Abstract
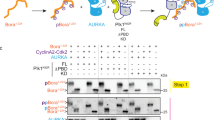
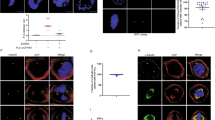
The cyclin-dependent kinase 1 (Cdc2)/cyclin B1 complex performs cardinal roles for eukaryotic mitotic progression. Phosphorylation of four serine residues within cyclin B1 promotes the rapid nuclear translocation of Cdc2/cyclin B1 at the G2/M transition. Still, the role of individual phosphorylation sites and their corresponding kinases remain to be elucidated. Polo-like kinase 1 (Plk1) shows a spatial and temporal distribution which makes it a candidate kinase for the phosphorylation of cyclin B1. We could demonstrate the interaction of both proteins in mammalian cells. Plk1 phosphorylated wild-type cyclin B1 expressed in bacteria and in mammalian cells. Ser-133 within the cytoplasmic retention signal (CRS) of cyclin B1, which regulates the nuclear entry of the heterodimeric complex during prophase, is a target of Plk1. In contrast, MAPK (Erk2) and MPF phosphorylate Ser-126 and Ser-128 within the CRS. Phosphorylation of CRS by MAPK (Erk2) prior to Plk1 treatment induced enhanced phosphorylation of cyclin B1 by Plk 1 suggesting a synergistic action of both enzymes towards cyclin B1. In addition, pretreatment of cyclin B1 by MAPK (Erk2) altered the phosphorylation pattern of Plk 1. Mutation of Ser-133 to Ala decreased the phosphorylation of cyclin B1 in vivo. An immunofluorescence study revealed that a mutation of Ser-133 reduced the nuclear import rate of cyclin B1. Still, multiple serine mutations are required to prevent nuclear translocation completely indicating that orchestrated phosphorylation within the CRS triggers rapid import of cyclin B1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Booher RN, Alfa CE, Hyams JS, Beach DH . 1989 Cell 58: 485–497
Borgne A, Ostvold AC, Flament S, Meijer L . 1999 J. Biol. Chem. 274: 11977–11986
Chen RH, Abate C, Blenis J . 1993 Proc. Natl. Acad. Sci. USA 90: 10952–10956
Clute P, Pines J . 1999 Nat. Cell Biol. 1: 82–87
Cogswell JP, Brown CE, Bisi JE, Neill SD . 2000 Cell Growth Differ. 11: 615–623
Gautier J, Maller JL . 1991 EMBO J. 10: 177–182
Gautier J, Minshull J, Lohka M, Glotzer M, Hunt T, Maller JL . 1990 Cell 60: 487–494
Gautier J, Solomon MJ, Booher RN, Bazan JF, Kirschner MW . 1991 Cell 67: 197–211
Glover DM, Hagan IM, Tavares AA . 1998 Genes Dev. 12: 3777–3787
Hagting A, Jackman M, Simpson K, Pines J . 1999 Curr. Biol. 9: 680–689
Hagting A, Karlsson C, Clute P, Jackman M, Pines J . 1998 EMBO J. 17: 4127–4138
Hock B, Bohme B, Karn T, Feller S, Rubsamen-Waigmann H, Strebhardt K . 1998a Oncogene 17: 255–260
Hock B, Bohme B, Karn T, Yamamoto T, Kaibuchi K, Holtrich U, Holland S, Pawson T, Rubsamen-Waigmann H, Strebhardt K . 1998b Proc. Natl. Acad. Sci. USA 95: 9779–9784
Holtrich U, Wolf G, Brauninger A, Karn T, Bohme B, Rubsamen-Waigmann H, Strebhardt K . 1994 Proc. Natl. Acad. Sci. USA 91: 1736–1740
Holtrich U, Wolf G, Yuan J, Bereiter-Hahn J, Karn T, Weiler M, Kauselmann G, Rehli M, Andreesen R, Kaufmann M, Kuhl D, Strebhardt K . 2000 Oncogene 19: 4832–4839
Horton RM, Cai ZL, Ho SN, Pease LR . 1990 Biotechniques 8: 528–535
Izumi T, Maller JL . 1991 Mol. Cell Biol. 11: 3860–3867
Kitada K, Johnson AL, Johnston LH, Sugino A . 1993 Mol. Cell Biol. 13: 4445–4457
Lake RJ, Jelinek WR . 1993 Mol. Cell Biol. 13: 7793–7801
Lane HA, Nigg EA . 1996 J. Cell Biol. 135: 1701–1713
Lee KS, Yuan YL, Kuriyama R, Erikson RL . 1995 Mol. Cell Biol. 15: 7143–7151
Li J, Meyer AN, Donoghue DJ . 1995 Mol. Biol. Cell 6: 1111–1124
Li J, Meyer AN, Donoghue DJ . 1997 Proc. Natl. Acad. Sci. USA 94: 502–507
Lin CY, Madsen ML, Yarm FR, Jang YJ, Liu X, Erikson RL . 2000 Proc. Natl. Acad. Sci. USA 97: 12589–12594
Meijer L, Azzi L, Wang JY . 1991 EMBO J. 10: 1545–1554
Morgan DO . 1997 Annu. Rev. Cell Dev. Biol. 13: 261–291
Mueller PR, Coleman TR, Kumagai A, Dunphy WG . 1995 Science 270: 86–90
Nigg EA . 2001 Nat. Rev. Mol. Cell Biol. 2: 21–32
Ohi R, Gould KL . 1999 Curr. Opin. Cell Biol. 11: 267–273
Porter CM, Havens MA, Clipstone NA . 2000 J. Biol. Chem. 275: 3543–3551
Solomon MJ, Lee T, Kirschner MW . 1992 Mol. Biol. Cell 3: 13–27
Strausfeld U, Labbe JC, Fesquet D, Cavadore JC, Picard A, Sadhu K, Russell P, Doree M . 1991 Nature 351: 242–245
Toyoshima-Morimoto F, Taniguchi E, Shinya N, Iwamatsu A, Nishida E . 2001 Nature 410: 215–220
Watanabe N, Broome M, Hunter T . 1995 EMBO J. 14: 1878–1891
Yang J, Bardes ES, Moore JD, Brennan J, Powers MA, Kornbluth S . 1998 Genes Dev. 12: 2131–2143
Yang J, Song H, Walsh S, Bardes ES, Kornbluth S . 2000 J. Biol. Chem. 276: 3604–3609
Yuan J, Horlin A, Hock B, Stutte HJ, Rubsamen-Waigmann H, Strebhardt K . 1997 Am. J. Pathol. 150: 1165–1172
Acknowledgements
We are grateful to A Krämer and O Möbert for their excellent technical support. We thank Dr J Pines for providing pCMX-cyclin B1 and Dr M Yoshida for leptomycin B. Many thanks also go to Drs Raymond L Erikson, Chin-Yo Lin and Xiaoqi Liu for kindly providing Plk1 from insect cells. This work was supported by grants from the Deutsche Krebshilfe (10-1212-St 1), the DFG (STR/8-1), the Messer Stiftung, the Sander Stiftung and the Dresdner Bank.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yuan, J., Eckerdt, F., Bereiter-Hahn, J. et al. Cooperative phosphorylation including the activity of polo-like kinase 1 regulates the subcellular localization of cyclin B1. Oncogene 21, 8282–8292 (2002). https://doi.org/10.1038/sj.onc.1206011
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1206011
Keywords
This article is cited by
-
Mechanisms for the temporal regulation of substrate ubiquitination by the anaphase-promoting complex/cyclosome
Cell Division (2019)
-
Targeting polo-like kinase 1 suppresses essential functions of alloreactive T cells
Immunologic Research (2016)
-
p21Waf1/Cip1 deficiency causes multiple mitotic defects in tumor cells
Oncogene (2014)
-
Transcriptome of Atlantic Cod (Gadus morhua L.) Early Embryos from Farmed and Wild Broodstocks
Marine Biotechnology (2013)
-
Polo-like kinase 1, on the rise from cell cycle regulation to prostate cancer development
Protein & Cell (2012)