Abstract
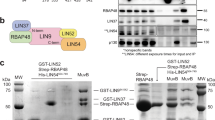
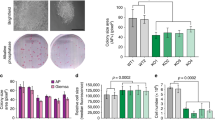
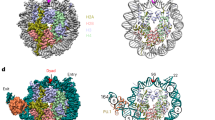
The activity of E2F transcription factors plays a crucial role in mammalian cell-cycle progression and is controlled by physical association with the pocket proteins (pRb and its related p107 and p130). The E2F1 promoter, which contains two overlapping E2F-binding sites, is activated at the G1/S transition in an E2F-dependent manner. Mutational experiments have shown that the distal E2F-binding site on the E2F1 promoter is required for transcriptional repression in the G0 phase, whereas the proximal E2F-binding site contributes to transcriptional activation at the G1/S boundary. Consistent with these results, chromatin immunoprecipitation assays have revealed that the E2F4/p130 repressor complex specifically binds to the distal E2F-binding site, whereas E2F1 and E2F3 activators preferentially bind to the proximal E2F-binding site. The assays also showed that the specific binding of E2F4/p130 complex to the distal site was dramatically impaired by a mutation introduced into the contiguous repression site (cell Cycle gene Homology Region; CHR). Taken together, these findings indicate that the two E2F-binding sites play distinct roles in the regulation of E2F1 transcription by interacting with different sets of E2F members and cooperating with the contiguous repressor element.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bennett JD, Farlie PG and Watson RJ . (1996). Oncogene, 13, 1073–1082.
Botz J, Zerfass-Thome K, Spitkovsky D, Delius H, Vogt B, Eilers M, Hatzigeorgiou A and Jansen-Durr P . (1996). Mol. Cell. Biol., 16, 3401–3409.
Cartwright P, Muller H, Wagener C, Holm K and Helin K . (1998). Oncogene, 17, 611–623.
Catchpole S, Tavner F, Le Cam L, Sardet C and Watson RJ . (2002). J. Biol. Chem., 277, 39015–39024.
Dalton S . (1992). EMBO J., 11, 1797–1804.
Di Fiore B, Guarguaglini G, Palena A, Kerkhoven RM, Bernards R and Lavia P . (1999). J. Biol. Chem., 274, 10339–10348.
Dou QP, Markell PJ and Pardee AB . (1992). Proc. Natl. Acad. Sci. USA, 89, 3256–3260.
Dyson N . (1998). Genes Dev., 12, 2245–2262.
Gaubatz S, Wood JG and Livingston DM . (1998). Proc. Natl. Acad. Sci. USA, 95, 9190–9195.
Harbour JW and Dean DC . (2000). Genes Dev., 14, 2393–2409.
Hateboer G, Wobst A, Petersen BO, Le Cam L, Vigo E, Sardet C and Helin K . (1998). Mol. Cell. Biol., 18, 6679–6697.
Hsiao KM, McMahon SL and Farnham PJ . (1994). Genes Dev., 8, 1526–1537.
Hurford RK, Cobrinik D, Lee MH and Dyson N . (1997). Genes Dev., 11, 1447–1463.
Iavarone A and Massague J . (1999). Mol. Cell. Biol., 19, 916–922.
Ikeda MA, Jakoi L and Nevins JR . (1996). Proc. Natl. Acad. Sci. USA, 93, 3215–3220.
Johnson DG, Ohtani K and Nevins JR . (1994). Genes Dev., 8, 1514–1525.
Karlseder J, Rotheneder H and Wintersberger E . (1996). Mol. Cell. Biol., 16, 1659–1667.
Lam EW and Watson RJ . (1993). EMBO J., 12, 2705–2713.
Leone G, DeGregori J, Van Z, Jakoi L, Ishida S, Williams RS and Nevins JR . (1998). Genes Dev., 12, 2120–2130.
Lin SY, Black AR, Kostic D, Pajovic S, Hoover CN and Azizkhan JC . (1996). Mol. Cell. Biol., 16, 1668–1675.
Lindeman GJ, Gaubatz S, Livingston DM and Ginsberg D . (1997). Proc. Natl. Acad. Sci. USA, 94, 5095–5100.
Liu N, Lucibello FC, Engeland K and Muller R . (1998). Oncogene, 16, 2957–2963.
Neuman E, Flemington EK, Sellers WR and Kaelin WG . (1994). Mol. Cell. Biol., 14, 6607–6615.
Ogawa H, Ishiguro K, Gaubatz S, Livingston DM and Nakatani Y . (2002). Science, 296, 1132–1136.
Ohtani K, DeGregori J and Nevins JR . (1995). Proc. Natl. Acad. Sci. USA, 92, 12146–12150.
Ohtani K, Tsujimoto A, Ikeda MA and Nakamura M . (1998). Oncogene, 17, 1777–1785.
Pearson BE, Nasheuer HP and Wang TS . (1991). Mol. Cell. Biol., 11, 2081–2095.
Rayman JB, Takahashi Y, Indjeian VB, Dannenberg JH, Catchpole S, Watson RJ, te Riele H and Dynlacht BD . (2002). Genes Dev., 16, 933–947.
Rotheneder H, Geymayer S and Haidweger E . (1999). J. Mol. Biol., 293, 1005–1015.
Schlisio S, Halperin T, Vidal M and Nevins JR . (2002). EMBO J., 21, 5775–5786.
Schulze A, Zerfass K, Spitkovsky D, Middendorp S, Berges J, Helin K, Jansen-Durr P and Henglein B . (1995). Proc. Natl. Acad. Sci. USA, 92, 11264–11268.
Shan B, Chang CY, Jones D and Lee WH . (1994). Mol. Cell. Biol., 14, 299–309.
Smith EJ, Leone G, DeGregori J, Jakoi L and Nevins JR . (1996). Mol. Cell. Biol., 16, 6965–6976.
Takahashi Y, Rayman JB and Dynlacht BD . (2000). Genes Dev., 14, 804–816.
Tommasi S and Pfeifer GP . (1995). Mol. Cell. Biol., 15, 6901–6913.
Trimarchi JM and Lees JA . (2002). Nat. Rev. Mol. Cell Biol., 3, 11–20.
Vairo G, Livingston DM and Ginsberg D . (1995). Genes Dev., 9, 869–881.
Vigo E, Muller H, Prosperini E, Hateboer G, Cartwright P, Moroni MC and Helin K . (1999). Mol. Cell. Biol., 19, 6379–6395.
Wells J, Boyd KE, Fry CJ, Bartley SM and Farnham PJ . (2000). Mol. Cell. Biol., 20, 5797–5807.
Wu L, Goodwin EC, Naeger LK, Vigo E, Galaktionov K, Helin K and DiMaio D . (2000). Mol. Cell. Biol., 20, 7059–7067.
Zwicker J, Lucibello FC, Wolfraim LA, Gross C, Truss M, Engeland K and Muller R . (1995). EMBO J., 14, 4514–4522.
Zwicker J and Muller R . (1997). Trends. Genet., 13, 3–6.
Acknowledgements
This work was supported in part by the Grant-in-Aid for General Scientific Research and Cancer Research from the Ministry of Education, Science, Sports, and Culture of Japan and Japan Society for the Promotion of Science and in part by ‘Ground Research for Space Utilization’ promoted by NASDA and Japan Space Forum.
We thank Dr K Ohtani for pBS-GAPDH and for critically reading the manuscript, members of SME for support and useful discussions, and Solachuddin JA. Ichwan for excellent technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Araki, K., Nakajima, Y., Eto, K. et al. Distinct recruitment of E2F family members to specific E2F-binding sites mediates activation and repression of the E2F1 promoter. Oncogene 22, 7632–7641 (2003). https://doi.org/10.1038/sj.onc.1206840
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206840
Keywords
This article is cited by
-
ALKBH4 promotes tumourigenesis with a poor prognosis in non-small-cell lung cancer
Scientific Reports (2021)
-
E2F1 and E2F7 differentially regulate KPNA2 to promote the development of gallbladder cancer
Oncogene (2019)
-
Cyclin D–CDK4 relieves cooperative repression of proliferation and cell cycle gene expression by DREAM and RB
Oncogene (2019)
-
TACC3 transcriptionally upregulates E2F1 to promote cell growth and confer sensitivity to cisplatin in bladder cancer
Cell Death & Disease (2018)
-
E2F1/TS Immunophenotype and Survival of Patients with Colorectal Cancer Treated with 5FU-Based Adjuvant Therapy
Pathology & Oncology Research (2016)