Abstract
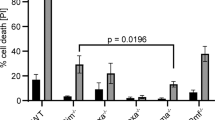
Bfl-1 is an antiapoptotic Bcl-2 family member and a mouse A1 homologue. The mouse A1 has been reported to have three isoforms, but little is known about human Bfl-1. By reverse–transcriptase polymerase chain reaction analysis, we have identified Bfl-1S (short form), an alternative splice variant of Bfl-1. The Bfl-1S primary sequence contains four conserved Bcl-2 homology (BH) domains and a positive-charged C-terminus containing KKRK amino acids. The expression of Bfl-1S mRNA was detected predominantly in normal lymph nodes and in B-lymphoid leukemia cells. Confocal microscopic analysis using green fluorescence protein fusion proteins demonstrated that Bfl-1S is localized in the nucleus by its C-terminus as an intrinsic nuclear localization sequence. Bfl-1S acts as an antiapoptotic agent in coexpression experiments with Bax, a proapoptotic molecule. The expression of Bfl-1S provided significant resistance against staurosporine (STS) treatments in Molt-4 human T-leukemia cells. Bfl-1S also significantly inhibited the cleavage of Bid, and of caspases 3 and 8 against STS treatment. These results indicate that Bfl-1S is a novel human Bcl-2 family member that possesses antiapoptotic function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Abbreviations
- RT–PCR:
-
reverse–transcriptase polymerase chain reaction
- PI:
-
propidium iodide
- GFP:
-
green fluorescence protein
- BH domain:
-
Bcl-2 homology domain
- NLS:
-
nuclear localization sequence
- STS:
-
staurosporine
- CHX:
-
cycloheximide
- 5-FU:
-
5-fluorouracil
- ΔΨm:
-
mitochondrial transmembrane potential
References
Boise LH, Gonzalez-Garcia M, Postema CE, Ding L, Lindsten T, Turka LA, Mao X, Nunez G and Thompson CB . (1993). Cell, 44, 597–608.
Cheng Q, Lee HH, Li Y, Parks TP and Cheng G . (2000). Oncogene, 19, 4936–4940.
Chao DT and Korsmeyer SJ . (1998). Annu. Rev. Immunol., 16, 395–419.
Cheung KJ and Li G . (2001). Exp. Cell. Res., 268, 1–6.
Choi SS, Park IC, Yun JW, Sung YC, Hong SI and Shin HS . (1995). Oncogene, 11, 1693–1698.
D'Sa-Eipper C and Chinnadurai G . (1998). Oncogene, 16, 3105–3114.
D'Sa-Eipper C, Subramanian T and Chinnadurai G . (1996). Cancer Res., 56, 3879–3882.
Duriez PJ, Wong F, Dorovini-Zis K, Shahidi R and Karsan A . (2000). J. Biol. Chem., 275, 18099–18107.
Ha S, Lee S, Chung M and Choi Y . (2002). Cancer Res., 62, 1275–1278.
Han J, Wallen HD, Nunez G and White E . (1998). Mol. Cell. Biol., 18, 6052–6062.
Han Z, Balla K, Pantazis P, Hendrickson EA and Wyche JH . (1999). Mol. Cell. Biol., 19, 1381–1389.
Hatakeyama S, Hamasaki A, Negishi I, Loh DY, Sendo F and Nakayama K . (1998). Int. Immunol., 10, 631–637.
Hockenbery D, Nunez G, Milliman C, Schreiber RD and Korsmeyer SJ . (1990). Nature, 348, 334–336.
Holmgreen SP, Huang DC, Adams JM and Cory S (1999) Cell Death and Differ., 6, 525–532.
Jung-Ha H, Kim D, Lee SB, Hong SI, Park SY, Huh J, Kim CW, Kim SS, Lee Y, Choi SS and Shin HS . (1998). Hum. Pathol., 29, 723–728.
Karsan A, Yee E and Harlan JM . (1996a). J. Biol. Chem., 271, 27201–27204.
Karsan A, Yee E, Kaushansky K and Harlan JM . (1996b). Blood, 87, 3089–3096.
Kasof GM, Goyal L and White E . (1999). Mol. Cell. Biol., 19, 4390–4404.
Kepler TB and Perelson AS . (1993). Immunol. Today, 14, 412–415.
Lee HH, Dadgostar H, Cheng Q, Shu J and Cheng G . (1999). Proc. Natl. Acad. Sci. USA, 96, 9136–9141.
Li H, Zhu H, Xu CJ and Yuan J . (1998). Cell, 94, 491–501.
Luo X, Budihardjo I, Zou H, Slaughter C and Wang X . (1998). Cell, 94, 481–490.
Perez D and White E . (1998). J. Cell. Biol., 141, 1255–1266.
Priault M, Camougrand N, Chaudhuri B, Manon S . (1999) FEBS Lett., 443, 225–228.
Rao L, Modha D and White E . (1997). Oncogene, 15, 1587–1597.
Reed JC . (1998). Oncogene, 17, 3225–3236.
Shim YH, Byun EK, Lee MJ, Huh J and Kim CW . (2000). Int. J. Hematol., 72, 484–490.
Somogyi RD, Wu A, Orlofsky A and Prystowsky MB . (2001). Cell Death Differ., 8, 785–793.
Tomayko MM and Cancro MP . (1998). J. Immunol., 160, 107–111.
Wang CY, Guttridge DC, Mayo MW and Baldwin Jr AS . (1999). Mol. Cell. Biol., 19, 5923–5929.
Wang L, Miura M, Bergeron L, Zhu H and Yuan J . (1994). Cell, 78, 739–750.
Werner AB, De Vries E, Tait SW, Bontjer I and Borst J . (2002). J. Biol. Chem., 277, 22781–22788.
Yin XM, Oltvai ZN and Korsmeyer SJ . (1994). Nature, 369, 321–323.
Zhang H, Cowan-Jacob SW, Simonen M, Greenhalf W, Heim J and Meyhack B . (2000). J. Biol. Chem., 275, 11092–11099.
Zong WX, Edelstein LC, Chen C, Bash J and Gelinas C . (1999). Genes Dev., 3, 382–387.
Acknowledgements
This work was supported by grants from the Korea Ministry of Health and Welfare (01-PJ1-PG3-20700-0033) and Seoul National University Hospital (03-2002-051). We thank Dr Masao Seto for a critical reading of this manuscript and helpful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ko, JK., Lee, MJ., Cho, SH. et al. Bfl-1S, a novel alternative splice variant of Bfl-1, localizes in the nucleus via its C-terminus and prevents cell death. Oncogene 22, 2457–2465 (2003). https://doi.org/10.1038/sj.onc.1206274
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206274
Keywords
This article is cited by
-
BCL-2 family isoforms in apoptosis and cancer
Cell Death & Disease (2019)
-
BCL2A1: the underdog in the BCL2 family
Cell Death & Differentiation (2012)
-
The C-terminal region of Bfl-1 sensitizes non-small cell lung cancer to gemcitabine-induced apoptosis by suppressing NF-κB activity and down-regulating Bfl-1
Molecular Cancer (2011)
-
Highly Conserved Caspase and Bcl-2 Homologues from the Sea Anemone Aiptasia pallida: Lower Metazoans as Models for the Study of Apoptosis Evolution
Journal of Molecular Evolution (2006)
-
Constitutive proteasome-mediated turnover of Bfl-1/A1 and its processing in response to TNF receptor activation in FL5.12 pro-B cells convert it into a prodeath factor
Cell Death & Differentiation (2005)