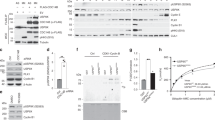
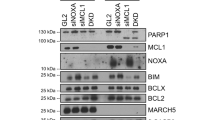
Abstract
A putative ubiquitin conjugating enzyme known as UBE2Q2 was previously identified in a microarray screen for mitotic regulatory proteins. UBE2Q2 is very similar to another human protein, UBE2Q1 and orthologs from other higher eukaryotic species. In these studies, we demonstrate that UBE2Q2 can covalently bind ubiquitin on the active site cysteine in vitro and show that inhibition of this protein in vivo causes an early mitotic arrest and increased cytotoxicity when cells are treated with microtubule inhibiting agents (MIAs). Changes in cell cycle progression and viability are not observed in the absence of MIA treatment, indicating that UBE2Q2 is involved in the response to MIAs rather than performing a more general function in mitosis. Inhibition of the UBE2Q2 protein causes cells to undergo a prolonged prophase arrest suggesting that UBE2Q2 normally functions to antagonize an early mitotic checkpoint. Furthermore, UBE2Q2 inhibition sensitizes cells to the cytotoxic effects of MIAs through caspase-mediated apoptosis that is correlated with PARP-1 cleavage. These data provide insights into the cellular response to MIAs and demonstrate that inhibition of UBE2Q2 protein function may be useful in the treatment of malignancies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Aristarkhov A, Eytan E, Moghe A, Admon A, Hershko A, Ruderman JV . (1996). E2-C, a cyclin-selective ubiquitin carrier protein required for the destruction of mitotic cyclins. Proc Natl Acad Sci USA 93: 4294–4299.
Baker DJ, Chen J, van Deursen JM . (2005). The mitotic checkpoint in cancer and aging: what have mice taught us? Curr Opin Cell Biol 17: 583–589.
Banerjee A, Deshaies RJ, Chau V . (1995). Characterization of a dominant negative mutant of the cell cycle ubiquitin-conjugating enzyme Cdc34. J Biol Chem 270: 26209–26215.
Bhalla KN . (2003). Microtubule-targeted anticancer agents and apoptosis. Oncogene 22: 9075–9086.
Chen C, Okayama H . (1987). High-efficiency transformation of mammalian cells by plasmid DNA. Mol Cell Biol 7: 2745–2752.
Corn PG, Summers MK, Fogt F, Virmani AK, Gazdar AF, Halazonetis TD et al. (2003). Frequent hypermethylation of the 5′ CpG island of the mitotic stress checkpoint gene Chfr in colorectal and non-small cell lung cancer. Carcinogenesis 24: 47–51.
Crawford DF, Piwnica-Worms H . (2001). The G(2) DNA damage checkpoint delays expression of genes encoding mitotic regulators. J Biol Chem 276: 37166–37177.
Gorbsky GJ . (2001). The mitotic spindle checkpoint. Curr Biol 11: R1001–R1004.
Gu H, Roizman B . (2003). The degradation of promyelocytic leukemia and Sp100 proteins by herpes simplex virus 1 is mediated by the ubiquitin-conjugating enzyme UbcH5a. Proc Natl Acad Sci USA 100: 8963–8968.
Huang Y, Fang Y, Wu J, Dziadyk JM, Zhu X, Sui M et al. (2004). Regulation of Vinca alkaloid-induced apoptosis by NF-kappaB/IkappaB pathway in human tumor cells. Mol Cancer Ther 3: 271–277.
Koh DW, Dawson TM, Dawson VL . (2005). Mediation of cell death by poly(ADP-ribose) polymerase-1. Pharmacol Res 52: 5–14.
Kung AL, Sherwood SW, Schimke RT . (1990). Cell line-specific differences in the control of cell cycle progression in the absence of mitosis. Proc Natl Acad Sci USA 87: 9553–9557.
Lew DJ, Burke DJ . (2003). The spindle assembly and spindle position checkpoints. Annu Rev Genet 37: 251–282.
Li L, Aggarwal BB, Shishodia S, Abbruzzese J, Kurzrock R . (2004). Nuclear factor-kappaB and IkappaB kinase are constitutively active in human pancreatic cells, and their down-regulation by curcumin (diferuloylmethane) is associated with the suppression of proliferation and the induction of apoptosis. Cancer 101: 2351–2362.
Li YP, Lecker SH, Chen Y, Waddell ID, Goldberg AL, Reid MB . (2003). TNF-alpha increases ubiquitin-conjugating activity in skeletal muscle by up-regulating UbcH2/E220k. FASEB J 17: 1048–1057.
Liu Q, Shang F, Whitcomb E, Guo W, Li W, Taylor A . (2006). Ubiquitin-conjugating enzyme 3 delays human lens epithelial cells in metaphase. Invest Ophthalmol Vis Sci 47: 1302–1309.
Matsusaka T, Pines J . (2004). Chfr acts with the p38 stress kinases to block entry to mitosis in mammalian cells. J Cell Biol 166: 507–516.
Melner MH, Ducharme NA, Brash AR, Winfrey VP, Olson GE . (2004). Differential expression of genes in the endometrium at implantation: upregulation of a novel member of the E2 class of ubiquitin-conjugating enzymes. Biol Reprod 70: 406–414.
Melner MH, Haas AL, Klein JM, Brash AR, Boeglin WE, Nagdas SK et al. (2006). Demonstration of ubiquitin thiolester formation of UBE2Q2 (UBCi), a novel ubiquitin conjugating enzyme with implantation site-specific expression. Biol Reprod 75: 395–406.
Mizuno K, Osada H, Konishi H, Tatematsu Y, Yatabe Y, Mitsudomi T et al. (2002). Aberrant hypermethylation of the CHFR prophase checkpoint gene in human lung cancers. Oncogene 21: 2328–2333.
Mollinedo F, Gajate C . (2003). Microtubules, microtubule-interfering agents and apoptosis. Apoptosis 8: 413–450.
Musacchio A, Hardwick KG . (2002). The spindle checkpoint: structural insights into dynamic signalling. Nat Rev Mol Cell Biol 3: 731–741.
Pallardy M, Biola A, Lebrec H, Breard J . (1999). Assessment of apoptosis in xenobiotic-induced immunotoxicity. Methods 19: 36–47.
Pati D, Meistrich ML, Plon SE . (1999). Human Cdc34 and Rad6B ubiquitin-conjugating enzymes target repressors of cyclic AMP-induced transcription for proteolysis. Mol Cell Biol 19: 5001–5013.
Pinkerton CR, McDermott B, Philip T, Biron P, Ardiet C, Vandenberg H et al. (1988). Continuous vincristine infusion as part of a high dose chemoradiotherapy regimen: drug kinetics and toxicity. Cancer Chemother Pharmacol 22: 271–274.
Pray TR, Parlati F, Huang J, Wong BR, Payan DG, Bennett MK et al. (2002). Cell cycle regulatory E3 ubiquitin ligases as anticancer targets. Drug Resist Updat 5: 249–258.
Rieder CL, Cole R . (2000). Microtubule disassembly delays the G2-M transition in vertebrates. Curr Biol 10: 1067–1070.
Rieder CL, Palazzo RE . (1992). Colcemid and the mitotic cycle. J Cell Sci 102 (Part 3): 387–392.
Rieder CL, Schultz A, Cole R, Sluder G . (1994). Anaphase onset in vertebrate somatic cells is controlled by a checkpoint that monitors sister kinetochore attachment to the spindle. J Cell Biol 127: 1301–1310.
Schulze E, Altmann ME, Adham IM, Schulze B, Frode S, Engel W . (2003). The maintenance of neuromuscular function requires UBC-25 in Caenorhabditis elegans. Biochem Biophys Res Commun 305: 691–699.
Scolnick DM, Halazonetis TD . (2000). Chfr defines a mitotic stress checkpoint that delays entry into metaphase. Nature 406: 430–435.
Seghatoleslam A, Zambrano A, Millon R, Ganguli G, Argentini M, Cromer A et al. (2006). Analysis of a novel human gene, LOC92912, over-expressed in hypopharyngeal tumours. Biochem Biophys Res Commun 339: 422–429.
Skerra A, Schmidt TG . (1999). Applications of a peptide ligand for streptavidin: the strep-tag. Biomol Eng 16: 79–86.
Tashiro K, Pando MP, Kanegae Y, Wamsley PM, Inoue S, Verma IM . (1997). Direct involvement of the ubiquitin-conjugating enzyme Ubc9/Hus5 in the degradation of IkappaBalpha. Proc Natl Acad Sci USA 94: 7862–7867.
Townsley FM, Aristarkhov A, Beck S, Hershko A, Ruderman JV . (1997). Dominant-negative cyclin-selective ubiquitin carrier protein E2-C/UbcH10 blocks cells in metaphase. Proc Natl Acad Sci USA 94: 2362–2367.
Toyota M, Sasaki Y, Satoh A, Ogi K, Kikuchi T, Suzuki H et al. (2003). Epigenetic inactivation of CHFR in human tumors. Proc Natl Acad Sci USA 100: 7818–7823.
Vodermaier HC . (2004). APC/C and SCF: controlling each other and the cell cycle. Curr Biol 14: R787–R796.
Weissman AM . (2001). Themes and variations on ubiquitylation. Nat Rev Mol Cell Biol 2: 169–178.
Xu B, Kim S, Kastan MB . (2001). Involvement of Brca1 in S-phase and G(2)-phase checkpoints after ionizing irradiation. Mol Cell Biol 21: 3445–3450.
Zhang YW, Otterness DM, Chiang GG, Xie W, Liu YC, Mercurio F et al. (2005). Genotoxic stress targets human Chk1 for degradation by the ubiquitin-proteasome pathway. Mol Cell 19: 607–618.
Acknowledgements
We appreciate the assistance of Drs Xinbin Chen and Michael Ruppert for critical review of this manuscript. We appreciate the use of the fluorescence microscope as well as assistance provided by Drs Louise Chow and Thomas Broker and members of their laboratory. This work was supported by NIH grant 5K08CA86941-5 from the NCI as well as a grant from The Research Institute at Children's Hospital, Birmingham, Alabama, USA.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on the Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Banerjee, S., Brooks, W. & Crawford, D. Inactivation of the ubiquitin conjugating enzyme UBE2Q2 causes a prophase arrest and enhanced apoptosis in response to microtubule inhibiting agents. Oncogene 26, 6509–6517 (2007). https://doi.org/10.1038/sj.onc.1210471
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210471
Keywords
This article is cited by
-
Upregulated expression of ubiquitin-conjugating enzyme E2Q1 (UBE2Q1) is associated with enhanced cell proliferation and poor prognosis in human hapatocellular carcinoma
Journal of Molecular Histology (2015)
-
Small molecule antagonist of the bone morphogenetic protein type I receptors suppresses growth and expression of Id1 and Id3 in lung cancer cells expressing Oct4 or nestin
Molecular Cancer (2013)