Abstract
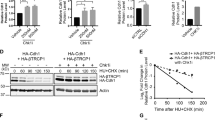
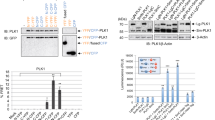
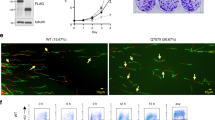
The Polo-like kinases (Plks) are a conserved family of kinases that contribute to cell cycle regulation, particularly in G2 and mitosis. In mammals, there are at least three members of the Plk family. Here we show that Plk3 is a stress response protein that becomes phosphorylated following DNA damage or mitotic spindle disruption. Phosphorylation enhances its kinase activity and is dependent upon ataxia telangiectasia-mutated (ATM) in the former case but not the latter. Plk3 associates with complexes of multiple sizes ranging from 150 to greater then 600 kDa. In its unphosphorylated form it elutes from a sizing column at about 400 kDa whereas it associates with complexes of 150 and 600 kDa when phosphorylated. Among the proteins with which it physically associates and utilizes, as substrates are Chk2 and P53. It phosphorylates Chk2 on a residue different from threonine 68 (Thr68), the principal target for ATM. While ATM is necessary for phosphorylation and activation of Chk2 in vivo, Plk3 seems to contribute to its full activation. In its phosphorylated form it also coelutes and forms a complex with unpolymerized tubulin. In aggregate, the data argue that Plk3 is a multifunctional protein that associates with multiple complexes and that contributes to response to stress incurred by DNA damage and mitotic spindle disruption, albeit via different pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- ATM:
-
ataxia telangiectasia mutated
- CIP:
-
calf intestinal phosphatase
- FPLC:
-
fast protein liquid chromatography
- Plk:
-
polo-like kinase
References
Bahassi E, Hennigan R . 2001 Cancer Research Alert 12: 133–144
Blasina A, de Weyer I, Laus M, Luyten W, Parker A, McGowan C . 1999a Curr. Biol. 9: 1–10
Blasina A, Price B, Turenne G, McGowan C . 1999b Curr. Biol. 9: 1135–1138
Carmena M, Riparbelli M, Minestrini G, Tavares A, Adams R, Callaini G, Glover D . 1998 J. Cell. Biol. 143: 659–671
Chase D, Feng Y, Hanshew B, Winkles J, Longo D, Ferris D . 1998 Biochem. J. 333: 655–660
Cheng L, Hunke L, Hardy C . 1998 Mol. Cell Biol. 18: 7360–7370
Conn CW, Hennigan RF, Dai W, Sanchez Y, Stambrook PJ . 2000 Cancer Res. 60: 6826–6831
Donohue P, Alberts G, Guo Y, Winkles J . 1995 J. Biol. Chem. 270: 10351–10357
Ellinger-Ziegelbauer H, Karasuyama H, Yamada E, Tsujikawa K, Todokoro K, Nishida E . 2000 Genes Cells 5: 491–498
Falck J, Maitland N, Syljnasen R, Bartek J, Lukas J . 2001 Nature 410: 842–847
Golsteyn R, Lane H, Mundt K, Arnaud L, Nigg E . 1996 Prog. Cell Cycle Res. 2: 107–114
Golsteyn R, Mundt K, Fry A, Nigg E . 1995 J. Cell Biol. 129: 1617–1628
Graves P, Lovly C, Uy G, Piwnica-Worms H . 2001 Oncogene 20: 1839–1851
Hirao A, Kong Y, Matsuoka S, Wakeham A, Ruland J, Yoshida H, Liu D, Elledge S, Mak T . 2000 Science 287: 1824–1827
Kim S, Lim D, Canman C, Kastan M . 1999 J. Biol. Chem. 274: 37538–37543
Li B, Ouyang B, Pan H, Reissmann P, Slamon D, Arceci R, Lu L, Dai W . 1996 J. Biol. Chem. 271: 19402–19408
Llamazares S, Moreira A, Tavares A, Girdham C, Spruce B, Gonzalez C, Karess R, Glover D, Sunkel C . 1991 Genes Dev. 5: 2153–2165
Matsuoka S, Huang M, Elledge S . 1998 Science 282: 1893–1897
Matsuoka S, Rotman G, Ogawa A, Shiloh Y, Tamai K, Elledge S . 2000 Proc. Natl. Acad. Sci. USA 97: 10389–10394
Melchionna R, Chen X, Blasina A, McGowan C . 2000 Nat. Cell Biol. 2: 762–765
Nigg E . 1998 Curr. Opin. Cell Biol. 10: 776–783
Ouyang B, Pan H, Lu L, Li J, Stambrook P, Li B, Dai W . 1997 J. Biol. Chem. 272: 28646–28651
Peng C, Graves P, Thoma R, Wu Z, Shaw A, Piwnica-Worms H . 1997 Science 277: 1501–1505
Qian Y, Erikson E, Li C, Maller J . 1998 Mol. Cell. Biol. 18: 4262–4271
Sanchez Y, Bachant J, Wang H, Hu F, Liu D, Tetzlaff M, Elledge SJ . 1999 Science 286: 1166–1171
Sanchez Y, Wong C, Thoma R, Richman R, Wu Z, Piwnica-Worms H, Elledge S . 1997 Science 277: 1497–1501
Smith M, Wilson M, Hamanaka R, Chase D, Kung H, Longo D, Ferris D . 1997 Biochem. Biophys. Res. Commun. 234: 397–405
Smits V, Klompmaker R, Arnaud L, Rijksen G, Nigg E, Medema R . 2000 Nat. Cell Biol. 2: 672–676
Song S, Lee K . 2001 J. Cell Biol. 152: 451–469
Sunkel C, Glover D . 1988 J. Cell Sci. 89: 25–38
Xie S, Wang Q, Wu H, Cogswell J, Lu L, Jhanwar-Uniyal M, Dai W . 2001a J. Biol. Chem. 276: 36194–36199
Xie S, Wu H, Wang Q, Cogswell J, Husain I, Conn C, Stambrook P, Jhanwar-Uniyal M, Dai W . 2001b J. Biol. Chem. 276: 43305–43312
Acknowledgements
This work was supported by the Ruth Lyons Fund and by NIH R01 CA90934, and NIH UO1 ES011038. CW Conn was supported by training grant T32 ES07250.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bahassi, E., Conn, C., Myer, D. et al. Mammalian Polo-like kinase 3 (Plk3) is a multifunctional protein involved in stress response pathways. Oncogene 21, 6633–6640 (2002). https://doi.org/10.1038/sj.onc.1205850
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1205850
Keywords
This article is cited by
-
PLK3 promotes the proneural–mesenchymal transition in glioblastoma via transcriptional regulation of C5AR1
Molecular Biology Reports (2023)
-
Phosphorylation Dynamics Dominate the Regulated Proteome during Early Xenopus Development
Scientific Reports (2017)
-
The role of Plk3 in oncogenesis
Oncogene (2016)
-
Putting a bit into the polo-box domain of polo-like kinase 1
Journal of Analytical Science and Technology (2015)
-
Understanding the Polo Kinase machine
Oncogene (2015)