Abstract
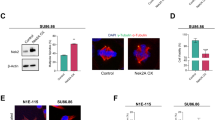
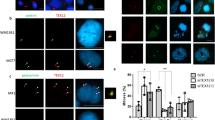
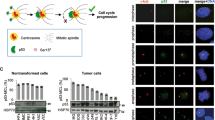
The centrosome, an organelle that functions as the major microtubule-organizing center, plays an essential role in the formation of the mitotic spindle and guiding accurate chromosome segregation. Centrosome aberrations are frequently associated with various forms of human cancers and it is thought that defects in this organelle contribute to genomic instability and malignant transformation. We recently identified and characterized a centrosome-localized protein complex that is comprised of Su48 and Nde1. Disruption of the normal function of these proteins leads to abnormal cell division. To extend our understanding of how this protein complex operates, we sought to identify Nde1-interacting molecules by the yeast two-hybrid screening method. Here, we demonstrate that both Nde1 and Su48 can associate with p78/MCRS1, a protein implicated in cancer development. We found that, whereas the majority of p78 localizes to the nucleus as reported in earlier studies, a fraction of the p78 protein can be detected in the centrosome. Moreover, we determined that a region containing the forkhead-associated domain of p78 is involved in association with Nde1 and Su48, as well as in centrosomal localization. We also provide evidence that the association between p78 and Nde1 is regulated by phosphorylation on Nde1. Furthermore, abrogation of the endogenous p78 function by small interfering RNA knockdown causes cell death and a modest delay in mitosis. These results indicate that a subset of the p78 proteins comprises a component of the centrosome and that p78 is essential for cell viability.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bader AG, Schneider ML, Bister K, Hartl M . (2001). Oncogene 20: 7524–7535.
Bruni R, Roizman B . (1998). J Virol 72: 8525–8531.
Doxsey S . (2001). Nat Rev Mol Cell Biol 2: 688–698.
Durocher D, Jackson SP . (2002). FEBS Lett 513: 58–66.
Efimov VP, Morris NR . (2000). J Cell Biol 150: 681–688.
Engel E, McGee BJ, Harris H . (1969a). J Cell Sci 5: 93–119.
Engel E, McGee BJ, Harris H . (1969b). Nature 223: 152–155.
Faulkner NE, Dujardin DL, Tai CY, Vaughan KT, O'Connell CB, Wang Y, Vallee RB . (2000). Nat Cell Biol 2: 784–791.
Feng Y, Olson EC, Stukenberg PT, Flanagan LA, Kirschner MW, Walsh CA . (2000). Neuron 28: 665–679.
Feng Y, Walsh CA . (2004). Neuron 44: 279–293.
Howell BJ, Hoffman DB, Fang G, Murray AW, Salmon ED . (2000). J Cell Biol 150: 1233–1250.
Howell BJ, Moree B, Farrar EM, Stewart S, Fang G, Salmon ED . (2004). Curr Biol 14: 953–964.
Lin DY, Shih HM . (2002). J Biol Chem 277: 25446–25456.
Lingle WL, Lutz WH, Ingle JN, Maihle NJ, Salisbury JL . (1998). Proc Natl Acad Sci USA 95: 2950–2955.
Moritz M, Braunfeld MB, Sedat JW, Alberts B, Agard DA . (1995). Nature 378: 638–640.
Moritz M, Zheng Y, Alberts BM, Oegema K . (1998). J Cell Biol 142: 775–786.
Niethammer M, Smith DS, Ayala R, Peng J, Ko J, Lee MS et al. (2000). Neuron 28: 697–711.
Nigg EA . (2002). Nat Rev Cancer 2: 815–825.
Okuda M, Horn HF, Tarapore P, Tokuyama Y, Smulian AG, Chan PK et al. (2000). Cell 103: 127–140.
Okumura K, Zhao M, Depinho RA, Furnari FB, Cavenee WK . (2005). Proc Natl Acad Sci USA 102: 2703–2706.
Paluh JL, Nogales E, Oakley BR, McDonald K, Pidoux AL, Cande WZ . (2000). Mol Biol Cell 11: 1225–1239.
Pihan GA, Purohit A, Wallace J, Knecht H, Woda B, Quesenberry P et al. (1998). Cancer Res 58: 3974–3985.
Pihan GA, Purohit A, Wallace J, Malhotra R, Liotta L, Doxsey SJ . (2001). Cancer Res 61: 2212–2219.
Prigozhina NL, Oakley CE, Lewis AM, Nayak T, Osmani SA, Oakley BR . (2004). Mol Biol Cell 15: 1374–1386.
Quintyne NJ, Reing JE, Hoffelder DR, Gollin SM, Saunders WS . (2005). Science 307: 127–129.
Salisbury JL, Whitehead CM, Lingle WL, Barrett SL . (1999). Biol Cell 91: 451–460.
Sasaki S, Shionoya A, Ishida M, Gambello MJ, Yingling J, Wynshaw-Boris A et al. (2000). Neuron 28: 681–696.
Sato N, Mizumoto K, Nakamura M, Nakamura K, Kusumoto M, Niiyama H et al. (1999). Clin Cancer Res 5: 963–970.
Schnackenberg BJ, Khodjakov A, Rieder CL, Palazzo RE . (1998). Proc Natl Acad Sci USA 95: 9295–9300.
Shono M, Sato N, Mizumoto K, Maehara N, Nakamura M, Nagai E et al. (2001). Lab Invest 81: 945–952.
Song H, Li Y, Chen G, Xing Z, Zhao J, Yokoyama KK et al. (2004). Biochem Biophys Res Commun 316: 1116–1123.
Starita LM, Machida Y, Sankaran S, Elias JE, Griffin K, Schlegel BP et al. (2004). Mol Cell Biol 24: 8457–8466.
Strome S, Powers J, Dunn M, Reese K, Malone CJ, White J et al. (2001). Mol Biol Cell 12: 1751–1764.
Sun Z, Hsiao J, Fay DS, Stern DF . (1998). Science 281: 272–274.
Tokuyama Y, Horn HF, Kawamura K, Tarapore P, Fukasawa K . (2001). J Biol Chem 276: 21529–21537.
Wang Q, Hirohashi Y, Furuuchi K, Zhao H, Liu Q, Zhang H et al. (2004). DNA Cell Biol 23: 475–489.
Wang Q, Zhang H, Kajino K, Greene MI . (1998). Oncogene 17: 1939–1948.
Yan X, Li F, Liang Y, Shen Y, Zhao X, Huang Q et al. (2003). Mol Cell Biol 23: 1239–1250.
Zheng Y, Wong ML, Alberts B, Mitchison T . (1995). Nature 378: 578–583.
Acknowledgements
This work was supported by funds from the Abramson Family Cancer Research Institute of the University of Pennsylvania and from the National Institute of Health.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc).
Supplementary information
Rights and permissions
About this article
Cite this article
Hirohashi, Y., Wang, Q., Liu, Q. et al. p78/MCRS1 forms a complex with centrosomal protein Nde1 and is essential for cell viability. Oncogene 25, 4937–4946 (2006). https://doi.org/10.1038/sj.onc.1209500
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209500
Keywords
This article is cited by
-
MCRS1 associates with cytoplasmic dynein and mediates pericentrosomal material recruitment
Scientific Reports (2016)
-
The candidate oncogene (MCRS1) promotes the growth of human lung cancer cells via the miR–155–Rb1 pathway
Journal of Experimental & Clinical Cancer Research (2015)
-
Increased expression of the 58-kD microspherule protein (MSP58) is correlated with poor prognosis in glioma patients
Medical Oncology (2013)
-
K-fibre minus ends are stabilized by a RanGTP-dependent mechanism essential for functional spindle assembly
Nature Cell Biology (2011)
-
A new cap for kinetochore fibre minus ends
Nature Cell Biology (2011)