’ through motivational states and emotions generated by the corticolimbic reward circuitry. The major interface between the homeostatic, motivational/emotional, and motor components is located within the lateral hypothalamus (LH; DiLeone et al, 2003; Saper et al, 2002). This area is involved in reinforcement (Berridge and Valenstein, 1991; Wise, 1996) and is interconnected with the major hypothalamic nuclei (ie, ARC and PVN) implicated in the metabolic sensing (Kalra and Kalra, 2004) and with the brainstem nuclei, mediating eating-related motor function and autonomic activity. Pleasure and positive affective states produced by consumption of palatable food, that is, food ‘liking’ is conveyed to the frontotemporal cortical structures (Berridge, 2003; Kelley, 2004b) through μ-opioid neurotransmission within the network of subcortical and brain stem nuclei (Berridge, 2003; Berridge and Robinson, 2003; Berthoud, 2004b; Kelley, 2004b; Pecina et al, 2003; Saper et al, 2002; Tanda and Di Chiara, 1998; Will et al, 2004), including the nucleus accumbens (NAc), vental tegmental area (VTA), ventral pallidum (VP), nucleus of the solitary tract (NTS), parabrachial nucleus (PBN), and the amygdala (Amy). Mesolimbic dopaminergic pathways projecting from the VTA to the NAc, along with the LH, Amy and prefrontal cortex (PFC; Goldstein and Volkow, 2002; Volkow et al, 2004a) are responsible for food ‘wanting’ (Kelley, 2004b; Kelley and Berridge, 2002; Pecina et al, 2003). The liking and wanting together constitute full reward system (Berridge, 2004). Although these are dissociable phenomena, they are closely linked as liking food usually evokes wanting and eventually eating it (Berthoud, 2004a). Food reward is modulated by a variety of peripheral signals such as nutrition and adiposity, smell, taste, orosensory, and visual stimuli along with environmental factors (eg, food composition and availability, media effects, and social situations). More specifically, metabolic signals of nutrient metabolites and of adiposity, that is, insulin and leptin decrease rewarding value of food (and other rewards) by binding insulin and leptin receptors, in the NAc, LH, and VTA (Figlewicz, 2003a, 2003b, 2004). For the clarity of presentation the scheme was rendered out-of-scale and simplified to reduce the numbers of the displayed neuroanatomical/neurochemical links and structures to those of direct relevance to the main themes of this review.
As depicted in Figure 1, peripheral adiposity- and nutrient-related signals converge on the level of the medial hypothalamus (Schwartz et al, 2000). Fasting or decrease in body energy stores and in circulating nutrients, signaled by gastric hormone, ghrelin, and/or by hypothalamic intracellular agent, AMP-activated protein kinase, promote anabolic processes (ie, hunger and conservation of energy) and restrain catabolic processes (ie, satiety signals) via hypothalamic orexigenic pathways (Schwartz and Porte, 2005). Food intake, conversely, increases body fat content and plasma levels of the nutrients’ metabolites (eg, glucose and free fatty acids) and triggers (among other metabolic events) release of catabolic hormones, insulin (from pancreas) and leptin (from adipose tissues). These hormones, respectively, suppress and activate hypothalamic orexigenic and anorexigenic networks, thus inhibiting further eating and enhancing energy expenditure (Morton et al, 2005; Schwartz and Porte, 2005). In sum, brain metabolic control apparatus is tightly regulated via antagonistic ying- and yang-type processes (Wynne et al, 2005).
The neural orexigenic networks, originating in the arcuate nucleus (ARC) of the hypothalamus and projecting to the paraventricular nucleus (PVN), coexpress neuropeptide Y, gamma-aminobutyric acid, and agouti-related protein (AgRP). The anorexigenic networks also project from ARC to PVN and are comprised of neurons coreleasing proopiomelanocortin, alpha-melanocyte-stimulating hormone, brain-derived neurotrophic factor (BDNF) and cocaine- and amphetamine-regulated transcript (Saper et al, 2002; Gerozissis, 2004; Kalra and Kalra, 2004; Schwartz and Porte, 2005). Orexin and melanin concentrating hormone (MCH) from neurons in the lateral hypothalamus (LH) stimulate the orexigenic networks; ventromedial nucleus exerts an opposite effect via histaminergic mechanisms (Mercer et al, 1994; Sakata et al, 1997). Histamine also has an important role in catabolic processes by mediating the effects of leptin (Masaki et al, 2004) that promotes histamine release (Itateyama et al, 2003). Other important anorexigenic factors include serotonin (via 5HT1b, 5HT2A, and 5HT2C receptors) and corticotropin-releasing hormone (CRH; Kalra et al, 1999; Makarenko et al, 2002; Muller et al, 2004), whereas orexigenic processes are favored by endogenous opiate peptides (Morley, 1987; Horvath et al, 1992; Kalra et al, 1999) and glucocorticoids (Kalra et al, 1999). Lastly, basal ganglia dopamine is involved in nonhedonic metabolic drive and goal-directed motor behavior towards procurement of food (Volkow et al, 2002c; Cannon et al, 2004).
As a whole, this well-orchestrated machinery is almost infallible in preventing caloric deficit states as it sets of hunger (and supresses satiety), accompanied by negative subjective experiences ranging from slight discomfort to restlessness and food obsession. Eating can therefore be construed as a negative reinforcer as it brings an end to the discomfort induced by hunger (Saper et al, 2002). Food intake is, however, not merely an automatic action aimed at regaining comfort and tranquility. It is highly rewarding and positively reinforcing process. Indeed, palatability, derived from high levels of sugar and fat, is the most important (aside from the energy need) factor determining the amount of consumed food (Berthoud, 2004a). This is perhaps to encourage ingestion of scarce (at the earlier stages of evolution) high-energy content foods (Ulijaszek, 2002; Kelley, 2004b), and to secure steady supply of sugar, which is the primary energy source throughout the central nervous system that is neither stored nor produced there.
NEUROBIOLOGY OF SYSTEMS MEDIATING FOOD REWARD
Neuroanatomy, neurophysiology, and neurochemistry of reward have been at the outset investigated using drug addiction paradigms. Over the last decade, these findings were expended and generalized to food reward (Figure 2), given the well-described substantial overlap between both phenomena (Mercer and Holder, 1997b; Kelley and Berridge, 2002; Berridge and Robinson, 2003; Wang et al, 2004; Volkow and Wise, 2005).
Incentive motivation theory (Berridge and Robinson, 2003), largely based on rodent research, appears to be applicable for both food and drug reward even though it has not yet received substantial clinical support. According to this theory, brain reward function is not a homogeneous entity. Rather, it can be dissected, using neurochemical, neuroanatomical, and functional criteria, into two core processes, namely ‘liking’ and ‘wanting’ (Berridge and Robinson, 2003). Drug-induced changes in the mesolimbic dopaminergic circuitry underlying the wanting but not liking purposes is purportedly responsible for transforming regular wanting responses into heightened incentive salience assigned to drugs or drug-related cues. This incentive sensitization process is construed to be an animal homolog of human craving (Berridge and Robinson, 2003).
A closely related concept, derived from primate work, is the aberrant learning theory suggesting that learning of new rewards is encoded via interactions between tonic (baseline) and phasic spikes in dopaminegic neurons; phasic firing predicts new rewards (Schultz, 2001). Hence, neural adaptations to excessive dopaminergic trafficking in response to drugs (Breiter et al, 1997) or during psychotic episodes (Grace, 1991; Lieberman et al, 1997; Laruelle, 2000; West et al, 2003) may lead to low signal-to-noise detection capability for natural rewards along with ‘profound overlearning of the motivational significance of cues that [either] predict delivery of drugs’ (Hyman, 2005) or are associated with psychotic contents resulting in ‘aberrant salience’ (Kapur, 2003) for drug- or psychosis-related stimuli (detailed in the following sections).
Mesolimbic dopaminergic pathways projecting from the vental tegmental area (VTA) to the nucleus accumbens (NAc), along with the LH, amygdala, and prefrontal cortex (PFC; Goldstein and Volkow, 2002; Volkow et al, 2004a), are responsible for the incentive motivational aspects of reward function. These are collectively termed ‘wanting’ processes (Kelley and Berridge, 2002; Pecina et al, 2003; Kelley, 2004b) and include conditioned learning of stimulus–reward association (McClure et al, 2003; Tobler et al, 2003; Berridge, 2004; Wise, 2004), reward prediction, and attribution of incentive salience to rewarding stimuli (Horvitz, 2000; McClure et al, 2003) by ‘gating’ of stimulus representations in the PFC (Montague et al, 2004). The latter region also integrates peripheral and environmental stimuli and exercises cognitive control over drives and emotions gated from the limbic regions (Goldstein and Volkow, 2002).
Consumption of palatable food produces pleasure and positive affective states. Food ‘liking’ is conveyed to the frontotemporal cortical structures (Berridge, 2003; Kelley, 2004b) through μ-opioid neurotransmission within the scattered network of subcortical and brainstem nuclei (Tanda and Di Chiara, 1998; Saper et al, 2002; Berridge, 2003; Berridge and Robinson, 2003; Pecina et al, 2003; Berthoud, 2004b; Kelley, 2004b; Will et al, 2004), including the NAc, VTA, ventral pallidum, nucleus of the solitary tract, parabrachial nucleus, and the amygdala. Opioids enhance food pleasantness (Doyle et al, 1993; Pecina and Berridge, 1995; Kelley et al, 2002; Will et al, 2003), whereas consumption of high-energy sweet and fat food increases endogenous opiates release (ie, feedforward interaction; Tanda and Di Chiara, 1998; Colantuoni et al, 2002; Grigson, 2002) so that analgesia (Mercer and Holder, 1997a; Segato et al, 1997; Lewkowski et al, 2003), opioid dependence (Schoenbaum et al, 1989, 1990; Colantuoni et al, 2001, 2002), and compensatory decrease of opiate gene expression in the reward structures (Kelley et al, 2003, 2005) may ensue.
The liking and wanting processes, contributing to the fully functional reward system (Berridge, 2004), are dissociable but closely related phenomena. This is obvious on the intuitive level as food desire is usually triggered by palatable, but not bland types of cuisine (Yeomans et al, 1997, 2004; Berthoud, 2004a). Accordingly, opioid-induced feeding depends on the NAc's dopamine receptors signaling (Schmauss and Emrich, 1985; Nylander and Terenius, 1987; Sivam, 1989; You et al, 1994; MacDonald et al, 2004), and in the reversed order, opioids cause dopamine releases in the NAc (Devine et al, 1993a, 1993b; Tanda and Di Chiara, 1998; Yoshida et al, 1999; Schad et al, 2002).
CENTRAL MECHANISMS FOR ENERGY BALANCE AND FOR FOOD REWARD ACT TOGETHER TO ENSURE ADEQUATE NUTRITION
As presented in Figure 3, appetite or incentive motivational drive to seek and consume food is determined by interrelated psychobiological factors including food's rewarding properties, individual's homeostatic needs, and cognitive ability to favor alternative (to eating) behaviors (Saper et al, 2002; Hinton et al, 2004; Kelley, 2004b). Regular (ie, predominately bland) food does not fully engage reward centers and its consumption is effectively terminated with sensation of satiety (Erlanson-Albertsson, 2005) when fulfillment of homeostatic needs is relayed to the brainstem nucleus of the solitary tract (Appleyard et al, 2005; Broberger, 2005) by cranial afferents from the alimentary tract organs (Harding and Leek, 1973).
Schematic overview of the interface between biopsychological factors governing eating behaviors. Appetite, that is, incentive motivational drive to seek and consume food, is derived from interrelated psychobiological factors including food's rewarding properties, individual's homeostatic needs, and cognitive ability to favor alternative (to eating) behaviors (Saper et al, 2002; Hinton et al, 2004; Kelley, 2004b). Satiety is associated with decreased reward function secondary to insulin and leptin actions (Figlewicz, 2003a, 2003b, 2004; Berthoud, 2004b) during caloric overflow signaled by these hormones, whereas caloric restriction, reflected in subjective sensation of hunger, increases food reward (Carr, 2002; Fulton et al, 2004) by augmenting dopaminergic (Carr et al, 2003) and opioidergic (Carr and Wolinsky, 1993) neurotransmission. On the other hand, reward centers activation by high sugar and fat content palatable foods promotes desire ‘to come back for more’ (Kelley et al, 2002) by upregulating of hunger signaling (orexin, agouti-related protein), melanin concentrating hormone) in hypothalamic orexigenic networks and by blunting brain responses to the peripheral satiety hormones, insulin and leptin (Erlanson-Albertsson, 2005; Isganaitis and Lustig, 2005). Prefrontal cortex (PFC) modulates these effects via reciprocal innervation with the hypothalamic–limbic areas (Berthoud, 2004a) by forming subjective hedonic perceptions and integrating them with metabolic signals and with intrapsychic and environmental contexts (Kringelbach, 2005). For the clarity of presentation, the scheme was rendered out-of-scale and simplified to reduce the numbers of the displayed links and structures to those of direct relevance to the main themes of this review.
Satiety is associated with decreased reward function secondary to insulin and leptin actions in the NAc, LH, VTA, taste receptor cells, and cortical areas (Figlewicz, 2003a, 2003b, 2004; Fulton et al, 2004; Berthoud, 2004b) during caloric overflow signaled by these hormones, whereas caloric restriction, reflected in subjective sensation of hunger, increases food reward (Carr, 2002; Fulton et al, 2004) by augmenting dopaminergic (Carr et al, 2003) and opioidergic (Carr and Wolinsky, 1993) neurotransmissions.
On the other hand, reward centers activation by high sugar and fat content palatable foods promotes desire ‘to come back for more’ (Kelley et al, 2002) by upregulating of hunger signaling (orexin, AgRP, and MCH) in hypothalamic orexigenic networks and by blunting brain responses to the peripheral satiety hormones, insulin and leptin (Erlanson-Albertsson, 2005; Isganaitis and Lustig, 2005). Prefrontal cortex modulates these effects via reciprocal innervation with the hypothalamic–limbic areas (Berthoud, 2004a) by forming subjective hedonic perceptions and integrating them with metabolic signals and with intrapsychic and environmental contexts (Kringelbach, 2005).
REPETITIVE PALATABLE FOOD CONSUMPTION MAY DYSREGULATE HOMEOSTATIC AND REWARD PROCESSES
To maintain stability of the body adipose content, energy expenditure should tightly match caloric intake. This is accomplished through negative feedback regulation exerted by the endocrine markers of body fat mass, insulin and leptin; both are also affected by recent food intake and its macronutrient composition (Havel, 2001; Schwartz et al, 2003). In the basal state (Figure 1), insulin and leptin inhibit anabolic pathways (appetite and energy conservation) while maintaining tonic activation of the catabolic pathways (satiety and energy outflow).
Such regulatory system may be adequate for regular diets but is ‘inherently biased toward weight gain’ (Havel, 2001; Schwartz et al, 2003; Schwartz and Niswender, 2004) in ‘obesigenic’ environments abound in ‘supersized’ meals, more than twice caloric density (energy per weight) and content of healthy diets (Prentice and Jebb, 2003). High caloric density food contributes to abnormally elevated total caloric intake as weight and volume of meals ingested by humans tend to remain relatively steady (Rolls et al, 1998). Such rich in caloric content diets robustly activate reward centers and fail to produce proportional (to the amount of ingested calories) suppression of hunger signals, as CNS insulin and leptin stop boosting and restraining, respectively, already activated (in the basal state) catabolic pathways and already inhibited (in the basal state) anabolic pathways, that is, ceiling effect (Schwartz et al, 2003). Thus, metabolic restraint on reward function via insulin/leptin mechanism becomes inefficient at about 40% fat concentration, typical of the Western diet (Figlewicz, 2003a, 2003b, 2004), allowing reward mechanisms to over-ride metabolic requirements (perhaps owing to evolutionary pressure giving preferentiality to food intake over fasting) and underscoring the role of cortical cognitive control mechanisms in the determination of the amount of eaten food.
Pleasurable hedonic impact and inefficient suppression of hunger by palatable foods predictably brings increases in the amount of consumed food and weight gain (in the absence of compensatory caloric loss) with ensuing hyperinsulinemia and insulin resistance (Kahn and Flier, 2000; Homko et al, 2003), which drive weight further upward (Sigal et al, 1997; Odeleye et al, 1997). Moreover, weight gain and insulin resistance from habitual consumption of palatable foods decrease insulin and leptin blood–brain barrier (BBB) penetrability (Caro et al, 1996; Kaiyala et al, 2000; Banks, 2003; Banks and Farrell, 2003; Woods et al, 2003) and their CNS effects (Couce et al, 2001; Banks and Farrell, 2003; Lindqvist et al, 2005; Porte et al, 2005), albeit some homeostatic hypothalamic areas lack BBB (Peruzzo et al, 2000; Ganong, 2000). The resultant brain insulin and leptin ‘resistance’ renders normal satiety signals even more ineffective (Erlanson-Albertsson, 2005; Isganaitis and Lustig, 2005) leading to further impairments in physiologic mechanisms regulating food intake (eg, overeating) and shifting the set point for energy homeostasis towards the development of overweight and obesity (Levine et al, 2003; Erlanson-Albertsson, 2005; Isganaitis and Lustig, 2005). It is quite difficult to reverse the developing vicious cycle (schematically illustrated in Figure 4) as attempted weight loss by caloric restriction results in insulin and leptin declines triggering robust activation of anabolic pathways and suppression of catabolic pathways; the magnitude of these responses exceeds by far that during caloric overflow (Niswender et al, 2004).
Diagram describing repetitive and compulsive nature of palatable food consumption. The figure depicts transition from occasional palatable food intake to dysregulation of homeostatic and reward processes leading to impairments in normal signals of hunger and satiety along with adaptations and deviations of physiological set points (cf, Koob and Le Moal, 2001).
STRESS EXPOSURE ALSO AFFECTS EATING HABITS
Stress is another factor controlling eating behavior and its effects on reward processes are quite similar to addicitive drugs (see Table 1 for comparison of these effects). Acute stress activates dopamine transmission in the reward circuitry through enhancement of extracellular dopamine release and/or potentiation of dopamine receptors’ affinity and their activity (Thierry et al, 1976; Imperato et al, 1989). Chronic stress exerts an opposite action by decreasing dopaminergic (Puglisi-Allegra et al, 1991; Imperato et al, 1993) neurotransmission and is accompanied by decreased motivation towards normally pleasurable stimuli (Willner et al, 1987; Papp et al, 1991). Furthermore, stress-related negative affective states drive comfort or palatable food consumption as it can alleviate these states (ie, ‘self-medication’) via temporary normalization of reward function (Wang et al, 2001), via inhibition of hypothalamic CRF secretion involved in stress-induced anxiety and depression (Dallman et al, 2003) or via priming effects (Wise, 2004) on the mesocorticolimbic dopaminergic system (Thierry et al, 1976; Imperato et al, 1989).
Finally, stress-induced hypercortisolemia contributes to the development of obesity as a result of visceral fat accumulation (Ryan and Thakore, 2002) consequentially to more abundant intra-abdominal adipose tissue's (vs subcutaneous fat mass) concentration of glucocorticoid receptors that activate cortisol-sensitive lipoprotein lipase (Rebuffe-Scrive et al, 1985; Pedersen et al, 1994; Fernandez-Real et al, 1997), an anabolic enzyme promoting buildup of fat (Bjorntorp, 1996). Cotisol's role in stress-induced obesity in schizophrenic patients has been undermined, though, by (1) the lack of consistent hypercortisolemia in nonacutely ill schizophrenia samples (Gispen-de Wied, 2000, 2) the lack of visceral fat accumulation and obesity in drug-naive schizophrenia samples from non-Westernized populations (Zhang et al, 2004), and (3) weight gain commonly occurring in the setting of cortisol decreases during SGAs treatment (Newcomer et al, 2002; Mann et al, 2006).
KEY FACTORS INVOLVED IN THE NORMAL REWARD FUNCTION ARE IMPAIRED IN SCHIZOPHRENIA
Given the metabolic abnormalities arising in healthy people exposed to fast food diets (see above), a compelling a fortiori argument could be that propensity for weight gain in ‘obesigenic’ environments is worsened by neuropathology afflicting the same factors involved in energy homeostasis and reward regulation (Thakore, 2005). Schizophrenia could be considered such a disorder (Figure 5a). One obvious factor is higher rates of peripheral glucose intolerance and insulin resistance observed in some schizophrenic patients (Mukherjee et al, 1996; Ryan et al, 2003) that could lead to CNS insulin and leptin resistance with the ensuing consequences as discussed above.
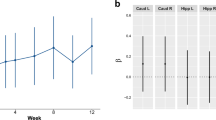
(a) Schematic overview of the psychological signals underlying manifestation of appetite and of the potential sites where schizophrenia neuropathology predisposes for overeating and obesity. Eating patterns are largely determined by homeostatic system responsible for total body energetic balance and by reward system ensuring repetitive activities essential for the survival of individuals and species. To maintain stability of the body adipose content, energy expenditure should tightly match caloric intake. This is accomplished through negative feedback regulation exerted by the endocrine markers of body fat mass, insulin and leptin; both are also affected by recent food intake and its macronutrient composition (Havel, 2001; Schwartz et al, 2003). Propensity for weight gain could be worsened by schizophrenic neuropathology afflicting the same factors involved in energy homeostasis and reward regulation (Thakore, 2005). One such cause is higher rates of peripheral glucose intolerance and insulin resistance in schizophrenic populations (Mukherjee et al, 1996; Ryan et al, 2003) that could lead to CNS insulin and leptin resistance. Mesolimbic hyperdopaminergic state presumably renders motivational/incentive reward system insensitive to low salience/palatability food. Also, palatable food may produce dysregulated dopamine release (Lieberman et al, 1997; Laruelle, 2000) in the ventral striatum, creating a rigid motivational state fixated on procurement of this type of food. These, together with poor cognitive control from the hypofunctional PFC and enhanced hedonic impact of food, owing to exaggerated opioidergic drive, may underlie increased intake of palatable food in patients with schizophrenia. (b) Treatment with SGAs may improve dopamine-mediated reward aspects, but at the cost of increased appetite and insulin resistance along with worsened or at least not improved opiodergic capacity; these effects can further increase intake of palatable food.
There are several lines of evidence that point to deficits of the brain circuits mediating reward and reinforcement in schizophrenic patients (Voruganti et al, 2001; Juckel et al, 2003; Voruganti and Awad, 2004). From a neuroanatomical perspective, reward circuitry structures including NAc, amygdala, and PFC have been reported to be abnormal in schizophrenia in neuropathological (Beckmann and Lauer, 1997; Bogerts et al, 1985; Lauer and Beckmann, 1997; Lauer et al, 2001; Selemon et al, 2002), in structural (Breier et al, 1992; Sigmundsson et al, 2001; Kawasaki et al, 2004), and in functional (see Table 2 for summary) neuroimaging studies.
The association between reward deficits and schizophrenia is also supported by the clinical phenomena typical of this illness: anhedonia, affective flattening, social isolation, avolition, apathy, high comorbidity of SUDs (Regier et al, 1990; American Psychiatric Association, 2000), and insensitivity to pain (discussed below). These characteristics support inclusion of schizophrenia within the spectrum of the ‘reward deficiency syndrome’ (Green et al, 1999), which is comprised of a broad range of personality traits and mental disorders (including, but not limited to schizoid/avoidant character styles, glucose binging and posttraumatic stress disorder) allegedly characterized by hypofunctionality of the reward circuitry, clinically manifested as diminution of drive/motivation and of capacity to enjoy or to experience pleasure (Comings and Blum, 2000).
SCHIZOPHRENIA AND UNHEALTHY FOOD PREFERENCES MAY BE EXPLAINED BY SHARED NEURAL SYSTEMS
Presently, no clinical studies directly link schizophrenia neuropathology and excessive consumption of palatable food. A cumulative body of evidence suggests, however, that (1) ‘predilection for addictive behavior’ is ingrained in schizophrenia neuropathology owing to ‘neural and motivational changes [that are] similar to long-term substance abuse but without the necessity of prior drug exposure’ (reviewed in Chambers et al, 2001) and (2) there is a ‘considerable overlap between brain circuitry that evolved in the service of body-weight regulation and brain circuitry that is usurped by exogenous drugs of abuse’ (reviewed in Volkow and Wise, 2005). The adaptation of these two ideas (discussed in the following sections) lends strength to the hypothesis that schizophrenia and unhealthy food preferences may be potentially explained by recursive partly shared neural systems.
NEURAL AND MOTIVATIONAL CHANGES IN SCHIZOPHRENIA AND IN SUDS
The first of the above-mentioned ideas (Table 3) is based on the neurodevelopmental model of schizophrenia (Weinberger, 1987) and is supported by neuroimaging research (Laruelle, 2000), and by ketamine and amphetamine challenge studies (Malhotra et al, 1997; Krystal et al, 2005). In addition, extensive preclinical work (Grace, 1991, 2000; Pierce and Kalivas, 1997) provides supportive evidence for distinct roles for tonic vs phasic dopaminergic firing and glutamatergic signaling in clinical symptomatology, motivational states, and in the outcomes of schizophrenia and SUDs (Lieberman et al, 1997; Kalivas and Volkow, 2005).
‘Hypofrontality’ or decrease in cerebral metabolism and blood flow have been observed in both schizophrenia (Weinberger and Berman, 1996) and in SUDs (Volkow et al, 1992; Kalivas et al, 2005; Garavan et al, 2000) at baseline or when challenged by cognitive tasks or when exposed to natural reinforcers. The hypofrontality phenomenon has been potentially related to the disease process- (Lieberman et al, 1997; West et al, 2003; Laruelle, 2000) or drug- (Kalivas and Volkow, 2005) related diminished dopaminergic tone with corresponding decrease in the tonic glutamatergic activity.
On the background of this diminished activity, hallucinations (McGuire et al, 1993; Silbersweig et al, 1995; Shergill et al, 2000) or other positive symptoms (Sabri et al, 1997) in schizophrenic patients or exposure to drugs (Volkow et al, 2005) or drug-related cues (Grant et al, 1996; Wang et al, 1999; Childress et al, 1999; Garavan et al, 2000) in drug addicts produce strong activations in the PFC. Prefrontal activations caused by drugs or drug cues in addicts profoundly increase PFC's glutamatergic output (Kalivas and Volkow, 2005; Kalivas et al, 2005) to already hypofunctional NAc (Volkow et al, 1997, 2005), thus further decreasing its dopaminergic activity. This triggers the drive to seek and consume drugs (ie, craving) with resultant drug-induced phasic dopaminergic bombardment (Grace, 2000).
Similarly, in schizophrenics, homeostatic adjustments (Laruelle et al, 2003) to diminished resting state tonic dopaminergic neurotransmission in the PFC and in ventral striatum result in robust augmentations of phasic dopamine responses to drugs and to behaviorally relevant stimuli. The limbic hyperdopaminergic state is further amplified through the long-term plasticity (Pierce and Kalivas, 1997) process of endogenous sensitization (Lieberman et al, 1997; Laruelle et al, 1999; Laruelle, 2000; Chambers et al, 2001; West et al, 2003). This is an autonomous, self-sustaining feedforward loop whereby a trivial event (eg, psychosocial stress) can mount escalating dopamine releases in the striatum, further worsening symptoms of psychosis (Laruelle, 2000).
OVERLAP BETWEEN FOOD AND DRUG REWARD SYSTEMS
Table 4 is a summary of basic neuroscience and limited clinical findings that support the assumed overlap between food and drug reward systems (Berridge, 1996; Volkow and Wise, 2005). Consumption of palatable food (Hajnal et al, 2004) and drugs (Hernandez and Hoebel, 1988) activates dopamine transmission in the mesolimbic dopaminergic system, whereas repeated exposure to both stimuli decreases dopaminergic firing (Bassareo and Di Chiara, 1999; Nestler, 2004) in the same neural structures (ie, habituation and tolerance). Repeated intermittent exposure to food (Rada et al, 2005) or to drugs (Steketee, 2003) conversely results in sensitization, that is, increased dopaminergic release in the NAc. The commonality of the circuitries is further supported by crosssensitization between palatable food with drugs and alcohol (Carr et al, 2003; Avena and Hoebel, 2003a, 2003b; Avena et al, 2004).
Palatable foods do not only engage dopaminergic reward system but also change it (Bello et al, 2002; Fetissov et al, 2002; Hajnal and Norgren, 2002; Bello et al, 2003). That is to say, qualitatively similar to the effects of psychosis or euphorogenic drugs, repeated artificial enhancement of dopaminergic neurotransmission in the reward system leads to a dysfunctional (Wang et al, 2001; Barrot et al, 2002; Bello et al, 2002; Hajnal and Norgren, 2002; Spangler et al, 2004) neuroadaptational state that renders it even less responsive to other natural reinforcers (Wang et al, 2001; Volkow et al, 2004b). Therefore, a common result in vulnerable individuals could be in a form of contribution to the ‘spiraling distress cycle’ (Goldstein and McEwen, 2002; Koob and Le Moal, 2001) depicted in Figure 4, whereby excessive palatable food consumption provoked by its enhanced salience and conditioned learning produces additional deterioration in emotional and behavioral problems, leading to further consumption that may eventually produce a transition from excessive eating to food addiction (Tuomisto et al, 1999).
Both palatable food (Tanda and Di Chiara, 1998; Colantuoni et al, 2002) and drugs (Tanda and Di Chiara, 1998; Roth-Deri et al, 2003) also release endogenous opiates, that is, endorphins and enkephalins. Their chronic consumption leads to neuroadaptations in opioid receptors accompanied by signs and symptoms of opioid dependence (Zubieta et al, 1996; Colantuoni et al, 2002; Bailey et al, 2005; Heinz et al, 2005). Only limited human data (Ibanez-Rojo et al, 1993; Bencherif et al, 2005) are available, though, to extend preclinical palatable food opioid findings to humans.
Neuroimaging studies in obese people reported increased PFC activity when food-related stimuli were presented (Del Parigi et al, 2002) in the face of decreased performance on a cognitive task recruiting PFC (Davis et al, 2004). As discussed above, hypo- (Volkow et al, 1992; Kalivas et al, 2005; Garavan et al, 2000) and hyperfrontality have been reported in drug addicts at baseline and during exposure to drugs (Volkow et al, 2005) or drug-related cues (Grant et al, 1996; Wang et al, 1999; Childress et al, 1999; Garavan et al, 2000), respectively. While the nature of these effects in obesity has not been elucidated, in addicts they have been attributed to drug-induced disintegration between dopaminergic and glutamatergic firing (Grace, 2000; Kelley and Schiltz, 2004; Kalivas and Volkow, 2005; Goto and Grace, 2005).
Although BDNF has been widely recognized as a neurotrophic growth factor (Huang and Reichardt, 2001) and orexin as a regulator of sleep/wake cycle (Takahashi, 1999), the opposing actions (anorexigenic and orexigenic, respectively) of these two neuropeptides in food and drug reward have been also identified. In mesolimbic dopaminergic pathways, BDNF regulates appetitive behavior (Eisch et al, 2003; Itoh et al, 2004) along with behavioral sensitization to drugs (Guillin et al, 2001) and potentially to palatable food. Through its hypothalamic effects, BDNF controls energy balance by enhancing catabolic processes (Xu et al, 2003). Similarly important are the actions of orexin neurons, situated in the LH and projecting to the NAc and VTA (Fadel and Deutch, 2002), to control food intake (Edwards et al, 1999; Harris et al, 2005) as well as drug reward and craving (Harris et al, 2005). In fact, a finding of a correlation between SGAs weight liability and the degree of LH orexin neurons stimulation (Fadel et al, 2002) is provocative and may offer important insights into homeostatic dysregulation associated with antipsychotic agents.
ABNORMAL DOPAMINERGIC FUNCTION IMPAIRS WANTING PROCESSES IN SCHIZOPHRENIA
As discussed above, dopamine theory of psychosis postulates that hyperactivity of mesolimbic dopaminergic pathways mediates the key characteristic symptoms of schizophrenia, delusions and hallucination (Laruelle et al, 1996; Breier et al, 1997; Abi-Dargham et al, 1998; Moore et al, 1999; Laruelle et al, 2003). To put it in more metaphoric terms: mesolimbic dopamine is conceptualized to be ‘the wind’ (Laruelle and Abi-Dargham, 1999), whereas delusions and hallucinations could be the smoke ‘of the psychotic fire’.
Given the central role played by mesolimbic dopamine in the wanting processes, psychotic symptoms in schizophrenic patients could originate from sporadic or stress-induced dopamine spikes causing attribution of salience to random events and/or to trivial subjective experiences (Taylor and Liberzon, 1999; Floresco et al, 2003; Kapur, 2003; Taylor et al, 2005), that is, aberrant conditioned learning (Schultz, 2001). Deficits of the neural mechanism involved in attribution of emotional salience in schizophrenics (Kohler et al, 2003; Moberg et al, 2003; Turetsky et al, 2003) are supported by functional brain imaging (summarized in Table 2) and electrophysiological (Pfefferbaum et al, 1989) studies, employing various natural probes ranging from odors to facial images and aversive vs neutral scenes (Taylor et al, 2002, 2005; Gur et al, 2002; Paradiso et al, 2003; Takahashi et al, 2004; Williams et al, 2004).
Similarly to SUDs, which is another condition associated with erratic supraphysiological dopamine surges in the limbic system (Volkow et al, 2004a), aberrant salience in schizophrenia creates rigid motivational states fixated on irrelevant and idiosyncratic stimuli with diminution of mesolimbic neurons’ ‘signal detection capability’ (Green et al, 2002) for normal salience (Taylor and Liberzon, 1999; Taylor et al, 2005) and with the loss of normal modulation of reinforcers’ values by nonpsychotic contexts (eg, caloric need state; Breier, 1989). Thus, palatable food (eg, ice cream) may be salient enough to compete with the psychosis-related reinforcers as a result of its effects on reward circuitry (Hajnal et al, 2004) and owing to aggressive and ubiquitous advertisement (Wansink, 2004), whereas less salient and palatable nutrition (eg, lettuce and broccoli), lacking these characteristics, may fail to elicit incentive motivation towards its consumption even within the context of caloric deficits (Breier, 1989). Preference for highly palatable food may be further enhanced, despite considerable efforts exerted by public health authorities to explain the risks of unhealthy diets, owing to a hypofunctional PFC involved in the inhibitory control (Volkow and Fowler, 2000; Goldstein and Volkow, 2002; Volkow et al, 2004a) over eating behavior.
ABNORMAL OPIOIDERGIC FUNCTION MAY IMPAIR LIKING PROCESSES IN SCHIZOPHRENIA
Robust elevations of endogenous opiates concentrations in the cerebral spinal fluid and in plasma (Terenius et al, 1976; Lindstrom et al, 1978, 1986; Rimon et al, 1980; Brambilla et al, 1984; Marchesi et al, 1995) is a relatively consistent clinical finding in schizophrenia. Opiates levels tend to parallel the severity of psychosis (Terenius et al, 1976; Lindstrom et al, 1978; Rimon et al, 1980; Marchesi et al, 1995), suggesting involvement of this neurochemical effect in schizophrenia neuropathology (Volavka et al, 1979; Schmauss and Emrich, 1985; Nemeroff and Bissette, 1988).
One possible involvement could be through opiate's interference with the neurotrophines (eg, BDNF) supporting neuronal survival (Weickert et al, 2003; Angelucci et al, 2005) with consequential damage of mesolimbic dopaminergic neurons (Sklair-Tavron et al, 1996). Furthermore, similarly to methadone-maintained patients (Willenbring et al, 1989; Zador et al, 1996), exaggerated opioidergic activity could enhance hedonic preference (ie, liking) for sweet and fatty foods (Doyle et al, 1993; Pecina and Berridge, 1995; Kelley et al, 2002; Will et al, 2003). The consumption of these foods further reduces BDNF efficiency in preventing neuronal death (Molteni et al, 2002) and in regulating reward function (Horger et al, 1999; Kernie et al, 2000; Nakagawa et al, 2003), glucose metabolism (Tonra et al, 1999; Nakagawa et al, 2000; Ono et al, 2000), appetitive behaviors (Eisch et al, 2003; Itoh et al, 2004), and other important homeostatic processes (Xu et al, 2003).
Another manifestation of alterations in endogenous opioids in schizophrenics may relate to pain insensitivity. This and other aberrations in protective mechanisms afforded by the pain system, noted in a number of reports (Davis et al, 1979; Evans, 1980; Davis and Buchsbaum, 1981; Fishbain, 1982; Bickerstaff et al, 1988; Rosenthal et al, 1990; Dworkin, 1994; Kudoh et al, 2000; Torrey, 2002), some of which date back to the days of Haslam (Haslam, 1798; 1809 cited in Torrey, 2002), Kraepelin (Kraepelin, 1919 cited in Hooley and Delgado, 2001), and Bleuler (Bleuler, 1924 cited in Hooley and Delgado, 2001), could be yet another aspect of excessive/altered endogenous opioid function in schizophrenia (Davis et al, 1979; Davis and Buchsbaum, 1981; Davis, 1983; Wiegant et al, 1992). Such a notion is supported clinically by reversal of pain insensitivity by opioid antagonism (Davis et al, 1979) and by molecular abnormalities in specific opioid genes, for example, prodynorphin (Ventriglia et al, 2002) or proenkephalin (Mikesell et al, 1996) in schizophrenic patients. Importantly, pain problems in schizophrenics are apparent in the context of real-life situations, for example, tissue damage following surgical treatment (Kudoh et al, 2000; Murthy et al, 2004), and their consequences range from finger burns caused by cigarette smoking (Jenkins et al, 1996) to such grave medical outcomes as silent myocardial infarction (Marchand, 1955) or delays in management of abdominal emergencies (Katz et al, 1990; Bickerstaff et al, 1988) that could be followed by perforated bowel (Rosenthal et al, 1990) and ruptured appendix (Geschwind, 1977).
ADVANTAGEOUS THERAPEUTIC PROPERTIES OF SGAS HAVE BEEN QUESTIONED
The first-generation antipsychotics (FGAs) improve the perceptual disturbances and other positive symptoms and can quieten down the ‘the wind of psychotic fire’ (Laruelle and Abi-Dargham, 1999) by aborting stress-induced dopamine releases (Laruelle, 2000). Their therapeutic properties are, however, tarnished by serious side effects, including those resulting from dopamine blockade in the cortical and subcortical areas with secondary worsening of reward and motivational deficits (Knable et al, 1997; Voruganti et al, 2001; Voruganti and Awad, 2004). In contrast, SGAs produce net increases in dopamine activity in the cortical dopaminergic pathways and substantially lesser blockade (in comparison to FGAs) of the limbic dopaminergic areas (Stahl, 2002).
Thus, all in all, SGAs share advantageous therapeutic properties for the treatment of motivational, emotional, and cognitive deficits (Stahl, 2002; Stahl and Grady, 2004) as well as for the reduction of dopaminergic sensitivity to stress (Dazzi et al, 2004). These qualities partially explain the emergence of SGAs during the past decade as the first line of choice in the pharmacotherapy of schizophrenia and related disorders (Stahl and Grady, 2004).
However, SGAs beneficial features have been put into question by recent studies. Numerous epidemiological surveys have found BWG to be a prevalent outcome in patients treated with clozapine, olanzapine, and to lesser degree quetiapine and risperidone, but not ziprasidone or aripiprazole (Allison et al, 1999b; American Diabetes Association, 2004; Kane et al, 2004; Newcomer, 2005). For clozapine, one study suggested that its advantageous therapeutic properties may be offset by serious morbidity and mortality associated with the BWG (Fontaine et al, 2001). The longest and the most comprehensive of the prospective trials randomized 1493 schizophrenics to treatment with olanzapine, risperidone, quetiapine, ziprasidone, or perphenazine, and found no differences in discontinuation rates (about 74% at 18 month) between the SGAs and the FGA, perphenazine (Lieberman et al, 2005). Although the medications were discontinued because of different reasons, olanzapine was mostly stopped because of the BWG or related metabolic side effects.
PUTATIVE MECHANISMS OF SGAS-INDUCED WEIGHT GAIN
The mechanisms of SGAs-induced weight gain are likely to be multifactorial and to involve both peripheral and central factors (summarized in Table 5). Among peripheral factors, glucoregulatory abnormalities probably play an important role (Newcomer, 2005). In vitro (Melkersson et al, 2001; Melkersson, 2004; Johnson et al, 2005, but Melkersson and Jansson, 2005) and preclinical studies (Ader et al, 2005) demonstrated that SGAs alter insulin secretion. However, in clinical samples, SGAs appear to spare pancreatic cells function (Ebenbichler et al, 2003; Laimer et al, 2005), but to change insulin signaling (Engl et al, 2005) and produce glucose intolerance and insulin resistance (Ebenbichler et al, 2003; Newcomer et al, 2002; Graham et al, 2005; Henderson et al, 2005). Although majority of glucose metabolism irregularities associated with SGAs therapy are secondary to weight gain (Newcomer, 2005), that causality could run in the opposite direction (Figure 4). That is to say, in addition to decreasing the ability of homeostatic and reward regions sensing of adiposity- or food-related signals (Figure 5b; Figlewicz, 2003a, 2003b, 2004; Berthoud, 2004b), insulin resistance increases body fat mass as it tends not to afflict adipose tissue that retains insulin sensitivity even in the face of other tissues’ resistance (Mizuno et al, 2004; Virtanen et al, 2005; Isganaitis and Lustig, 2005).
In contrast, SGAs’ leptin effects (Monteleone et al, 2002; Melkersson and Dahl, 2003; Ebenbichler et al, 2005; Murashita et al, 2005; Sporn et al, 2005; Theisen et al, 2005) seem to be entirely attributable to increased adiposity (Haupt et al, 2005; Sporn et al, 2005). In a cross-sectional study of plasma leptin and adiposity in schizophrenic patients treated with olanzapine (N=27), risperidone (N=24), or a typical antipsychotic (N=21), and in 124 healthy controls, BMI correlated with leptin levels in the entire sample. No SGAs effects on leptin were observed, despite adequate power, when SGAs-treated patients were individually matched to the controls by their BMI values (Haupt et al, 2005).
Increases (Murashita et al, 2005; Palik et al, 2005), decreases (Togo et al, 2004), and no change (Sporn et al, 2005; Theisen et al, 2005; Himmerich et al, 2005) in plasma ghrelin levels were reported in patients treated with SGAs; a consensus regarding clinical significance of these changes has not yet transpired. Likewise, potential contributions to weight gain of norepinephrine (Elman et al, 2002, 2004) and prolactin (Melkersson, 2005; Mann et al, 2006) increases associated with SGA treatment are still unclear. The findings of SGA effects on the brain BDNF (Bai et al, 2003; Luo et al, 2004; Angelucci et al, 2005) and orexin (Fadel et al, 2002) in preclinical literature may be also of potential importance as both neurochemicals are involved in reward and appetitive functions; however, their precise role and how they are influenced by SGAs in humans (Dalal et al, 2003; Weickert et al, 2005) are intriguing topics for future research. In addition, further studies are needed to examine a potential involvement of the endogenous cannabinoid system that has been implicated in schizophrenia (Weiser and Noy, 2005), in obesity (Engeli et al, 2005), and in the mechanisms of action of SGAs, for example, clozapine (Sundram et al, 2005).
EFFECTS OF SGAS ON FOOD INTAKE
Observational preclinical (Thornton-Jones et al, 2002; Ota et al, 2002; Hartfield et al, 2003a; Arjona et al, 2004; Cope et al, 2005) and clinical data suggest that one of the ways SGAs can lead to changes in the patterns of food intake is by increasing appetite (Figure 5b; Bromel et al, 1998; Briffa and Meehan, 1998; Eder et al, 2001; Basson et al, 2001; Kinon et al, 2005) and subsequent consumption of the previously preferred diet (Gothelf et al, 2002; Kane et al, 2004).
Additionally, SGAs can contribute to decreased caloric expenditure and subsequent BWG via diminished physical activity (Arjona et al, 2004) owing to their pronounced sedative effects (Lublin et al, 2005). However, the relative contribution of SGAs effects on energy intake and/or expenditure remains largely unquantified by experimental studies. To begin exploring this issue, Gothelf et al (2002) prospectively measured BMIs, caloric intake, and energy expenditure in 10 olanzapine-treated subjects and compared them to a matched group of patients treated with haloperidol. At the 4-week follow-up time point, the patients had increases in both caloric intake and in the BMI. Their resting and daily energy expenditure remained unchanged. The results of this pilot study add further support to the hypothesis that caloric intake plays an important role in the SGAs-induced weight gain.
An important candidate mechanism for SGAs-induced increases in appetite is blockade of receptors systems such as 5-HT2A, 5-HT2C, serotonin; D1, D2, D3, and D4 dopamine; H1 histamine, α1 and α2 adrenergic and acetylcholine (Van Tol et al, 1991; Zeng et al, 1997; Seeman et al, 1997; Wirshing et al, 1999; Meltzer, 1999; Richelson, 1999; Richelson and Souder, 2000; Casey and Zorn, 2001; Muller et al, 2004; Kroeze et al, 2003; Templeman et al, 2005; Johnson et al, 2005; Reynolds et al, 2005), involved in appetite regulation in hypothalamus and elsewhere in the brain. Greater part of the receptor data are based on preclinical models (eg, knockout mice). However, some but not all (Tsai et al, 2002; Basile et al, 2002) clinical studies implicate polymorphism of the 5-HT2C receptor gene in BWG in response to SGAs (Reynolds et al, 2002, 2005; Ellingrod et al, 2005). In addition, one study (Wirshing et al, 1999) found a logarithmic relationship between H1 receptor affinity in patients treated with clozapine (N=20), olanzapine (N=13), risperidone (N=38), haloperidol (N=43), and sertindole (N=8). This study is echoed by another article (Kroeze et al, 2003), demonstrating that H1 receptor affinity predicted weight gain for both FGA- and SGA-treated patients enrolled in meta-analysis of 418 antipsychotic drug-induced estimates of weight change (Allison et al, 1999b).
Nevertheless, rank ordering of specific SGAs’ propensities to increase body weight against all their binding affinities at the human brain receptors argues against antagonism of a single receptor as a sufficient cause for SGA-induced BWG (Richelson, 1999; Richelson and Souder, 2000). For instance, an SGA that does not appear to produce BWG, ziprasidone, also binds with substantial affinity to the 5-HT2C and H1 receptors (Richelson, 1999; Richelson and Souder, 2000; Pouzet et al, 2003; Kirk et al, 2004). Hence, it is possible that simultaneous blockade of several (rather than single) receptor, for example, H1, 5-HT2, D2, and acetylcholine receptors may have a synergistic effect on increased appetite and food intake (Masand, 1999; Wetterling, 2001; Hartfield et al, 2003b).
OPIOID ANTAGONISTS MAY IMPROVE HEDONIC DEFICITS IN PATIENTS WITH SCHIZOPHRENIA
Dopamine remains the most extensively investigated neurotransmitter system in the mechanism of the SGAs action (Kapur and Mamo, 2003). However, among the complex concoction of their pharmacological properties, some SGAs (clozapine, olanzapine, and risperidone) may also be associated with enhancements in the opioidergic activity, which could further deteriorate the pre-existing hedonic alterations in patients with schizophrenia.
Several lines of clinical and preclinical evidence support this sort of effect. These include clinical presentation of olanzapine overdose, which is indistinguishable from opioid intoxication (O’Malley et al, 1999; Kochhar et al, 2002; Palenzona et al, 2004) and analgesic/antinociceptive properties of clozapine, olanzapine, and rispiridone observed in both human (Kiser et al, 2001; Silberstein et al, 2002; Fe-Bornstein et al, 2002; Khojainova et al, 2002; Gorski and Willis, 2003) and rodent (Schreiber et al, 1997, 1999; Weizman et al, 2003) models and ascribed to the opioid mechanisms (Schreiber et al, 1997, 1999; Weizman et al, 2003). Taken together, these results suggest that SGAs are likely to have positive impact on the incentive/motivational reward aspects (including drive to procure food), but at the cost of worsened, or at least not improved opioid-mediated hedonic capacity.
If excess of central opioid activity, which is consequential to the schizophrenia neuropathology with or without SGAs pharmacotherapy, creates metabolic problems for the patients, it is reasonable to expect amelioration of the symptoms through blockade of opioid receptors. Opioid antagonism enhances sensitivity of opioid receptors (Zukin et al, 1982; Lesscher et al, 2003) and diminishes tolerance and physical dependence on opioids (Powell et al, 2002). In rodent studies, opioid antagonists block hedonic responses to sweet foods and sugar-induced analgesia (Blass et al, 1987; Blass and Fitzgerald, 1988) without affecting total energy intake (Berridge, 1996; Rodefer et al, 1999).
Moreover, all but one human study (Hetherington et al, 1991) employing opioid receptor antagonists, including naloxone (Thompson et al, 1982; Trenchard and Silverstone, 1983; Cohen et al, 1985; Drewnowski et al, 1992; MacIntosh et al, 2001), naltrexone (Fantino et al, 1986; Jonas and Gold, 1986; Melchior et al, 1989; Bertino et al, 1991; Chatoor et al, 1994; Yeomans and Gray, 1996, 1997), and nalmefene (Yeomans et al, 1990; Yeomans and Wright, 1991), found significant decreases in caloric intake (Yeomans and Gray, 2002).
In clinical populations, opioid antagonism successfully curbed excessive food intake in bulimic patients (Jonas and Gold, 1986; Chatoor et al, 1994; Marrazzi et al, 1995a, 1995b) as well as opioid (Shufman et al, 1994) and alcohol (O’Mara and Wesley, 1994) abuse. Above and beyond their dietary effects, opioid antagonists may also contribute to weight reduction by reasons of diminished hypeinsulinemia and improved insulin sensitivity (Cucinelli et al, 2002; Fruzzetti et al, 2002). Overall, these findings support the hypotheses that contrary to dopamine antagonism that worsen reward function (Knable et al, 1997; Voruganti et al, 2001; Voruganti and Awad, 2004), blockade of opioid receptors may actually improve it and that these agents can be successfully used in clinical settings.
On the basis of the hypothesis that opioidergic mechanisms are involved in the pathophysiology and symptoms of schizophrenia (Volavka et al, 1979; Schmauss and Emrich, 1985; Nemeroff and Bissette, 1988), opioid antagonists were evaluated for potential antipsychotic efficacy in numerous clinical trials (McNicholas and Martin, 1984; Nemeroff and Bissette, 1988; Welch and Thompson, 1994). On the whole, they were safe and well tolerated and resulted in significant improvements of various aspects of schizophrenic symtomatology such as positive and deficit symptoms, polydipsia, alcoholism, and tardive dyskinesia in some (Watson et al, 1978; Lehmann et al, 1979; Berger et al, 1981; Nishikawa et al, 1994a, 1994b; Marchesi et al, 1995; Petrakis et al, 2004; Wonodi et al, 2004), but not all (Volavka et al, 1977; Pickar et al, 1982) participants.
Possible explanations for these partial treatment responses may have to do with varying dosing strategies, oral vs injectable preparations, open-label vs blind designs, different lengths of treatments, and use of the antagonists as sole agents vs augmentation strategies of various antipsychotic medications. Thus, double-blind placebo-controlled dose–response trials employing the only available (and therefore mostly clinically relevant) oral antagonist form, naltrexone, as an add-on treatment to a standard SGA (eg, olanzapine) may be an important future direction. The potential clinical utility of naltrexone for reward deficiency spectrum problems in schizophrenia is supported by its ability to curb alcohol consumption when administered in a double-blind placebo-controlled manner to comorbid schizophrenic/alcohol-dependent patients on both SGAs and FGAs (Petrakis et al, 2004). In short, these preliminary data suggest that blockade of opioid receptors may have beneficial effects in patients with schizophrenia, especially in the domain of hedonic alterations, but without impairing motivational aspects of reward or aggravating positive symptoms. Definite treatment recommendations are, however, pending rigorously designed clinical trials.
CONCLUSIONS
Overeating, driven by ubiquitously available fast food, appears to be a major cause of the obesity epidemic in the industrialized world. The coalescence of preclinical, neuroimaging, and clinical data suggests that schizophrenic patients may have a predilection for excessive consumption of fast food-type nutrition owing to functionally impaired neural substrate comprising homeostatic and reward mechanisms. Treatment with SGAs does not fully address this problem and may even worsen it.
If confirmed in clinical studies, our insights could have important implications for the primary and secondary prevention of obesity in schizophrenia. If neurobiologic vulnerability factors for obesity could be identified, they might be used to screen patients at risk for the development of such condition. Patients found to possess high vulnerability for the development of obesity owing to schizophrenia-related alterations in glucoregulatory or reward function might be counseled to avoid excessive sweet and fat consumption (primary prevention), or targeted for early intervention even in the presence of mild weight problems (secondary prevention).
The proposed model of excessive BWG in schizophrenia could also have treatment implications, as it implies use of opioid receptors antagonists for amelioration of weight problems in schizophrenics. Clinical experience suggests that opioid antagonists utilization is lower than what could be expected from positive outcomes of clinical trials (Yeomans and Gray, 2002). We therefore believe that this Perspective Article will equip practitioners with the essential knowledge base for understanding the rationale for naltrexone therapy and will sharpen their sensitivity to the unmet medical needs of their schizophrenic patients. Lastly, this model may also provide important leads for recognition and treatment of hazardous eating habits in patients with other disorders, including obesity (Wang et al, 2001), SUDs (Kampov-Polevoy et al, 2001), and major depression (Papakostas et al, 2005).
References
Abi-Dargham A, Gil R, Krystal J, Baldwin RM, Seibyl JP, Bowers M et al (1998). Increased striatal dopamine transmission in schizophrenia: confirmation in a second cohort. Am J Psychiatry 155: 761–767.
Ader M, Kim SP, Catalano KJ, Ionut V, Hucking K, Richey JM et al (2005). Metabolic dysregulation with atypical antipsychotics occurs in the absence of underlying disease: a placebo-controlled study of olanzapine and risperidone in dogs. Diabetes 54: 862–871.
Allison DB, Fontaine KR, Heo M, Mentore JL, Cappelleri JC, Chandler LP et al (1999a). The distribution of body mass index among individuals with and without schizophrenia. J Clin Psychiatry 60: 215–220.
Allison DB, Mentore JL, Heo M, Chandler LP, Cappelleri JC, Infante MC et al (1999b). Antipsychotic-induced weight gain: a comprehensive research synthesis. Am J Psychiatry 156: 1686–1696.
American Psychiatric Association (2000). Diagnostic and Statistical Manual of Mental Disorders DSM-IV-TR (Text Revision). American Psychiatric Press: Washington, DC.
American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity (2004). Consensus development conference on antipsychotic drugs and obesity and diabetes. J Clin Psychiatry 65: 267–272.
Andreasen NC, Rezai K, Alliger R, Swayze II VW, Flaum M, Kirchner P et al (1992). Hypofrontality in neuroleptic-naive patients and in patients with chronic schizophrenia. Assessment with xenon 133 single-photon emission computed tomography and the Tower of London. Arch Gen Psychiatry 49: 943–958.
Angelucci F, Brene S, Mathe AA (2005). BDNF in schizophrenia, depression and corresponding animal models. Mol Psychiatry 10: 345–352.
Arjona AA, Zhang SX, Adamson B, Wurtman RJ (2004). An animal model of antipsychotic-induced weight gain. Behav Brain Res 152: 121–127.
Appleyard SM, Bailey TW, Doyle MW, Jin YH, Smart JL, Low MJ (2005). Proopiomelanocortin neurons in nucleus tractus solitarius are activated by visceral afferents: regulation by cholecystokinin and opioids. J Neurosci 6: 3578–3585.
Avena NM, Carrillo CA, Needham L, Leibowitz SF, Hoebel BG (2004). Sugar-dependent rats show enhanced intake of unsweetened ethanol. Alcohol 34: 203–209.
Avena NM, Hoebel BG (2003a). Amphetamine-sensitized rats show sugar-induced hyperactivity (cross-sensitization) and sugar hyperphagia. Pharmacol Biochem Behav 74: 635–639.
Avena NM, Hoebel BG (2003b). A diet promoting sugar dependency causes behavioral cross-sensitization to a low dose of amphetamine. Neuroscience 122: 17–20.
Bai O, Chlan-Fourney J, Bowen R, Keegan D, Li XM (2003). Expression of brain-derived neurotrophic factor mRNA in rat hippocampus after treatment with antipsychotic drugs. J Neurosci Res 71: 127–131.
Bailey A, Gianotti R, Ho A, Kreek MJ (2005). Persistent upregulation of mu-opioid, but not adenosine, receptors in brains of long-term withdrawn excalating dose ‘binge’ cocaine-treated rats. Synapse 57: 160–166.
Banks WA (2003). Is obesity a disease of the blood–brain barrier? Physiological, pathological, and evolutionary considerations. Curr Pharm Des 9: 801–809.
Banks WA, Farrell CL (2003). Impaired transport of leptin across the blood–brain barrier in obesity is acquired and reversible. Am J Physiol Endocrinol Metab 285: E10–E15.
Barrett JM, Spealman RD (1978). Behavior simulataneously maintained by both presentation and termination of noxious stimuli. J Exp Anal Behav 29: 375–383.
Barrot M, Olivier JD, Perrotti LI, DiLeone RJ, Berton O, Eisch AJ et al (2002). CREB activity in the nucleus accumbens shell controls gating of behavioral responses to emotional stimuli. Proc Natl Acad Sci USA 99: 11435–11440.
Basile VS, Masellis M, De Luca V, Meltzer HY, Kennedy JL (2002). 759C/T genetic variation of 5HT(2C) receptor and clozapine-induced weight gain. Lancet 360: 1790–1791.
Bassareo V, Di Chiara G (1999). Differential responsiveness of dopamine transmission to food-stimuli in nucleus accumbens shell/core compartments. Neuroscience 89: 637–641.
Basson BR, Kinon BJ, Taylor CC, Szymanski KA, Gilmore JA, Tollefson GD (2001). Factors influencing acute weight change in patients with schizophrenia treated with olanzapine, haloperidol, or risperidone. J Clin Psychiatry 62: 231–238.
Becerra L, Breiter HC, Wise R, Gonzalez RG, Borsook D (2001). Reward circuitry activation by noxious thermal stimuli. Neuron 32: 927–946.
Beckmann H, Lauer M (1997). The human striatum in schizophrenia. II. Increased number of striatal neurons in schizophrenics. Psychiatry Res 68: 99–109.
Bello NT, Lucas LR, Hajnal A (2002). Repeated sucrose access influences dopamine D2 receptor density in the striatum. Neuroreport 13: 1575–1578.
Bello NT, Sweigart KL, Lakoski JM, Norgren R, Hajnal A (2003). Restricted feeding with scheduled sucrose access results in an upregulation of the rat dopamine transporter. Am J Physiol Regul Integr Comp Physiol 284: R1260–R1268.
Bencherif B, Guarda AS, Colantuoni C, Ravert HT, Dannals RF, Frost JJ (2005). Regional mu-opioid receptor binding in insular cortes is decreased in bulimia nervosa and correlates inversely with fasting behavior. J Nucl Med 46: 1349–1351.
Berger PA, Watson SJ, Akil H, Barchas JD (1981). The effects of naloxone in chronic schizophrenia. Am J Psychiatry 138: 913–918.
Berridge KC (1996). Food reward: brain substrates of wanting and liking. Neurosci Biobehav Rev 20: 1–25.
Berridge KC (2003). Pleasures of the brain. Brain Cogn 52: 106–128.
Berridge KC (2004). Motivation concepts in behavioral neuroscience. Physiol Behav 81: 179–209.
Berridge KC, Robinson TE (2003). Parsing reward. Trends Neurosci 26: 507–513.
Berridge KC, Valenstein ES (1991). What psychological process mediates feeding evoked by electrical stimulation of the lateral hypothalamus? Behav Neurosci 105: 3–14.
Berthoud HR (2004a). Mind versus metabolism in the control of food intake and energy balance. Physiol Behav 81: 781–793.
Berthoud HR (2004b). Neural control of appetite: cross-talk between homeostatic and non-homeostatic systems. Appetite 43: 315–317.
Bertino M, Beauchamp GK, Engelman K (1991). Naltrexone, an opioid blocker, alters taste perception and nutrient intake in humans. Am J Physiol 261: R59–R63.
Bickerstaff LK, Harris SC, Leggett RS, Cheah KC (1988). Pain insensitivity in schizophrenic patients. A surgical dilemma. Arch Surg 123: 49–51.
Bjorntorp P (1996). The regulation of adipose tissue distribution in humans. Int J Obes Relat Metab Disord 20: 291–302.
Blass E, Fitzgerald E, Kehoe P (1987). Interactions between sucrose, pain and isolation distress. Pharmacol Biochem Behav 26: 483–489.
Blass EM, Fitzgerald E (1988). Milk-induced analgesia and comforting in 10-day-old rats: opioid mediation. Pharmacol Biochem Behav 29: 9–13.
Bogerts B, Meertz E, Schonfeldt-Bausch R (1985). Basal ganglia and limbic system pathology in schizophrenia. A morphometric study of brain volume and shrinkage. Arch Gen Psychiatry 42: 784–791.
Bozarth MA (1987). Methods of Assessing the Reinforcing Properties of Abused Drugs. Springer-Verlag: New York.
Brady KT, Sinha R (2005). Co-occurring mental and substance use disorders: the neurobiological effects of chronic stress. Am J Psychiatry 62: 1483–1493.
Brambilla F, Facchinetti F, Petraglia F, Vanzulli L, Genazzani AR (1984). Secretion pattern of endogenous opioids in chronic schizophrenia. Am J Psychiatry 141: 1183–1189.
Brar JS, Ganguli R, Pandina G, Turkoz I, Berry S, Mahmoud R (2005). Effects of behavioral therapy on weight loss in overweight and obese patients with schizophrenia or schizoaffective disorder. J Clin Psychiatry 66: 205–212.
Breier A (1989). Experimental approaches to human stress research: assessment of neurobiological mechanisms of stress in volunteers and psychiatric patients. Biol Psychiatry 26: 438–462.
Breier A, Berg PH, Thakore JH, Naber D, Gattaz WF, Cavazzoni P et al (2005). Olanzapine versus ziprasidone: results of a 28-week double-blind study in patients with schizophrenia. Am J Psychiatry 162: 1879–1887.
Breier A, Buchanan RW, Elkashef A, Munson RC, Kirkpatrick B, Gellad F (1992). Brain morphology and schizophrenia. A magnetic resonance imaging study of limbic, prefrontal cortex, and caudate structures. Arch Gen Psychiatry 49: 921–926.
Breier A, Su TP, Saunders R, Carson RE, Kolachana BS, de Bartolomeis A et al (1997). Schizophrenia is associated with elevated amphetamine-induced synaptic dopamine concentrations: evidence from a novel positron emission tomography method. Proc Natl Acad Sci USA 94: 2569–2574.
Breiter HC, Gollub RL, Weisskoff RM, Kennedy DN, Makris N, Berke JD et al (1997). Acute effects of cocaine on human brain activity and emotion. Neuron 19: 591–611.
Briffa D, Meehan T (1998). Weight changes during clozapine treatment. Aust NZ J Psychiatry 32: 718–721.
Broberger C (2005). Brain regulation of food intake and appetite: molecules and networks. J Intern Med 258: 301–327.
Bromel T, Blum WF, Ziegler A, Schulz E, Bender M, Fleischhaker C (1998). Serum leptin levels increase rapidly after initiation of clozapine therapy. Mol Psychiatry 3: 76–80.
Brown S, Birtwistle J, Roe L, Thompson C (1999). The unhealthy lifestyle of people with schizophrenia. Psychol Med 29: 697–701.
Brown S, Inskip H, Barraclough B (2000). Causes of the excess mortality of schizophrenia. Br J Psychiatry 177: 212–217.
Butovsky E, Juknat A, Goncharov I, Elbaz J, Eilam R, Zangen A et al (2005). In vivo up-regulation of brain-derived neurotropic factor in specific brain areas by chronic exposure of Delta-tetrahydrocannabinol. Neurochem 93: 802–811.
Cannon CM, Abdallah L, Tecott LH, During MJ, Palmiter RD (2004). Dysregulation of striatal dopamine signaling by amphetamine inhibits feeding by hungry mice. Neuron 44: 509–520.
Carlsson M, Carlsson A (1990). Schizophrenia: a subcortical neurotransmitter imbalance syndrome? Schizophr Bull 16: 425–432.
Caro JF, Kolaczynski JW, Nyce MR, Ohannesian JP, Opentanova I, Goldman WH et al (1996). Decreased cerebrospinal-fluid/serum leptin ratio in obesity: a possible mechanism for leptin resistance. Lancet 348: 159–161.
Carr KD (2002). Augmentation of drug reward by chronic food restriction: behavioral evidence and underlying mechanisms. Physiol Behav 76: 353–364.
Carr KD, Tsimberg Y, Berman Y, Yamamoto N (2003). Evidence of increased dopamine receptor signaling in food-restricted rats. Neuroscience 119: 1157–1167.
Carr KD, Wolinsky TD (1993). Chronic food restriction and weight loss produce opioid facilitation of perifornical hypothalamic self-stimulation. Brain Res 607: 141–148.
Casey DE, Zorn SH (2001). The pharmacology of weight gain with antipsychotics. J Clin Psychiatry 62 (Suppl 7): 4–10.
Chambers RA, Krystal JH, Self DW (2001). A neurobiological basis for substance abuse comorbidity in schizophrenia. Biol Psychiatry 50: 71–83.
Chatoor I, Herman BH, Hartzler J (1994). Effects of the opiate antagonist, naltrexone, on binging antecedents and plasma beta-endorphin concentrations. J Am Acad Child Adolesc Psychiatry 33: 748–752.
Cheng TO (2005). The Framingham heart study functions cannot be applied to the Chinese population for risk assessment for coronary heart disease. Int J Cardiol 104: 228.
Cheta D, Dumitrescu C, Georgescu M, Cocioaba G, Lichiardopol R, Stamoran M (1990). A study on the types of diabetes mellitus in first degree relatives of diabetic patients. Diabete Metab 16: 11–15.
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O’Brien CP (1999). Limbic activitation during cue-induced cocaine craving. Am J Psychiatry 156: 11–18.
Christensen O, Christensen E (1988). Fat consumption and schizophrenia. Acta Psychiatr Scand 78: 587–591.
Cohen MR, Cohen RM, Pickar D, Murphy DL (1985). Naloxone reduces food intake in humans. Psychosom Med 47: 132–138.
Colantuoni C, Rada P, McCarthy J, Patten C, Avena NM, Chadeayne A et al (2002). Evidence that intermittent, excessive sugar intake causes endogenous opioid dependence. Obes Res 10: 478–488.
Colantuoni C, Schwenker J, McCarthy J, Rada P, Ladenheim B, Cadet JL et al (2001). Excessive sugar intake alters binding to dopamine and mu-opioid receptors in the brain. Neuroreport 12: 3549–3552.
Comings DE, Blum K (2000). Reward deficiency syndrome: genetic aspects of behavioral disorders. Prog Brain Res 126: 325–341.
Cope MB, Nagy TR, Fernandez JR, Geary N, Casey DE, Allison DB (2005). Antipsychotic drug-induced weight gain: development of an animal model. Int J Obes Relat Metab Disord (London) 29: 607–614.
Couce ME, Green D, Brunetto A, Achim C, Lloyd RV, Burguera B (2001). Limited brain access for leptin in obesity. Pituitary 4: 101–110.
Crespo-Facorro B, Paradiso S, Andreasen NC, O’Leary DS, Watkins GL, Ponto LL et al (2001). Neural mechanisms of anhedonia in schizophrenia: a PET study of response to unpleasant and pleasant odors. JAMA 286: 427–435.
Cucinelli F, Soranna L, Perri C, Romualdi D, Barini A, Mancuso S et al (2002). Naloxone decreases insulin secretion in hyperinsulinemic postmenopausal women and may positively affect hormone replacement therapy. Fertil Steril 78: 1017–1024.
Dalal MA, Schuld A, Pollmacher T (2003). Lower CSF orexin A (hypocretin-1) levels in patients with schizophrenia treated with haloperidol compared to unmedicated subjects. Mol Psychiatry 8: 836–837.
Dallman MF, Pecoraro N, Akana SF, La Fleur SE, Gomez F, Houshyar H et al (2003). Chronic stress and obesity: a new view of ‘comfort food’. Proc Natl Acad Sci USA 100: 11670–11696.
Davis C, Levitan RD, Muglia P, Bewell C, Kennedy JL (2004). Decision-making deficits and overeating: a risk model for obesity. Obes Res 12: 929–935.
Davis GC (1983). Endorphins and pain. Psychiatr Clin North Am 6: 473–478.
Davis GC, Buchsbaum MS (1981). Pain sensitivity and endorphins in functional psychoses. Mod Probl Pharmacopsychiatry 17: 97–108.
Davis GC, Buchsbaum MS, van Kammen DP, Bunney Jr WE (1979). Analgesia to pain stimuli in schizophrenics and its reversal by naltrexone. Psychiatry Res 1: 61–69.
Dazzi L, Seu E, Cherchi G, Biggio G (2004). Inhibition of stress-induced dopamine output in the rat prefrontal cortex by chronic treatment with olanzapine. Biol Psychiatry 55: 477–483.
Del Parigi A, Gautier JF, Hen K, Salbe AD, Ravussin E, Reiman E et al (2002). Neuroimaging and obesity: maping the brain responses to hunger and satiation in humans using positron emission tomography. Ann NY Acad Sci 967: 389–397.
Devine DP, Leone P, Pocock D, Wise RA (1993a). Differential involvement of ventral tegmental mu, delta and kappa opioid receptors in modulation of basal mesolimbic dopamine release: in vivo microdialysis studies. J Pharmacol Exp Ther 266: 1236–1246.
Devine DP, Leone P, Wise RA (1993b). Mesolimbic dopamine neurotransmission is increased by administration of mu-opioid receptor antagonists. Eur J Pharmacol 243: 55–64.
DiLeone RJ, Georgescu D, Nestler EJ (2003). Lateral hypothalamic neuropeptides in reward and drug addiction. Life Sci 73: 759–768.
Dixon L, Weiden P, Delahanty J, Goldberg R, Postrado L, Lucksted A et al (2000). Prevalence and correlates of diabetes in national schizophrenia samples. Schizophr Bull 26: 903–912.
Doyle TG, Berridge KC, Gosnell BA (1993). Morphine enhances hedonic taste palatability in rats. Pharmacol Biochem Behav 46: 745–749.
Drewnowski A, Krahn DD, Demitrack MA, Nairn K, Gosnell BA (1992). Taste responses and preferences for sweet high-fat foods: evidence for opioid involvement. Physiol Behav 51: 371–379.
Dworkin RH (1994). Pain insensitivity in schizophrenia: a neglected phenomenon and some implications. Schizophr Bull 20: 235–248.
Dynes JB (1969). Diabetes in schizophrenia and diabetes in nonpsychotic medical patients. Dis Nerv Syst 30: 341–344.
Ebenbichler C, Laimer M, Kranebitter M, Lechleitner M, Patsch JR, Baumgartner S et al (2005). The soluble leptin receptor in olanzapine-induced weight gain: results from a prospective study. Schizophr Res 75: 143–146.
Ebenbichler CF, Laimer M, Eder U, Mangweth B, Weiss E, Hofer A et al (2003). Olanzapine induces insulin resistance: results from a prospective study. J Clin Psychiatry 64: 1436–1439.
Eder U, Mangweth B, Ebenbichler C, Weiss E, Hofer A, Hummer M et al (2001). Association of olanzapine-induced weight gain with an increase in body fat. Am J Psychiatry 158: 1719–1722.
Edwards CM, Abusnana S, Sunter D, Murphy KG, Ghatei MA, Bloom SR (1999). The effect of the orexins on food intake: comparison with neuropeptide Y, melanin-concentrating hormone and galanin. J Endocrinol 160: R7–R12.
Erlanson-Albertsson C (2005). How palatable food disrupts appetite regulation. Basic Clin Pharmacol Toxicol 97: 61–73.
Eisch AJ, Bolanos CA, de Wit J, Simonak RD, Pudiak CM, Barrot M et al (2003). Brain-derived neurotrophic factor in the ventral midbrain–nucleus accumbens pathway: a role in depression. Biol Psychiatry 54: 994–1005.
Ellingrod VL, Perry PJ, Ringold JC, Lund BC, Bever-Stille K, Fleming F et al (2005). Weight gain associated with the −759C/T polymorphism of the 5HT2C receptor and olanzapine. Am J Med Genet B 134: 76–78.
Elman I, Ariely D, Mazar N, Aharon I, Lasko NB, Macklin ML et al (2005). Probing reward function in post-traumatic stress disorder with beautiful facial images. Psychiatry Res 135: 179–183.
Elman I, Goldstein DS, Green AI, Eisenhofer G, Folio CJ, Holmes CS et al (2002). Effects of risperidone on the peripheral noradrenegic system in patients with schizophrenia: a comparison with clozapine and placebo. Neuropsychopharmacology 27: 293–300.
Elman I, Rott D, Green AI, Langleben DD, Lukas SE, Goldstein DS et al (2004). Effects of pharmacological doses of 2-deoxyglucose on plasma catecholamines and glucose levels in patients with schizophrenia. Psychopharmacology (Berlin) 176: 369–375.
Engeli S, Bohnke J, Feldpausch M, Gorzelniak K, Janke J, Batkai S et al (2005). Activation of the peripheral endocannabinoid system in human obesity. Diabetes 54: 2838–2843.
Engl J, Laimer M, Niederwanger A, Kranebitter M, Starzinger M, Pedrini MT (2005). Olanzapine impairs glycogen synthesis and insulin signaling in L6 skeletal muscle cells. Mol Psychiatry 10: 1089–1096.
Evans DL (1980). Pain insensitivity in psychotic patients. Am J Psychiatry 137: 507–508.
Fadel J, Bubser M, Deutch AY (2002). Differential activation of orexin neurons by antipsychotic drugs associated with weight gain. J Neurosci 22: 6742–6746.
Fadel J, Deutch AY (2002). Anatomical substrates of orexin–dopamine interactions: lateral hypothalamic projections to the ventral tegmental area. Neuroscience 111: 379–387.
Fahim C, Stip E, Mancini-Marie A, Gendron A, Mensour B, Beauregard M (2005a). Differential hemodynamic brain activity in schizophrenia patients with blunted affect during quetiapine treatment. J Clin Psychopharmacol 25: 367–371.
Fahim C, Stip E, Mancini-Marie A, Mensour B, Boulay LJ, Leroux JM et al (2005b). Brain activity during emotionally negative pictures in schizophrenia with and without flat affect: An fMRI study. Psychiatry Res 140: 1–15.
Fantino M, Hosotte J, Apfelbaum M (1986). An opioid antagonist, naltrexone, reduces preference for sucrose in humans. Am J Physiol 251: R91–R96.
Fe-Bornstein M, Watt SD, Gitlin MC (2002). Improvement in the level of psychosocial functioning in chronic pain patients with the use of risperidone. Pain Med 3: 128–131.
Fernandez-Real JM, Ricart W, Casamitjana R (1997). Lower cortisol levels after oral glucose in subjects with insulin resistance and abdominal obesity. Clin Endocrinol (Oxford) 47: 583–588.
Fessler DM (2003). The implications of starvation induced psychological changes for the ethical treatment of hunger strikers. J Med Ethics 29: 243–247.
Fetissov SO, Meguid MM, Sato T, Zhang LH (2002). Expression of dopaminergic receptors in the hypothalamus of lean and obese Zucker rats and food intake. Am J Physiol Regul Integr Comp Physiol 283: R905–R910.
Figlewicz DP (2003a). Adiposity signals and food reward: expanding the CNS roles of insulin and leptin. Am J Physiol Regul Integr Comp Physiol 284: R882–R982.
Figlewicz DP (2003b). Insulin, food intake, and reward. Semin Clin Neuropsychiatry 8: 82–93.
Figlewicz DP, Bennett J, Evans SB, Kaiyala K, Sipols AJ, Benoit SC (2004). Intraventricular insulin and leptin reverse place preference conditioned with high-fat diet in rats. Behav Neurosci 118: 479–487.
Finney GO (1989). Juvenile onset diabetes and schizophrenia? Lancet 2: 1214–1215.
Fishbain DA (1982). Pain insensitivity in psychosis. Ann Emerg Med 11: 630–632.
Floresco SB, West AR, Ash B, Moore H, Grace AA (2003). Afferent modulation of dopamine neuron firing differentially regulates tonic and phasic dopamine transmission. Nat Neurosci 6: 968–973.
Fontaine KR, Heo M, Harrigan EP, Shear CL, Lakshminarayanan M, Casey DE et al (2001). Estimating the consequences of anti-psychotic induced weight gain on health and mortality rate. Psychiatry Res 101: 277–288.
Food and Agriculture Organization (2002). Food and Agriculture Organization of the United Nations Statistical Database. http://apps.fao.org/default.jsp.
Foster-Powell K, Holt SH, Brand-Miller JC (2002). International table of glycemic index and glycemic load values: 2002. Am J Clin Nutr 76: 5–56.
Fruzzetti F, Bersi C, Parrini D, Ricci C, Genazzani AR (2002). Effect of long-term naltrexone treatment on endocrine profile, clinical features, and insulin sensitivity in obese women with polycystic ovary syndrome. Fertil Steril 77: 936–944.
Fulton S, Richard D, Woodside B, Shizgal P (2004). Food restriction and leptin impact brain reward circuitry in lean and obese Zucker rats. Behav Brain Res 155: 319–329.
Ganong WF (2000). Circumventricular organs: definition and role in the regulation of endocrine and autonomic function. Clin Exp Pharmacol Physiol 27: 422–427.
Garavan H, Pankiewicz J, Bloom A, Cho JK, Sperry L, Ross TJ et al (2000). Cue-induced cocaine craving: neuroanatomical specificity for drug users and drug stimuli. Am J Psychiatry 157: 1789–1798.
Gawin FH, Khalsa-Denison ME (1996). Is craving mood-driven or self-propelled? Sensitization and ‘street’ stimulant addiction. NIDA Res Monogr 163: 224–250.
Gerozissis K (2004). Brain insulin and feeding: a bi-directional communication. Eur J Pharmacol 490: 59–70.
Geschwind N (1977). Insensitivity to pain in psychotic patients. N Engl J Med 296: 1480.
Gispen-de Wied CC (2000). Stress in schizophrenia: an integrative view. Eur J Pharmacol 405: 375–384.
Goeders NE (2002). Stress and cocaine addiction. J Pharmacol Exp Ther 301: 785–789.
Goldstein DS, McEwen B (2002). Allostasis, homeostats, and the nature of stress. Stress 5: 55–58.
Goldstein RZ, Volkow ND (2002). Drug addiction and its underlying neurobiological basis: neuroimaging evidence for the involvement of the frontal cortex. Am J Psychiatry 159: 1642–1652.
Gorski ED, Willis KC (2003). Report of three case studies with olanzapine for chronic pain. J Pain 4: 166–168.
Gothelf D, Falk B, Singer P, Kairi M, Phillip M, Zigel L et al (2002). Weight gain associated with increased food intake and low habitual activity levels in male adolescent schizophrenic inpatients treated with olanzapine. Am J Psychiatry 159: 1055–1057.
Goto Y, Grace AA (2005). Dopamine-dependent interactions between limbic and prefrontal cortical plasticity in the nucleus accumbens: disruption by cocaine sensitization. Neuron 47: 255–266.
Grace AA (1991). Phasic versus tonic dopamine release and the modulation of dopamine system responsivity: a hypothesis for the etiology of schizophrenia. Neuroscience 41: 1–24.
Grace AA (2000). The tonic/phasic model of dopamine system regulation and its implications for understanding alcohol and psychostimulant craving. Addiction 95 (Suppl 2): S119–S128.
Graham KA, Perkins DO, Edwards LJ, Barrier Jr RC, Lieberman JA, Harp JB (2005). Effect of olanzapine on body composition and energy expenditure in adults with first-episode psychosis. Am J Psychiatry 162: 118–123.
Grant S, London ED, Newlin DB, Villemagne VL, Liu X, Contoreggi C et al (1996). Activation of memory circuits during cue-elicited cocaine craving. Proc Natl Acad Sci USA 93: 12040–12045.
Green AI, Salomon MS, Brenner MJ, Rawlins K (2002). Treatment of schizophrenia and comorbid substance use disorder. Curr Drug Targets CNS Neurol Disord 1: 129–139.
Green AI, Zimmet SV, Strous RD, Schildkraut JJ (1999). Clozapine for comorbid substance use disorder and schizophrenia: do patients with schizophrenia have a reward-deficiency syndrome that can be ameliorated by clozapine? Harvard Rev Psychiatry 6: 287–296.
Grigson PS (2002). Like drugs for chocolate: separate rewards modulated by common mechanisms? Physiol Behav 76: 389–395.
Grigson PS, Twining RC (2002). Cocaine-induced suppression of saccharin intake: a model of drug-induced devaluation of natural rewards. Behav Neurosci 116: 321–333.
Grimm JW, Lu L, Hayashi T, Hope BT, Su TP, Shaham Y (2003). Time-dependent increases in brain-derived neurotrophic factor protein levels within the mesolimbic dopamine system after withdrawal from cocaine: implications for incubationof cocaine craving. J Neurosci 23: 742–747.
Guillin O, Diaz J, Carroll P, Griffon N, Schwartz JC, Sokoloff P (2001). BDNF controls dopamine D3 receptor expresion and triggers behavioural sensitization. Nature 411: 86–89.
Gupta S (1992). Cross-national differences in the frequency and outcome of schizophrenia: a comparison of five hypotheses. Soc Psychiatry Psychiatr Epidemiol 27: 249–252.
Gur RE, McGrath C, Chan RM, Schroeder L, Turner T, Turetsky BI et al (2002). An fMRI study of facial emotion processing in patients with schizophrenia. Am J Psychiatry 159: 1992–1999.
Hajnal A, Norgren R (2002). Repeated access to sucrose augments dopamine turnover in the nucleus accumbens. Neuroreport 13: 2213–2216.
Hajnal A, Smith GP, Norgren R (2004). Oral sucrose stimulation increases accumbens dopamine in the rat. Am J Physiol Regul Integr Comp Physiol 286: R31–R37.
Hall FS, Drgonova J, Goeb M, Uhl GR (2003). Reduced behavioral effects of cocaine in heterozygous brain-derived neurotrophic factor (BDNF) knockout mice. Neuropsychopharmacology 28: 1485–1490.
Harding R, Leek BF (1973). Central projections of gastric afferent vagal inputs. J Physiol 228: 73–90.
Harris GC, Wimmer M, Aston-Jones G (2005). A role for lateral hypothalamic orexin neurons in reward seeking. Nature 437: 556–559.
Hartfield AW, Moore NA, Clifton PG (2003a). Effects of clozapine, olanzapine and haloperidol on the microstructure of ingestive behaviour in the rat. Psychopharmacology (Berlin) 167: 115–122.
Hartfield AW, Moore NA, Clifton PG (2003b). Serotonergic and histaminergic mechanisms involved in intralipid drinking? Pharmacol Biochem Behav 76: 251–258.
Haupt DW, Luber A, Maeda J, Melson AK, Schweiger JA, Newcomer JW (2005). Plasma leptin and adiposity during antipsychotic treatment of schizophrenia. Neuropsychopharmacology 30: 184–191.
Haupt DW, Newcomer JW (2002). Abnormalities in glucose regulation associated with mental illness and treatment. J Psychosom Res 53: 925–933.
Havel PJ (2001). Peripheral signals conveying metabolic information to the brain: short-term and long-term regulation of food intake and energy homeostasis. Exp Biol Med 226: 963–977.
Heinz A, Reimold M, Wrase J, Hermann D, Croissant B, Mundle G et al (2005). Correlation of stable elevations in striatal mu-opioid receptor availability in detoxified alcoholic patients with alcohol craving: a positron emission tomography study using carbon 11-labeled carfentanil. Arch Gen Psychiatry 62: 57–64.
Henderson DC, Cagliero E, Copeland PM, Borba CP, Evins E, Hayden D et al (2005). Glucose metabolism in patients with schizophrenia treated with atypical antipsychotic agents: a frequently sampled intravenous glucose tolerance test and minimal model analysis. Arch Gen Psychiatry 62: 19–28.
Hernandez L, Hoebel BG (1988). Food reward and cocaine increase extracellular dopamine in the nucleus accumbens as measured by microdialysis. Life Sci 42: 1705–1712.
Hetherington MM, Vervaet N, Blass E, Rolls BJ (1991). Failure of naltrexone to affect the pleasantness or intake of food. Pharmacol Biochem Behav 40: 185–190.
Himmerich H, Fulda S, Kunzel HE, Pfennig A, Dzaja A, Cummings DE et al (2005). Ghrelin plasma levels during psychopharmacological treatment. Neuropsychobiology 52: 11–16.
Hinton EC, Parkinson JA, Holland AJ, Arana FS, Roberts AC, Owen AM (2004). Neural contributions to the motivational control of appetite in humans. Eur J Neurosci 20: 1411–1418.
Holt RI, Peveler RC, Byrne CD (2004). Schizophrenia, the metabolic syndrome and diabetes. Diabetes Med 21: 515–523.
Homko CJ, Cheung P, Boden G (2003). Effects of free fatty acids on glucose uptake and utilization in healthy women. Diabetes 52: 487–491.
Hooley JM, Delgado ML (2001). Pain insensitivity in the relatives of schizophrenia patients. Schizophr Res 47: 265–273.
Horger BA, Iyasere CA, Berhow MT, Messer CJ, Nestler EJ, Taylor JR (1999). Enhancement of locomotor activity and conditioned reward to cocaine by brain-derived neurotrophic factor. J Neurosci 19: 4110–4122.
Horvath TL, Naftolin F, Kalra SP, Leranth C (1992). Neuropeptide-Y innervation of beta-endorphin-containing cells in the rat mediobasal hypothalamus: a light and electron microscopic double immunostaining analysis. Endocrinology 131: 2461–2467.
Horvitz JC (2000). Mesolimbocortical and nigrostriatal dopamine responses to salient non-reward events. Neuroscience 96: 651–656.
Hosojima H, Togo T, Odawara T, Hasegawa K, Miura S, Kato Y et al (2006). Early effects of olanzapine on serum levels of ghrelin, adiponectin and leptin in patients with schizophrenia. J Psychopharmacol 20: 75–79.
Huang EJ, Reichardt LF (2001). Neurotrophins: roles in neuronal development and function. Annu Rev Neurosci 24: 677–736.
Hyman SE (2005). Addiction: a disease of learning and memory. Am J Psychiatry 162: 1414–1422.
Ibanez-Rojo V, Palnca I, Irela LM, Oliveros SC, Caballero L, Baca E (1993). Case report 4: naltrexone-induced opiate withdrawal like syndrome in a non-addict obese woman. J Drug Dev 6: 75–76.
Imperato A, Cabib S, Puglisi-Allegra S (1993). Repeated stressful experiences differently affect the time-dependent responses of the mesolimbic dopamine system to the stressor. Brain Res 601: 333–336.
Imperato A, Puglisi-Allegra S, Casolini P, Zocchi A, Angelucci L (1989). Stress-induced enhancement of dopamine and acetylcholine release in limbic structures: role of corticosterone. Eur J Pharmacol 165: 337–338.
Isganaitis E, Lustig RH (2005). Fast food, central nervous system insulin resistance, and obesity. Arterioscler Thromb Vasc Biol 6: 245–260.
Itateyama E, Chiba S, Sakata T, Yoshimatsu H (2003). Hypothalamic neuronal histamine in genetically obese animals: its implication of leptin action in the brain. Exp Biol Med 228: 1132–1137.
Itoh K, Hashimoto K, Kumakiri C, Shimizu E, Iyo M (2004). Association between brain-derived neurotrophic factor 196 G/A polymorphism and personality traits in healthy subjects. Am J Med Genet B 124: 61–63.
Jablensky A, Sartorius N, Emberg G, Anker M, Korten A, Cooper JE et al (1992). Schizophrenia: manifestations, incidence and course in different cultures: a World Health Organization 10-country study. Psychol Med Monograph Suppl 20: 1–97.
Jenkins L, Malik MA, Lik M, Vaczi P (1996). Multiple cigarette burn wounds in a chronic paranoid schizophrenic. S Afr Med J 86: 572.
Jonas JM, Gold MS (1986). Naltrexone reverses bulimic symptoms. Lancet 1: 807.
Johnson DE, Yamazaki H, Ward KM, Schmidt AW, Lebel WS, Treadway JL et al (2005). Inhibitory effects of antipsychotics on carbachol-enhanced insulin secretion from perifused rat islets: role of muscarinic antagonism in antipsychotic-induced diabetes and hyperglycemia. Diabetes 54: 1552–1558.
Juckel G, Sass L, Heinz A (2003). Anhedonia, self-experience in schizophrenia, and implications for treatment. Pharmacopsychiatry 36: S176–S180.
Juckel G, Schlagenhauf F, Koslowski M, Wustenberg T, Villringer A, Knutson B et al (2006). Dysfunction of ventral striatal reward prediction in schizophrenia. Neuroimage 29: 409–416.
Kahn BB, Flier JS (2000). Obesity and insulin resistance. J Clin Invest 106: 473–481.
Kaiyala KJ, Prigeon RL, Kahn SE, Woods SC, Schwartz MW (2000). Obesity induced by a high-fat diet is associated with reduced brain insulin transport in dogs. Diabetes 49: 1525–1533.
Kalivas PW, Volkow ND (2005). The neural basis of addiction: a pathology of motivation and choice. Am J Psychiatry 162: 1403–1413.
Kalivas PW, Volkow ND, Seamans J (2005). Unmanageable motivation in addiction: a pathology in prefrontal–accumbens glutamate transmission. Neuron 45: 647–650.
Kalra SP, Dube MG, Pu S, Xu B, Horvath TL, Kalra PS (1999). Interacting appetite-regulating pathways in the hypothalamic regulation of body weight. Endocr Rev 20: 68–100.
Kalra SP, Kalra PS (2004). Overlapping and interactive pathways regulating appetite and craving. J Addict Dis 23: 5–21.
Kampov-Polevoy AB, Tsoi MV, Zvartau EE, Neznanov NG, Khalitov E (2001). Sweet liking and family history of alcoholism in hospitalized alcoholic and non-alcoholic patients. Alcohol Alcohol 36: 165–170.
Kane JM, Barrett EJ, Casey DE, Correll CU, Gelenberg AJ, Klein S et al (2004). Metabolic effects of treatment with atypical antipsychotics. J Clin Psychiatry 65: 1447–1455.
Kapur S (2003). Psychosis as a state of aberrant salience: a framework linking biology, phenomenology, and pharmacology in schizophrenia. Am J Psychiatry 160: 13–23.
Kapur S, Mamo D (2003). Half a century of antipsychotics and still a central role for dopamine D2 receptors. Prog Neuropsychopharmacol Biol Psychiatry 27: 1081–1090.
Katz E, Kluger Y, Rabinovici R, Stein D, Gimmon Z (1990). Acute surgical abdominal disease in chronic schizophrenic patients: a unique clinical problem. Isr J Med Sci 26: 275–277.
Kawasaki Y, Suzuki M, Nohara S, Hagino H, Takahashi T, Matsui M et al (2004). Structural brain differences in patients with schizophrenia and schizotypal disorder demonstrated by voxel-based morphometry. Eur Arch Psychiatry Clin Neurosci 254: 406–414.
Kelley AE (2004a). Memory and addiction: shared neural circuitry and molecular mechanisms. Neuron 44: 161–179.
Kelley AE (2004b). Ventral striatal control of appetitive motivation: role in ingestive behavior and reward-related learning. Neurosci Biobehav Rev 27: 765–776.
Kelley AE, Bakshi VP, Haber SN, Steininger TL, Will MJ, Zhang M (2002). Opioid modulation of taste hedonics within the ventral striatum. Physiol Behav 76: 365–377.
Kelley AE, Berridge KC (2002). The neuroscience of natural rewards: relevance to addictive drugs. J Neurosci 22: 3306–3311.
Kelley AE, Schiltz CA (2004). Accessories to addiction: G protein regulators play a key role in cocaine seeking and neuroplasticity. Neuron 42: 181–183.
Kelley AE, Schiltz CA, Landry CF (2005). Neural systems recruited by drug- and food-related cues: studies of gene activation in corticolimbic regions. Physiol Behav 86: 11–14.
Kelley AE, Will MJ, Steininger TL, Zhang M, Haber SN (2003). Restricted daily consumption of a highly palatable food (chocolate Ensure(R)) alters striatal enkephalin gene expression. Eur J Neurosci 18: 2592–2598.
Kernie SG, Liebl DJ, Parada LF (2000). BDNF regulates eating behavior and locomotor activity in mice. EMBO J 19: 1290–1300.
Kerns JG, Cohen JD, Macdonald III AW, Johnson MK, Stenger VA, Aizenstein H et al (2005). Decreased conflict- and error-related activity in the anterior cingulate cortex in subjects with schizophrenia. Am J Psychiatry 162: 1833–1839.
Khojainova N, Santiago-Palma J, Kornick C, Breitbart W, Gonzales GR (2002). Olanzapine in the management of cancer pain. J Pain Symptom Manage 23: 346–350.
Kinon BJ, Kaiser CJ, Ahmed S, Rotelli MD, Kollack-Walker S (2005). Association between early and rapid weight gain and change in weight over one year of olanzapine therapy in patients with schizophrenia and related disorders. J Clin Psychopharmacol 25: 255–258.
Kirk SL, Neill JC, Jones DN, Reynolds GP (2004). Ziprasidone suppresses olanzapine-induced increases in ingestive behaviour in the rat. Eur J Pharmacol 505: 253–254.
Kiser RS, Cohen HM, Freedenfeld RN, Jewell C, Fuchs PN (2001). Olanzapine for the treatment of fibromyalgia symptoms. J Pain Symptom Manage 22: 704–708.
Knable MB, Egan MF, Heinz A, Gorey J, Lee KS, Coppola R et al (1997). Altered dopaminergic function and negative symptoms in drug-free patients with schizophrenia. [123I]-iodobenzamide SPECT study. Br J Psychiatry 171: 574–577.
Kochhar S, Nwokike JN, Jankowitz B, Sholevar EH, Abed T, Baron DA (2002). Olanzapine overdose: a pediatric case report. J Child Adolesc Psychopharmacol 12: 351–353.
Kohen D (2004). Diabetes mellitus and schizophrenia: historical perspective. Br J Psychiatry Suppl 47: S64–S66.
Kohler CG, Turner TH, Bilker WB, Brensinger CM, Siegel SJ, Kanes SJ et al (2003). Facial emotion recognition in schizophrenia: intensity effects and error pattern. Am J Psychiatry 160: 1768–1774.
Koob GF, Le Moal M (2001). Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 24: 97–129.
Kringelbach ML (2005). The human orbitofrontal cortex: linking reward to hedonic experience. Nat Rev Neurosci 6: 691–702.
Kroeze WK, Hufeisen SJ, Popadak BA, Renock SM, Steinberg S, Ernsberger P et al (2003). H1-histamine receptor affinity predicts short-term weight gain for typical and atypical antipsychotic drugs. Neuropsychopharmacology 28: 519–526.
Krystal JH, Perry Jr EB, Gueorguieva R, Belger A, Madonick SH, Abi-Dargham A (2005). Comparative and interactive human psychopharmacologic effects of ketamine and amphetamine: implications for glutamatergic and dopaminergic model psychoses and cognitive function. Arch Gen Psychiatry 62: 985–994.
Kudoh A, Ishihara H, Matsuki A (2000). Current perception thresholds and postoperative pain in schizophrenic patients. Regul Anesth Pain Med 25: 475–479.
Kulhara P (1994). Outcome of schizophrenia: some transcultural observations with particular reference to developing countries. Eur Arch Psychiatry Clin Neurosci 244: 227–235.
Laimer M, Ebenbichler CF, Kranebitter M, Eder U, Mangweth B, Weiss E et al (2005). Olanzapine-induced hyperglycemia: role of humoral insulin resistance-inducing factors. J Clin Psychopharmacol 25: 183–185.
Lamberti JS, Crilly JF, Maharaj K, Olson D, Wiener K, Dvorin S et al (2004). Prevalence of diabetes mellitus among outpatients with severe mental disorders receiving atypical antipsychotic drugs. J Clin Psychiatry 65: 702–706.
Laruelle M (2000). The role of endogenous sensitization in the pathophysiology of schizophrenia: implications from recent brain imaging studies. Brain Res Brain Res Rev 31: 371–384.
Laruelle M, Abi-Dargham A (1999). Dopamine as the wind of the psychotic fire: new evidence from brain imaging studies. J Psychopharmacol 13: 358–371.
Laruelle M, Abi-Dargham A, Gil R, Kegeles L, Innis R (1999). Increased dopamine transmission in schizophrenia: relationship to illness phases. Biol Psychiatry 46: 56–72.
Laruelle M, Abi-Dargham A, van Dyck CH, Gil R, D’Souza CD, Erdos J et al (1996). Single photon emission computerized tomography imaging of amphetamine-induced dopamine release in drug-free schizophrenic subjects. Proc Natl Acad Sci USA 93: 9235–9240.
Laruelle M, Kegeles LS, Abi-Dargham A (2003). Glutamate, dopamine, and schizophrenia: from pathophysiology to treatment. Ann NY Acad Sci 1003: 138–158.
Lauer M, Beckmann H (1997). The human striatum in schizophrenia. I. Increase in overall relative striatal volume in schizophrenics. Psychiatry Res 68: 87–98.
Lauer M, Senitz D, Beckmann H (2001). Increased volume of the nucleus accumbens in schizophrenia. J Neural Transm 108: 645–660.
Lehmann H, Nair NP, Kline NS (1979). Beta-endorphin and naloxone in psychiatric patients: clinical and biological effects. Am J Psychiatry 136: 762–766.
Lesscher HM, Bailey A, Burbach JP, Van Ree JM, Kitchen I, Gerrits MA (2003). Receptor-selective changes in mu-, delta- and kappa-opioid receptors after chronic naltrexone treatment in mice. Eur J Neurosci 17: 1006–1012.
Levine AS, Kotz CM, Gosnell BA (2003). Sugars and fats: the neurobiology of preference. J Nutr 133: 831S–834S.
Lewis DA, Glantz LA, Pierri JN, Sweet RA (2003). Altered cortical glutamate neurotransmission in schizophrenia: evidence from morphological studies of pyramidal neurons. Ann NY Acad Sci 1003: 102–112.
Lewkowski MD, Ditto B, Roussos M, Young SN (2003). Sweet taste and blood pressure-related analgesia. Pain 106: 181–186.
Lieberman JA (2000). Clinical research in the age of neuroscience. Neuropsychopharmacology 22: 1–3.
Lieberman JA, Sheitman BB, Kinon BJ (1997). Neurochemical sensitization in the pathophysiology of schizophrenia: deficits and dysfunction in neuronal regulation and plasticity. Neuropsychopharmacology 17: 205–229.
Lieberman JA, Stroup TS, McEvoy JP, Swartz MS, Rosenheck RA, Perkins DO et al (2005). Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE). N Engl J Med 353: 1209–1223 (Neuropsychopharmacology 22: 1–3).
Lindenmayer JP, Czobor P, Volavka J, Citrome L, Sheitman B, McEvoy JP et al (2003). Changes in glucose and cholesterol levels in patients with schizophrenia treated with typical or atypical antipsychotics. Am J Psychiatry 160: 290–296.
Lindqvist A, de la Cour CD, Stegmark A, Hakanson R, Erlanson-Albertsson C (2005). Overeating of palatable food is associated with blunted leptin and ghrelin responses. Regul Peptide 130: 123–132.
Lindstrom LH, Besev G, Gunne LM, Terenius L (1986). CSF levels of receptor-active endorphins in schizophrenic patients: correlations with symptomatology and monoamine metabolites. Psychiatry Res 19: 93–100.
Lindstrom LH, Widerlov E, Gunne LM, Wahlstrom A, Terenius L (1978). Endorphins in human cerebrospinal fluid: clinical correlations to some psychotic states. Acta Psychiatr Scand 57: 153–164.
Little KY, Zhang L, Desmond T, Frey KA, Dalack GW, Cassin BJ (1999). Striatal dopaminergic abnormalities in human cocaine users. Am J Psychiatry 156: 238–245.
Lublin H, Eberhard J, Levander S (2005). Current therapy issues and unmet clinical needs in the treatment of schizophrenia: a review of the new generation antipsychotics. Int Clin Psychopharmacol 20: 183–198.
Luo C, Xu H, Li XM (2004). Post-stress changes in BDNF and Bcl-2 immunoreactivities in hippocampal neurons: effect of chronic administration of olanzapine. Brain Res 1025: 194–202.
Lyons WE, Mamounas LA, Ricaurte GA, Coppola V, Reid SW, Bora SH et al (1999). Brain derived neurotrophic factor-deficient mice develop aggressiveness nd hyperphagia in conjunction with brain serotonergic abnormalities. Proc Natl Acad Sci USA 96: 15239–15244.
MacDonald AF, Billington CJ, Levine AS (2004). Alterations in food intake by opioid and dopamine signaling pathways between the ventral tegmental area and the shell of the nucleus accumbens. Brain Res 1018: 78–85.
MacIntosh CG, Sheehan J, Davani N, Morley JE, Horowitz M, Chapman IM (2001). Effects of aging on the opioid modulation of feeding in humans. J Am Geriatr Soc 49: 1518–1524.
Makarenko IG, Meguid MM, Ugrumov MV (2002). Distribution of serotonin 5-hydroxytriptamine 1B (5-HT(1B)) receptors in the normal rat hypothalamus. Neurosci Lett 328: 155–159.
Malhotra AK, Pinals DA, Adler CM, Elman I, Clifton A, Pickar D et al (1997). Ketamine-induced exacerbation of psychotic symptoms and cognitive impairment in neuroleptic-free schizophrenics. Neuropsychopharmacology 17: 141–150.
Mann K, Rossbach W, Muller MJ, Muller-Siecheneder F, Pott T, Linde I et al (2006). Nocturnal hormone profiles in patients with schizophrenia treated with olanzapine. Psychoneuroendocrinology 31: 256–264.
Marchand WE (1955). Occurrence of painless myocardial infarction in psychotic patients. N Engl J Med 253: 51–55.
Marchesi GF, Santone G, Cotani P, Giordano A, Chelli F (1995). The therapeutic role of naltrexone in negative symptom schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry 19: 1239–1249.
Marder SR, Essock SM, Miller AL, Buchanan RW, Casey DE, Davis JM et al (2004). Physical health monitoring of patients with schizophrenia. Am J Psychiatry 161: 1334–1349.
Marrazzi MA, Bacon JP, Kinzie J, Luby ED (1995a). Naltrexone use in the treatment of anorexia nervosa and bulimia nervosa. Int Clin Psychopharmacol 10: 163–172.
Marrazzi MA, Kinzie J, Luby ED (1995b). A detailed longitudinal analysis on the use of naltrexone in the treatment of bulimia. Int Clin Psychopharmacol 10: 173–176.
Martin-Soelch C, Chevalley AF, Kunig G, Missimer J, Magyar S, Mino A et al (2001a). Changes in reward-induced brain activation in opiate addicts. Eur J Neurosci 14: 1360–1368.
Martin-Soelch C, Leenders KL, Chevalley AF, Missimer J, Kunig G, Magyar S et al (2001b). Reward mechanisms in the brain and their role in dependence: evidence from neurophysiological and neuroimaging studies. Brain Res Brain Res Rev 36: 139–149.
Martin-Solch C, Magyar S, Kunig G, Missimer J, Schultz W, Leenders KL (2001). Changes in brain activation associated with reward processing in smokers and nonsmokers. A positron emission tomography study. Exp Brain Res 139: 278–286.
Martins JM, Trinca A, Afonso A, Carreiras F, Falcao J, Nunes JS et al (2001). Psychoneuroendocrine characteristics of common obesity clinical subtypes. Int J Obes Relat Metab Disord 25: 24–32.
Masaki T, Chiba S, Yasuda T, Noguchi H, Kakuma T, Watanabe T et al (2004). Involvement of hypothalamic histamine H1 receptor in the regulation of feeding rhythm and obesity. Diabetes 53: 2250–2260.
Masand PS (1999). Relative weight gain among antipsychotics. J Clin Psychiatry 60: 706–708.
Maslow AH (1943). A preface to motivation theory. Psychosomatic Med 5: 85–92.
Mayo-Smith W, Hayes CW, Biller BM, Klibanski A, Rosenthal H, Rosenthal DI (1989). Body fat distribution measured with CT: correlations in healthy subjects, patients with anorexia nervosa, and patients with Cushing syndrome. Radiology 170: 515–518.
McClure SM, Daw ND, Montague PR (2003). A computational substrate for incentive salience. Trends Neurosci 26: 423–428.
McCreadie R, Macdonald E, Blacklock C, Tilak-Singh D, Wiles D, Halliday J et al (1998). Dietary intake of schizophrenic patients in Nithsdale, Scotland: case–control study. BMJ 317: 784–785.
McCreadie RG (2003). Diet, smoking and cardiovascular risk in people with schizophrenia: descriptive study. Br J Psychiatry 183: 534–539.
McCreadie RG, Kelly C, Connolly M, Williams S, Baxter G, Lean M et al (2005). Dietary improvement in people with schizophrenia: randomised controlled trial. Br J Psychiatry 187: 346–351.
McGuire PK, Shah GM, Murray RM (1993). Increased blood flow in Broca's area during auditory hallucinations in schizophrenia. Lancet 342: 703–706.
McIntosh A, Lawrie S (2004). Cross-national differences in diet, the outcome of schizophrenia and the prevalence of depression: you are (associated with) what you eat. Br J Psychiatry 184: 381–382.
McKee HA, D’Arcy PF, Wilson PJ (1986). Diabetes and schizophrenia—a preliminary study. J Clin Hosp Pharm 11: 297–299.
McNicholas LF, Martin WR (1984). New and experimental therapeutic roles for naloxone and related opioid antagonists. Drugs 27: 81–93.
Melchior JC, Fantino M, Rozen R, Igoin L, Rigaud D, Apfelbaum M (1989). Effects of a low dose of naltrexone on glucose-induced allesthesia and hunger in humans. Pharmacol Biochem Behav 32: 117–121.
Melkersson K (2004). Clozapine and olanzapine, but not conventional antipsychotics, increase insulin release in vitro. Eur Neuropsychopharmacol 14: 115–119.
Melkersson K (2005). Differences in prolactin elevation and related symptoms of atypical antipsychotics in schizophrenic patients. J Clin Psychiatry 66: 761–767.
Melkersson K, Jansson E (2005). The atypical antipsychotics quetiapine, risperidone and ziprasidone do not increase insulin release in vitro. Neuro Endocrinol Lett 26: 205–208.
Melkersson K, Khan A, Hilding A, Hulting AL (2001). Different effects of antipsychotic drugs on insulin release in vitro. Eur Neuropsychopharmacol 11: 327–332.
Melkersson KI, Dahl ML (2003). Relationship between levels of insulin or triglycerides and serum concentrations of the atypical antipsychotics clozapine and olanzapine in patients on treatment with therapeutic doses. Psychopharmacology (Berlin) 170: 157–166.
Meltzer HY (1999). The role of serotonin in antipsychotic drug action. Neuropsychopharmacology 21: 106S–115S.
Mercer LP, Kelley DS, Humphries LL, Dunn JD (1994). Manipulation of central nervous system histamine or histaminergic receptors (H1) affects food intake in rats. J Nutr 124: 1029–1036.
Mercer ME, Holder MD (1997a). Antinociceptive effects of palatable sweet ingesta on human responsivity to pressure pain. Physiol Behav 61: 311–318.
Mercer ME, Holder MD (1997b). Food cravings, endogenous opioid peptides, and food intake: a review. Appetite 29: 325–352.
Meredith GF, Callen S, Scheuer DA (2002). Brain-derived neurotroic factor expression is increased in the rat amygdala, piriform cortex and hypothalamus following repeated amphetmine administration. Brain Res 949: 218–227.
Mikesell MJ, Sobell JL, Sommer SS, McMurray CT (1996). Identification of a missense mutation and several polymorphisms in the proenkephalin A gene of schizophrenic patients. Am J Med Genet 67: 459–467.
Mizuno TM, Funabashi T, Kleopoulos SP, Mobbs CV (2004). Specific preservation of biosynthetic responses to insulin in adipose tissue may contribute to hyperleptinemia in insulin-resistant obese mice. J Nutr 134: 1045–1050.
Moberg PJ, Arnold SE, Doty RL, Kohler C, Kanes S, Seigel S et al (2003). Impairment of odor hedonics in men with schizophrenia. Am J Psychiatry 160: 1784–1789.
Molteni R, Barnard RJ, Ying Z, Roberts CK, Gomez-Pinilla F (2002). A high-fat, refined sugar diet reduces hippocampal brain-derived neurotrophic factor, neuronal plasticity, and learning. Neuroscience 112: 803–814.
Molteni R, Wu A, Vaynman S, Ying Z, Barnard RJ, Gomez-Pinilla F (2004). Exercise reverses the harmful effects of consumption of a high-fat diet on synaptic and behavioral plasticity associated to the action of brain-derived neurotrophic factor. Neuroscience 123: 429–440.
Montague PR, Hyman SE, Cohen JD (2004). Computational roles for dopamine in behavioural control. Nature 431: 760–767.
Monteleone P, Fabrazzo M, Tortorella A, La Pia S, Maj M (2002). Pronounced early increase in circulating leptin predicts a lower weight gain during clozapine treatment. J Clin Psychopharmacol 22: 424–426.
Moore H, West AR, Grace AA (1999). The regulation of forebrain dopamine transmission: relevance to the pathophysiology and psychopathology of schizophrenia. Biol Psychiatry 46: 40–55.
Morley JE (1987). Neuropeptide regulation of appetite and weight. Endocr Rev 8: 256–287.
Morton GJ, Blevins JE, Williams DL, Niswender KD, Gelling RW, Rhodes CJ et al (2005). Leptin action in the forebrain regulates the hindbrain response to satiety signals. J Clin Invest 115: 703–710.
Mukherjee S, Decina P, Bocola V, Saraceni F, Scapicchio PL (1996). Diabetes mellitus in schizophrenic patients. Compr Psychiatry 37: 68–73.
Mukherjee S, Schnur DB, Reddy R (1989). Family history of type 2 diabetes in schizophrenic patients. Lancet 1: 495.
Muller DJ, Muglia P, Fortune T, Kennedy JL (2004). Pharmacogenetics of antipsychotic-induced weight gain. Pharmacol Res 49: 309–329.
Murashita M, Kusumi I, Inoue T, Takahashi Y, Hosoda H, Kangawa K et al (2005). Olanzapine increases plasma ghrelin level in patients with schizophrenia. Psychoneuroendocrinology 30: 106–110.
Murthy BV, Narayan B, Nayagam S (2004). Reduced perception of pain in schizophrenia: its relevance to the clinical diagnosis of compartment syndrome. Injury 35: 1192–1193.
Meyer JM (2002). Awareness of obesity and weight issues among chronically mentally ill inpatients: a pilot study. Ann Clin Psychiatry 14: 39–45.
Nakagawa T, Ogawa Y, Ebihara K, Yamanaka M, Tsuchida A, Taiji M et al (2003). Anti-obesity and anti-diabetic effects of brain-derived neurotrophic factor in rodent models of leptin resistance. Int J Obes Relat Metab Disord 27: 557–565.
Nakagawa T, Tsuchida A, Itakura Y, Nonomura T, Ono M, Hirota F et al (2000). Brain-derived neurotrophic factor regulates glucose metabolism by modulating energy balance in diabetic mice. Diabetes 49: 436–444.
National Institute of Diabetes and Digestive and Kidney Diseases (2005). http://win.niddk.nih.gov/statistics/index.htm#preval.
Nemeroff CB, Bissette G (1988). Neuropeptides, dopamine, and schizophrenia. Ann NY Acad Sci 537: 273–291.
Nestler FJ (2004). Molecular mechanisms of drug addiction. Neuropharmacology Suppl 1: 24–32.
Newcomer JW (2005). Second-generation (atypical) antipsychotics and metabolic effects: a comprehensive literature review. CNS Drugs. 19 (Suppl 1): 1–93.
Newcomer JW, Haupt DW, Fucetola R, Melson AK, Schweiger JA, Cooper BP et al (2002). Abnormalities in glucose regulation during antipsychotic treatment of schizophrenia. Arch Gen Psychiatry 59: 337–345.
Nishikawa T, Tsuda A, Tanaka M, Nishikawa M, Koga I, Uchida Y (1994a). Decreased polydipsia in schizophrenic patients treated with naloxone. Am J Psychiatry 151: 947.
Nishikawa T, Tsuda A, Tanaka M, Nishikawa M, Koga I, Uchida Y (1994b). Naloxone attenuates drinking behavior in psychiatric patients displaying self-induced water intoxication. Prog Neuropsychopharmacol Biol Psychiatry 18: 149–153.
Niswender KD, Baskin DG, Schwartz MW (2004). Insulin and its evolving partnership with leptin in the hypothalamic control of energy homeostasis. Trends Endocrinol Metab 15: 362–369.
Nylander I, Terenius LH (1987). Dopamine receptors mediate alterations in striato-nigral dynorphin and substance P pathways. Neuropharmacology 26: 1295–1302.
Odeleye OE, de Courten M, Pettitt DJ, Ravussin E (1997). Fasting hyperinsulinemia is a predictor of increased body weight gain and obesity in Pima Indian children. Diabetes 46: 1341–1345.
O’Malley GF, Seifert S, Heard K, Daly F, Dart RC (1999). Olanzapine overdose mimicking opioid intoxication. Ann Emerg Med 34: 279–281.
O’Mara NB, Wesley LC (1994). Naltrexone in the treatment of alcohol dependence. Ann Pharmacother 28: 210–211.
Ono M, Itakura Y, Nonomura T, Nakagawa T, Nakayama C, Taiji M et al (2000). Intermittent administration of brain-derived neurotrophic factor ameliorates glucose metabolism in obese diabetic mice. Metabolism 49: 129–133.
Ota M, Mori K, Nakashima A, Kaneko YS, Fujiwara K, Itoh M et al (2002). Peripheral injection of risperidone, an atypical antipsychotic, alters the bodyweight gain of rats. Clin Exp Pharmacol Physiol 29: 980–989.
Palenzona S, Meier PJ, Kupferschmidt H, Rauber-Luethy C (2004). The clinical picture of olanzapine poisoning with special reference to fluctuating mental status. J Toxicol Clin Toxicol 42: 27–32.
Palik E, Birkas KD, Faludi G, Karadi I, Cseh K (2005). Correlation of serum ghrelin levels with body mass index and carbohydrate metabolism in patients treated with atypical antipsychotics. Diabetes Res Clin Pract 68: S60–S64.
Papakostas GI, Petersen T, Iosifescu DV, Burns AM, Nierenberg AA, Alpert JE et al (2005). Obesity among outpatients with major depressive disorder. Int J Neuropsychopharmacol 8: 59–63.
Papp M, Willner P, Muscat R (1991). An animal model of anhedonia: attenuation of sucrose consumption and place preference conditioning by chronic unpredictable mild stress. Psychopharmacology (Berlin) 104: 255–259.
Paradiso S, Andreasen NC, Crespo-Facorro B, O’Leary DS, Watkins GL, Boles Ponto LL et al (2003). Emotions in unmedicated patients with schizophrenia during evaluation with positron emission tomography. Am J Psychiatry 160: 1775–1783.
Pecina S, Berridge KC (1995). Central enhancement of taste pleasure by intraventricular morphine. Neurobiology (Bp) 3: 269–280.
Pecina S, Cagniard B, Berridge KC, Aldridge JW, Zhuang X (2003). Hyperdopaminergic mutant mice have higher ‘wanting’ but not ‘liking’ for sweet rewards. J Neurosci 23: 9395–9402.
Pedersen SB, Jonler M, Richelsen B (1994). Characterization of regional and gender differences in glucocorticoid receptors and lipoprotein lipase activity in human adipose tissue. J Clin Endocrinol Metab 78: 1354–1359.
Peet M (2004a). Diet, diabetes and schizophrenia: review and hypothesis. Br J Psychiatry 47: S102–S105.
Peet M (2004b). International variations in the outcome of schizophrenia and the prevalence of depression in relation to national dietary practices: an ecological analysis. Br J Psychiatry 184: 404–408.
Peet M (2004c). Nutrition and schizophrenia: beyond omega-3 fatty acids. Prostaglandins Leukot Essent Fatty Acids 70: 417–422.
Peruzzo B, Pastor FE, Blazquez JL, Schobitz K, Pelaez B, Amat P et al (2000). A second look at the barriers of the medial basal hypothalamus. Exp Brain Res 132: 10–26.
Petrakis IL, O’Malley S, Rounsaville B, Poling J, McHugh-Strong C, Krystal JH (2004). Naltrexone augmentation of neuroleptic treatment in alcohol abusing patients with schizophrenia. Psychopharmacology (Berlin) 172: 291–297.
Pfefferbaum A, Ford JM, White PM, Roth WT (1989). P3 in schizophrenia is affected by stimulus modality, response requirements, medication status, and negative symptoms. Arch Gen Psychiatry 46: 1035–1044.
Phillips ML, Williams L, Senior C, Bullmore ET, Brammer MJ, Andrew C et al (1999). A differential neural response to threatening and non-threatening negative facial expressions in paranoid and non-paranoid schizophrenics. Psychiatry Res 92: 11–31.
Phillips ML, Young AW, Scott SK, Calder AJ, Andrew C, Giampietro V et al (1998). Neural responses to facial and vocal expressions of fear and disgust. Proc R Soc Lond Ser B 265: 1809–1817.
Phillips WJ (1994). Starvation and survival: some military considerations. Mil Med 159: 513–516.
Pickar D, Vartanian F, Bunney Jr WE, Maier HP, Gastpar MT, Prakash R et al (1982). Short-term naloxone administration in schizophrenic and manic patients. A World Health Organization Collaborative Study. Arch Gen Psychiatry 39: 313–319.
Pierce RC, Kalivas PW (1997). A circuitry model of the expression of behavioral sensitization to amphetamine-like psychostimulants. Brain Res Brain Res Rev 25: 192–216.
Porte Jr D, Baskin DG, Schwartz MW (2005). Insulin signaling in the central nervous system: a critical role in metabolic homeostasis and disease from C. elegans to humans. Diabetes 54: 1264–1276.
Pouzet B, Mow T, Kreilgaard M, Velschow S (2003). Chronic treatment with antipsychotics in rats as a model for antipsychotic-induced weight gain in human. Pharmacol Biochem Behav 75: 133–140.
Powell KJ, Abul-Husn NS, Jhamandas A, Olmstead MC, Beninger RJ, Jhamandas K (2002). Paradoxical effects of the opioid antagonist naltrexone on morphine analgesia, tolerance, and reward in rats. J Pharmacol Exp Ther 300: 588–596.
Prentice AM, Jebb SA (2003). Fast foods, energy density and obesity: a possible mechanistic link. Obes Rev 4: 187–194.
Puglisi-Allegra S, Imperato A, Angelucci L, Cabib S (1991). Acute stress induces time-dependent responses in dopamine mesolimbic system. Brain Res 554: 217–222.
Rada P, Avena NM, Hoebel BG (2005). Daily bingeing on sugar repeatedly releases dopamine in the accumbens shell. Neuroscience 134: 737–744.
Rebuffe-Scrive M, Lundholm K, Bjorntorp P (1985). Glucocorticoid hormone binding to human adipose tissue. Eur J Clin Invest 15: 267–271.
Regier DA, Farmer ME, Rae DS, Locke BZ, Keith SJ, Judd LL et al (1990). Comorbidity of mental disorders with alcohol and other drug abuse. Results from the Epidemiologic Catchment Area (ECA) Study. JAMA 264: 2511–2518.
Reynolds GP, Templeman LA, Zhang ZJ (2005). The role of 5-HT2C receptor polymorphisms in the pharmacogenetics of antipsychotic drug treatment. Prog Neuropsychopharmacol Biol Psychiatry 29: 1021–1028.
Reynolds GP, Zhang ZJ, Zhang XB (2002). Association of antipsychotic drug-induced weight gain with a 5-HT2C receptor gene polymorphism. Lancet 359: 2086–2087.
Richelson E (1999). Receptor pharmacology of neuroleptics: relation to clinical effects. J Clin Psychiatry 60 (Suppl 10): 5–14.
Richelson E, Souder T (2000). Binding of antipsychotic drugs to human brain receptors focus on newer generation compounds. Life Sci 68: 29–39.
Rimon R, Terenius L, Kampman R (1980). Cerebrospinal fluid endorphins in schizophrenia. Acta Psychiatr Scand 61: 395–403.
Rios M, Fan G, Fekete C, Kelly J, Bates B, Kuehn R et al (2001). Conditional deletion of brain-derived neurotropic factor in the postnatal brain leads to obesity and hyperactivity. Mol Endocrinol 10: 1748–1757.
Rodefer JS, Campbell UC, Cosgrove KP, Carroll ME (1999). Naltrexone pretreatment decreases the reinforcing effectiveness of ethanol and saccharin but not PCP or food under concurrent progressive-ratio schedules in rhesus monkeys. Psychopharmacology (Berlin) 141: 436–446.
Rolls BJ, Castellanos VH, Halford JC, Kilara A, Panyam D, Pelkman CL et al (1998). Volume of food consumed affects satiety in men. Am J Clin Nutr 67: 1170–1177.
Rosenthal SH, Porter KA, Coffey B (1990). Pain insensitivity in schizophrenia. Case report and review of the literature. Gen Hosp Psychiatry 12: 319–322.
Roth-Deri I, Zangen A, Aleli M, Goelman RG, Pelled G, Nakash R et al (2003). Effect of experimenter-delivered and self-administered cocaine on extracellular beta-endorphin levels in the nucleus accumbens. J Neurochem 84: 930–938.
Ryan MC, Collins P, Thakore JH (2003). Impaired fasting glucose tolerance in first-episode, drug-naive patients with schizophrenia. Am J Psychiatry 160: 284–289.
Ryan MC, Flanagan S, Kinsella U, Keeling F, Thakore JH (2004). The effects of atypical antipsychotics on visceral fat distribution in first episode, drug-naive patients with schizophrenia. Life Sci 74: 1999–2008.
Ryan MC, Thakore JH (2002). Physical consequences of schizophrenia and its treatment: the metabolic syndrome. Life Sci 71: 239–257.
Sabri O, Erkwoh R, Schreckenberger M, Owega A, Sass H, Buell U (1997). Correlation of positive symptoms exclusively to hyperperfusion or hypoperfusion of cerebral cortex in never-treated schizophrenics. Lancet 349: 1735–1739.
Sakata T, Yoshimatsu H, Kurokawa M (1997). Hypothalamic neuronal histamine: implications of its homeostatic control of energy metabolism. Nutrition 13: 403–411.
Saper CB, Chou TC, Elmquist JK (2002). The need to feed: homeostatic and hedonic control of eating. Neuron 36: 199–211.
Sartorius N, Gulbinat W, Harrison G, Laska E, Siegel C (1996). Long-term follow-up of schizophrenia in 16 countries. A description of the International Study of Schizophrenia conducted by the World Health Organization. Soc Psychiatry Psychiatr Epidemiol 31: 249–258.
Schad CA, Justice Jr JB, Holtzman SG (2002). Endogenous opioids in dopaminergic cell body regions modulate amphetamine-induced increases in extracellular dopamine levels in the terminal regions. J Pharmacol Exp Ther 300: 932–938.
Schmauss C, Emrich HM (1985). Dopamine and the action of opiates: a reevaluation of the dopamine hypothesis of schizophrenia. With special consideration of the role of endogenous opioids in the pathogenesis of schizophrenia. Biol Psychiatry 20: 1211–1231.
Schneider F, Weiss U, Kessler C, Salloum JB, Posse S, Grodd W et al (1998). Differential amygdala activation in schizophrenia during sadness. Schizophr Res 34: 133–142.
Schoenbaum GM, Martin RJ, Roane DS (1989). Relationships between sustained sucrose-feeding and opioid tolerance and withdrawal. Pharmacol Biochem Behav 34: 911–914.
Schoenbaum GM, Martin RJ, Roane DS (1990). Discontinuation of sustained sucrose-feeding aggravates morphine withdrawal. Brain Res Bull 24: 565–568.
Schreiber S, Backer MM, Weizman R, Pick CG (1997). Augmentation of opioid induced antinociception by the atypical antipsychotic drug risperidone in mice. Neurosci Lett 228: 25–28.
Schreiber S, Getslev V, Backer MM, Weizman R, Pick CG (1999). The atypical neuroleptics clozapine and olanzapine differ regarding their antinociceptive mechanisms and potency. Pharmacol Biochem Behav 64: 75–80.
Schultz W (2001). Reward signaling by dopamine neurons. Neuroscientist 7: 293–302.
Schwartz MW, Niswender KD (2004). Adiposity signaling and biological defense against weight gain: absence of protection or central hormone resistance? J Clin Endocrinol Metab 89: 5889–5897.
Schwartz MW, Porte Jr D (2005). Diabetes, obesity, and the brain. Science 307: 375–379.
Schwartz MW, Woods SC, Porte Jr D, Seeley RJ, Baskin DG (2000). Central nervous system control of food intake. Nature 404: 661–671.
Schwartz MW, Woods SC, Seeley RJ, Barsh GS, Baskin DG, Leibel RL (2003). Is the energy homeostasis system inherently biased toward weight gain? Diabetes 52: 232–238.
Seeman P, Corbett R, Van Tol HH (1997). Atypical neuroleptics have low affinity for dopamine D2 receptors or are selective for D4 receptors. Neuropsychopharmacology 16: 93–110.
Segato FN, Castro-Souza C, Segato EN, Morato S, Coimbra NC (1997). Sucrose ingestion causes opioid analgesia. Braz J Med Biol Res 30: 981–984.
Selemon LD, Kleinman JE, Herman MM, Goldman-Rakic PS (2002). Smaller frontal gray matter volume in postmortem schizophrenic brains. Am J Psychiatry 159: 1983–1991.
Selye H (1976). The Stress of Life. McGraw-Hill: New York.
Shaham Y, Shalev U, Lu L, De Wit H, Stewart J (2002). The reinstatement model of drug relapse: history, methodology and major findings. Psychopharmacology (Berlin) 168: 3–20.
Shergill SS, Brammer MJ, Williams SC, Murray RM, McGuire PK (2000). Mapping auditory hallucinations in schizophrenia using functional magnetic resonance imaging. Arch Gen Psychiatry 57: 1033–1038.
Shufman EN, Porat S, Witztum E, Gandacu D, Bar-Hamburger R, Ginath Y (1994). The efficacy of naltrexone in preventing reabuse of heroin after detoxification. Biol Psychiatry 35: 935–945.
Sigal RJ, El-Hashimy M, Martin BC, Soeldner JS, Krolewski AS, Warram JH (1997). Acute postchallenge hyperinsulinemia predicts weight gain: a prospective study. Diabetes 46: 1025–1029.
Sigmundsson T, Suckling J, Maier M, Williams S, Bullmore E, Greenwood K et al (2001). Structural abnormalities in frontal, temporal, and limbic regions and interconnecting white matter tracts in schizophrenic patients with prominent negative symptoms. Am J Psychiatry 158: 234–243.
Silberstein SD, Peres MF, Hopkins MM, Shechter AL, Young WB, Rozen TD (2002). Olanzapine in the treatment of refractory migraine and chronic daily headache. Headache 42: 515–518.
Silbersweig DA, Stern E, Frith C, Cahill C, Holmes A, Grootoonk S et al (1995). A functional neuroanatomy of hallucinations in schizophrenia. Nature 378: 176–179.
Simpson GM, Weiden P, Pigott T, Murray S, Siu CO, Romano SJ (2005). Six-month, blinded, multicenter continuation study of ziprasidone versus olanzapine in schizophrenia. Am J Psychiatry 162: 1535–1538.
Sinha R (2001). How does stress increase risk of drug abuse and relapse? Psychopharmacology (Berlin) 158: 343–359.
Siuciak JA, Altar CA, Wiegand SJ, Lindsay RM. (1994). Antinociceptive effect of brain-derived neurotrophic factor and neurotrophin-3. Brain Res 633: 326–330.
Siuciak JA, Wong V, Pearsall D, Wiegand SJ, Lindsay RM. (1995). BDNF produces analgesia in the formalin test and modifies neuropeptide levels in rat brain and spinal cord areas associated with nociception. Eur J Neurosci 7: 663–670.
Sivam SP (1989). Cocaine selectively increases striatonigral dynorphin levels by a dopaminergic mechanism. J Pharmacol Exp Ther 250: 818–824.
Skidmore PM, Yarnell JW (2004). The obesity epidemic: prospects for prevention. Q J Med 97: 817–825.
Sklair-Tavron L, Shi WX, Lane SB, Harris HW, Bunney BS, Nestler EJ (1996). Chronic morphine induces visible changes in the morphology of mesolimbic dopamine neurons. Proc Natl Acad Sci USA 93: 11202–11207.
Solomon RL, Corbit JD (1974). An opponent-process theory of motivation. I. Temporal dynamics of affect. Psychol Rev 81: 119–145.
Soyka M, Koch W, Moller HJ, Ruther T, Tatsch K (2005). Hypermetabolic pattern in frontal cortex and other brain regions in unmedicated schizophrenia patients: results from a FDG-PET study. Eur Arch Psychiatry Clin Neurosci 255: 308–312.
Spangler R, Wittkowski KM, Goddard NL, Avena NM, Hoebel BG, Leibowitz SF (2004). Opiate-like effects of sugar on gene expression in reward areas of the rat brain. Brain Res Mol Brain Res 124: 134–142.
Sporn AL, Bobb AJ, Gogtay N, Stevens H, Greenstein DK, Clasen LS et al (2005). Hormonal correlates of clozapine-induced weight gain in psychotic children: an exploratory study. J Am Acad Child Adolesc Psychiatry 44: 925–933.
Stahl SM (2002). Essential Psychopharmacology of Antipsychotics and Mood Stabilizer. Cambride University Press: Cambridge, 137pp.
Stahl SM, Grady MM (2004). A critical review of atypical antipsychotic utilization: comparing monotherapy with polypharmacy and augmentation. Curr Med Chem 11: 313–327.
Stone WS, Faraone SV, Su J, Tarbox SI, Van Eerdewegh P, Tsuang MT (2004). Evidence for linkage between regulatory enzymes in glycolysis and schizophrenia in a multiplex sample. Am J Med Genet B 15: 5–10.
Steketee JD (2003). Neurotransmitter systems of the medial prefrontal cortex: potential role in sensitization to psychostimulants. Brain Res Brain Res Rev 41: 203–228.
Strassnig M, Brar JS, Ganguli R (2003a). Body mass index and quality of life in community-dwelling patients with schizophrenia. Schizophr Res 62: 73–76.
Strassnig M, Brar JS, Ganguli R (2003b). Nutritional assessment of patients with schizophrenia: a preliminary study. Schizophr Bull 29: 393–397.
Strassnig M, Singh Brar J, Ganguli R (2005). Dietary fatty acid and antioxidant intake in community-dwelling patients suffering from schizophrenia. Schizophr Res 15: 343–351.
Sturm R (2002). The effects of obesity, smoking, and drinking on medical problems and costs. Obesity outranks both smoking and drinking in its deleterious effects on health and health costs. Health Affair (Millwood) 21: 245–253.
Sundram S, Copolov D, Dean B (2005). Clozapine decreases [(3)H] CP 55940 binding to the cannabinoid (1) receptor in the rat nucleus accumbens. Naunyn Schmiedebergs Arch Pharmacol 37: 428–433.
Suzuki M, Zhou SY, Takashashi T, Hagino H, Kawasaki Y, Niu L (2005). Differential contributions of prefrontal and temporolimbic pathology to mechanisms of psychosis. Brain 128: 2109–2122.
Swanson LW (2000). Cerebral hemisphere regulation of motivated behavior. Brain Res 886: 113–164.
Sweet RA, Bergen SE, Sun Z, Sampson AR, Pierri JN, Lewis DA (2004). Pyramidal cell size reduction in schizophrenia: evidence for involvement of auditory feedforward circuits. Biol Psychiatry 55: 1128–1137.
Takahashi H, Koeda M, Oda K, Matsuda T, Matsushima E, Matsuura M et al (2004). An fMRI study of differential neural response to affective pictures in schizophrenia. Neuroimage 22: 1247–1254.
Takahashi JS (1999). Narcolepsy genes wake up the sleep field. Science 285: 2076–2077.
Tamminga A, Buschbaum MS (2004). Frontal cortex function. Am J Psychiatry 161: 2178.
Tanda G, Di Chiara G (1998). A dopamine-mu1 opioid link in the rat ventral tegmentum shared by palatable food (Fonzies) and non-psychostimulant drugs of abuse. Eur J Neurosci 10: 1179–1187.
Taylor SF, Liberzon I (1999). Paying attention to emotion in schizophrenia. Br J Psychiatry 174: 6–8.
Taylor SF, Liberzon I, Decker LR, Koeppe RA (2002). A functional anatomic study of emotion in schizophrenia. Schizophr Res 58: 159–172.
Taylor SF, Phan KL, Britton JC, Liberzon I (2005). Neural response to emotional salience in schizophrenia. Neuropsychopharmacology 30: 984–995.
Templeman LA, Reynolds GP, Arranz B, San L (2005). Polymorphisms of the 5-HT2C receptor and leptin genes are associated with antipsychotic drug-induced weight gain in Caucasian subjects with a first-episode psychosis. Pharmacogenet Genomics 15: 195–200.
Terenius L, Wahlström A, Lindström A, Widerlov E (1976). Increased CSF levels of endorphins in chronic psychosis. Neurosci Lett 3: 157–162.
Thakore JH (2004). Metabolic disturbance in first-episode schizophrenia. Br J Psychiatry Suppl 47: S76–S79.
Thakore JH (2005). Metabolic syndrome and schizophrenia. Br J Psychiatry 186: 455–456.
Thakore JH, Mann JN, Vlahos I, Martin A, Reznek R (2002). Increased visceral fat distribution in drug-naive and drug-free patients with schizophrenia. Int J Obes Relat Metab Disord 26: 137–141.
Theisen FM, Gebhardt S, Bromel T, Otto B, Heldwein W, Heinzel-Gutenbrunner M et al (2005). A prospective study of serum ghrelin levels in patients treated with clozapine. J Neural Transm 112: 1411–1416.
Thierry AM, Tassin JP, Blanc G, Glowinski J (1976). Selective activation of mesocortical DA system by stress. Nature 263: 242–244.
Thompson DA, Welle SL, Lilavivat U, Penicaud L, Campbell RG (1982). Opiate receptor blockade in man reduces 2-deoxy-D-glucose-induced food intake but not hunger, thirst, and hypothermia. Life Sci 31: 847–852.
Thornton-Jones Z, Neill JC, Reynolds GP (2002). The atypical antipsychotic olanzapine enhances ingestive behaviour in the rat: a preliminary study. J Psychopharmacol 16: 35–37.
Tobler PN, Dickinson A, Schultz W (2003). Coding of predicted reward omission by dopamine neurons in a conditioned inhibition paradigm. J Neurosci 23: 10402–10410.
Toda S, McGinty JF, Kalivas PW (2002). Repeated cocaine administration alters the expression of genes in corticolimbic circuitry after a 3-week withdrawal: A DNA macroarray Study. J Neurochem 82: 1290–1299.
Togo T, Hasegawa K, Miura S, Hosojima H, Kojima K, Shoji M et al (2004). Serum ghrelin concentrations in patients receiving olanzapine or risperidone. Psychopharmacology (Berlin) 172: 230–232.
Tonra JR, Ono M, Liu X, Garcia K, Jackson C, Yancopoulos GD et al (1999). Brain-derived neurotrophic factor improves blood glucose control alnd alleviates fasting hyperglycemia in C57BLKS-Lepr(db) mice. Diabetes 48: 588–594.
Torrey EF (2002). Studies of individuals with schizophrenia never treated with antipsychotic medications: a review. Schizophr Res 58: 101–115.
Trenchard E, Silverstone T (1983). Naloxone reduces the food intake of normal human volunteers. Appetite 4: 43–50.
Tsai SJ, Hong CJ, Yu YW, Lin CH (2002). −759C/T genetic variation of 5HT(2C) receptor and clozapine-induced weight gain. Lancet 360: 1790.
Tuomisto T, Hetherington MM, Morris MF, Tuomisto MT, Turjanmaa V, Lappalainen R (1999). Psychological and physiological characteristics of sweet food ‘addiction’. Int J Eat Disord 25: 169–175.
Turetsky BI, Moberg PJ, Owzar K, Johnson SC, Doty RL, Gur RE (2003). Physiologic impairment of olfactory stimulus processing in schizophrenia. Biol Psychiatry 53: 403–411.
Ulijaszek SJ (2002). Human eating behaviour in an evolutionary ecological context. Proc Nutr Soc 61: 517–526.
Van Tol HH, Bunzow JR, Guan HC, Sunahara RK, Seeman P, Niznik HB et al (1991). Cloning of the gene for a human dopamine D4 receptor with high affinity for the antipsychotic clozapine. Nature 350: 610–614.
Ventriglia M, Bocchio Chiavetto L, Bonvicini C, Tura GB, Bignotti S, Racagni G et al (2002). Allelic variation in the human prodynorphin gene promoter and schizophrenia. Neuropsychobiology 46: 17–21.
Virtanen KA, Iozzo P, Hallsten K, Huupponen R, Parkkola R, Janatuinen T et al (2005). Increased fat mass compensates for insulin resistance in abdominal obesity and type 2 diabetes: a positron-emitting tomography study. Diabetes 54: 2720–2726.
Volavka J, Davis LG, Ehrlich YH (1979). Endorphins, dopamine, and schizophrenia. Schizophr Bull 5: 227–239.
Volavka J, Mallya A, Baig S, Perez-Cruet J (1977). Naloxone in chronic schizophrenia. Science 196: 1227–1228.
Volkow ND, Fowler JS (2000). Addiction, a disease of compulsion and drive: involvement of the orbitofrontal cortex. Cereb Cortex 10: 318–325.
Volkow ND, Fowler JS, Wang GJ (2002a). Role of dopamine in drug reinforcement and addiction in humans: results from imaging studies. Behav Pharmacol 13: 355–366.
Volkow ND, Fowler JS, Wang GJ (2004a). The addicted human brain viewed in the light of imaging studies: brain circuits and treatment strategies. Neuropharmacology 47 (Suppl 1): 3–13.
Volkow ND, Fowler JS, Wang GJ, Goldstein RZ (2002b). Role of dopamine, the frontal cortex and memory circuits in drug addiction: insight from imaging studies. Neurobiol Learn Mem 78: 610–624.
Volkow ND, Fowler JS, Wang GJ, Swanson JM (2004b). Dopamine in drug abuse and addiction: results from imaging studies and treatment implications. Mol Psychiatry 9: 557–569.
Volkow ND, Wang GJ, Fowler JS, Logan J, Jayne M, Franceschi D et al (2002c). ‘Nonhedonic’ food motivation in humans involves dopamine in the dorsal striatum and methylphenidate amplifies this effect. Synapse 44: 175–180.
Volkow ND, Chang L, Wang GJ, Fowler JS, Ding YS, Sedler M et al (2001a). Low level of brain dopamine D2 receptors in methamphetamine abusers: association with metabolism in the orbitofrontal cortex. Am J Psychiatry 158: 2015–2021.
Volkow ND, Fowler JS, Wolf AP, Hitzemann R, Dewey S, Bendriem B et al (2001b). Changes in brain glucose metabolism in cocaine dependence and withdrawal. Am J Psychiatry 148: 621–626.
Volkow ND, Hitzemann R, Wang GJ, Fowler JS, Wolf AP, Dewey SL (1992). Long-term frontal brain metabolic changes in cocaine abusers. Synapse 11: 184–190.
Volkow ND, Wang GJ, Fowler JS, Franceschi D, Thanos PK, Wong C et al (2000). Cocaine abusers show a blunted response to alcohol intoxication in limbic brain regions. Life Sci 66: 161–167.
Volkow ND, Wang GJ, Fowler JS, Logan J, Gatley SJ, Hitzemann R et al (1997). Decreased striatal dopaminergic responsiveness in detoxified cocaine-dependent subjects. Nature 386: 830–833.
Volkow ND, Wang GJ, Ma Y, Fowler JS, Wong C, Ding YS et al (2005). Activation of orbital and medial prefrontal cortex by methylphenidate in cocaine-addicted subjects but not in controls; relevance to addiction. J Neurosci 25: 3932–3939.
Volkow ND, Wang GJ, Maynard L, Jayne M, Fowler JS, Zhu W et al (2003). Brain dopamine is associated with eating behaviors in humans. Int J Eat Disord 33: 136–142.
Volkow ND, Wise RA (2005). How can drug addiction help us understand obesity? Nat Neurosci 8: 555–560.
Voruganti L, Awad AG (2004). Neuroleptic dysphoria: towards a new synthesis. Psychopharmacology (Berlin) 171: 121–132.
Voruganti L, Slomka P, Zabel P, Costa G, So A, Mattar A et al (2001). Subjective effects of AMPT-induced dopamine depletion in schizophrenia: correlation between dysphoric responses and striatal D(2) binding ratios on SPECT imaging. Neuropsychopharmacology 25: 642–650.
Wang GJ, Volkow ND, Fowler JS (2002). The role of dopamine in motivation for food in humans: implications for obesity. Expert Opin Ther Targets 6: 601–609.
Wang VJ, Volkow ND, Fowler JS, Logan J, Abumrad NN, Hitzemann RJ et al (1997). Dopamine D2 receptor availability in opiate-dependent subjects before and after naloxone-precipitated withdrawal. Neuropsychopharmacology 16: 174–182.
Wang GJ, Volkow ND, Logan J, Pappas NR, Wong CT, Zhu W et al (2001). Brain dopamine and obesity. Lancet 357: 354–357.
Wang GJ, Volkow ND, Thanos PK, Fowler JS (2004). Similarity between obesity and drug addiction as assessed by neurofunctional imaging: a concept review. J Addict Dis 23: 39–53.
Wang VJ, Volkow ND, Cervany P, Hitzemann RJ, Pappas NR, Wong CT et al (1999). Regional brain metabolic activation during craving elicited by recall of previous drug experiences. Life Sci 64: 775–784.
Wang Y, Rimm EB, Stampfer MJ, Willett WC, Hu FB (2005). Comparison of abdominal adiposity and overall obesity in predicting risk of type 2 diabetes among men. Am J Clin Nutr 81: 555–563.
Wansink B (2004). Environmental factors that increase the food intake and consumption volume of unknowing consumers. Annu Rev Nutr 24: 455–479.
Watson SJ, Berger PA, Akil H, Mills MJ, Barchas JD (1978). Effects of naloxone on schizophrenia: reduction in hallucinations in a subpopulation of subjects. Science 201: 73–76.
Weickert CS, Hyde TM, Lipska BK, Herman MM, Weinberger DR, Kleinman JE (2003). Reduced brain-derived neurotrophic factor in prefrontal cortex of patients with schizophrenia. Mol Psychiatry 8: 592–610.
Weickert CS, Ligons DL, Romanczyk T, Ungaro G, Hyde TM, Herman MM et al (2005). Reductions in neurotrophin receptor mRNAs in the prefrontal cortex of patients with schizophrenia. Mol Psychiatry 10: 637–650.
Weinberger DR (1987). Implications of normal brain development for the pathogenesis of schizophrenia. Arch Gen Psychiatry 44: 660–669.
Weinberger DR, Berman KF (1996). Prefrontal function in schizophrenia: confounds and controversies. Philos Trans R Soc Lond Ser B 351: 1495–1503.
Weiser M, Noy S (2005). Interpreting the association between cannabis use and increased risk for schizophrenia. Dialog Clin Neurosci 7: 81–85.
Weizman T, Pick CG, Backer MM, Rigai T, Bloch M, Schreiber S (2003). The antinociceptive effect of amisulpride in mice is mediated through opioid mechanisms. Eur J Pharmacol 478: 155–159.
Welch EB, Thompson DF (1994). Opiate antagonists for the treatment of schizophrenia. J Clin Pharm Ther 19: 279–283.
West AR, Floresco SB, Charara A, Rosenkranz JA, Grace AA (2003). Electrophysiological interactions between striatal glutamatergic and dopaminergic systems. Ann NY Acad Sci 1003: 53–74.
Wetterling T (2001). Bodyweight gain with atypical antipsychotics. A comparative review. Drug Saf 24: 59–73.
Wiegant VM, Ronken E, Kovacs G, De Wied D (1992). Endorphins and schizophrenia. Prog Brain Res 93: 433–453.
Will MJ, Franzblau EB, Kelley AE (2003). Nucleus accumbens mu-opioids regulate intake of a high-fat diet via activation of a distributed brain network. J Neurosci 23: 2882–2888.
Will MJ, Franzblau EB, Kelley AE (2004). The amygdala is critical for opioid-mediated binge eating of fat. Neuroreport 15: 1857–1860.
Willenbring ML, Morley JE, Krahn DD, Carlson GA, Levine AS, Shafer RB (1989). Psychoneuroendocrine effects of methadone maintenance. Psychoneuroendocrinology 14: 371–391.
Williams LM, Das P, Harris AW, Liddell BB, Brammer MJ, Olivieri G et al (2004). Dysregulation of arousal and amygdala–prefrontal systems in paranoid schizophrenia. Am J Psychiatry 161: 480–489.
Willner P, Towell A, Sampson D, Sophokleous S, Muscat R (1987). Reduction of sucrose preference by chronic unpredictable mild stress, and its restoration by a tricyclic antidepressant. Psychopharmacology (Berlin) 93: 358–364.
Wirshing DA (2004). Schizophrenia and obesity: impact of antipsychotic medications. J Clin Psychiatry 65 (Suppl 18): 13–26.
Wirshing DA, Wirshing WC, Kysar L, Berisford MA, Goldstein D, Pashdag J et al (1999). Novel antipsychotics: comparison of weight gain liabilities. J Clin Psychiatry 60: 358–363.
Wise RA (1996). Addictive drugs and brain stimulation reward. Annu Rev Neurosci 19: 319–340.
Wise RA (2004). Dopamine, learning and motivation. Nat Rev Neurosci 5: 483–494.
Wolkin A, Sanfilipo M, Wolf AP, Angrist B, Brodie JD, Rotrosen J (1992). Negative symptoms and hypofrontality in chronic schizophrenia. Arch Gen Psychiatry 49: 959–965.
Wonodi I, Adami H, Sherr J, Avila M, Hong LE, Thaker GK (2004). Naltrexone treatment of tardive dyskinesia in patients with schizophrenia. J Clin Psychopharmacol 24: 441–445.
Woods SC, Seeley RJ, Baskin DG, Schwartz MW (2003). Insulin and the blood–brain barrier. Curr Pharm Des 9: 795–800.
World Health Organization (1979). Schizophrenia: An International Follow-up Study. John Wiley: New York.
Wynne K, Stanley S, McGowan B, Bloom S (2005). Appetite control. J Endocrinol 184: 291–318.
Xu B, Goulding EH, Zang K, Cepoi D, Cone RD, Jones KR et al (2003). Brain-derived neurotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat Neurosci 6: 736–742.
Yeomans MR, Gray RW (1996). Selective effects of naltrexone on food pleasantness and intake. Physiol Behav 60: 439–446.
Yeomans MR, Gray RW (1997). Effects of naltrexone on food intake and changes in subjective appetite during eating: evidence for opioid involvement in the appetizer effect. Physiol Behav 62: 15–21.
Yeomans MR, Gray RW (2002). Opioid peptides and the control of human ingestive behaviour. Neurosci Biobehav Rev 26: 713–728.
Yeomans MR, Gray RW, Mitchell CJ, True S (1997). Independent effects of palatability and within-meal pauses on intake and appetite ratings in human volunteers. Appetite 29: 61–76.
Yeomans MR, Tovey HM, Tinley EM, Haynes CJ (2004). Effects of manipulated palatability on appetite depend on restraint and disinhibition scores from the Three-Factor Eating Questionnaire. Int J Obes Relat Metab Disord 28: 144–151.
Yeomans MR, Wright P (1991). Lower pleasantness of palatable foods in nalmefene-treated human volunteers. Appetite 16: 249–259.
Yeomans MR, Wright P, Macleod HA, Critchley JA (1990). Effects of nalmefene on feeding in humans. Dissociation of hunger and palatability. Psychopharmacology (Berlin) 100: 426–432.
Yoshida Y, Koide S, Hirose N, Takada K, Tomiyama K, Koshikawa N et al (1999). Fentanyl increases dopamine release in rat nucleus accumbens: involvement of mesolimbic mu- and delta-2-opioid receptors. Neuroscience 92: 1357–1365.
You ZB, Herrera-Marschitz M, Nylander I, Goiny M, O’Connor WT, Ungerstedt U et al (1994). The striatonigral dynorphin pathway of the rat studied with in vivo microdialysis—II. Effects of dopamine D1 and D2 receptor agonists. Neuroscience 63: 427–434.
Zador D, Lyons Wall PM, Webster I (1996). High sugar intake in a group of women on methadone maintenance in south western Sydney, Australia. Addiction 91: 1053–1061.
Zeng XP, Le F, Richelson E (1997). Muscarinic m4 receptor activation by some atypical antipsychotic drugs. Eur J Pharmacol 321: 349–354.
Zhang ZJ, Yao ZJ, Liu W, Fang Q, Reynolds GP (2004). Effects of antipsychotics on fat deposition and changes in leptin and insulin levels. Magnetic resonance imaging study of previously untreated people with schizophrenia. Br J Psychiatry 184: 58–62.
Zubieta JK, Gorelick DA, Stauffer R, Ravert HT, Dannals RF, Frost JJ (1996). Increased mu opioid receptor binding detected by PET in cocaine-dependent men is associated with cocaine craving. Nat Med 2: 1225–1229.
Zukin RS, Sugarman JR, Fitz-Syage ML, Gardner EL, Zukin SR, Gintzler AR (1982). Naltrexone-induced opiate receptor supersensitivity. Brain Res 245: 285–292.
Acknowledgements
This work was supported by Grants DA# 017959 (IE) and DA# 00343 (SEL) from the National Institute on Drug Abuse. We thank Ronna Shostak for the help with the reference list.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Elman, I., Borsook, D. & Lukas, S. Food Intake and Reward Mechanisms in Patients with Schizophrenia: Implications for Metabolic Disturbances and Treatment with Second-Generation Antipsychotic Agents. Neuropsychopharmacol 31, 2091–2120 (2006). https://doi.org/10.1038/sj.npp.1301051
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.npp.1301051
Keywords
This article is cited by
-
Cebranopadol for the Treatment of Chronic Pain
Current Pain and Headache Reports (2023)
-
Prevalence of obesity and clinical and metabolic correlates in first-episode schizophrenia relative to healthy controls
Psychopharmacology (2021)
-
Metabolic and Addiction Indices in Patients on Opioid Agonist Medication-Assisted Treatment: A Comparison of Buprenorphine and Methadone
Scientific Reports (2020)
-
STructured lifestyle education for people WIth SchizophrEnia (STEPWISE): mixed methods process evaluation of a group-based lifestyle education programme to support weight loss in people with schizophrenia
BMC Psychiatry (2019)
-
Liraglutide and the management of overweight and obesity in people with schizophrenia, schizoaffective disorder and first-episode psychosis: protocol for a pilot trial
Trials (2019)