Abstract
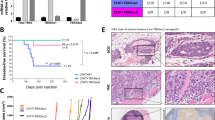
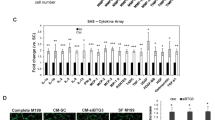
Tumor neovascularization is controlled by a balance between positive and negative effectors, whose production can be regulated by oncogenes and tumor suppressor genes. The aim of this study was to investigate whether the angiogenic potential of tumors could also be controlled by p73, a gene homologous to the tumor suppressor p53, whose involvement in tumor angiogenesis is known. We have studied the production of proangiogenic (VEGF, FGF-2, PIGF and PDGF) and antiangiogenic (TSP-1) factors in two p73 overexpressing clones obtained from the human ovarian carcinoma cells A2780. TSP-1 was downregulated in both clones compared to mock transfected cells, both at mRNA and protein level. Conversely, both clones showed an increased production of VEGF mRNA and protein. For both TSP-1 and VEGF, regulation of expression was partially due to modulation of the promoter activity, and was dependent on p53 status. Production of the other angiogenic factors FGF-2, PIGF and PDGF-B was also increased in p73 overexpressing clones. The two clones were more angiogenic than parental cells, as shown in vitro by their increased chemotactic activity for endothelial cells, and in vivo by the generation of more vascularized tumors. These findings suggest a potential role of p73 in tumor angiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bertin N, Clezardin P, Kubiak R, Frappart L . 1997 Cancer Res. 57: 396–399
Bouck N . 1996 Adv. Canc. Res. 69: 135–174
Chen CL, Ip SM, Cheng D, Wong LC, Ngan HY . 2000 Clin. Cancer Res. 6: 3910–3915
Chi SG, Chang SG, Lee SJ, Lee CH, Kim JI, Park JH . 1999 Cancer Res. 59: 2791–2793
Dameron KM, Volpert OV, Tainsky MA, Bouck N . 1994 Science 265: 1582–1584
Debernardis D, Sire EG, De Feudis P, Vikhanskaya F, Valenti M, Russo P, Parodi S, D'Incalci M, Broggini M . 1997 Cancer Res. 57: 870–874
Dejong V, Degeorges A, Filleur S, Ait-Si-Ali S, Mettouchi A, Bornstein P, Binetruy B, Cabon F . 1999 Oncogene 18: 3143–3151
Grossfeld GD, Ginsberg DA, Stein JP, Bochner BH, Esrig D, Groshen S, Dunn M, Nichols PW, Taylor CR, Skinner DG, Cote RJ . 1997 J. Natl. Cancer Inst. 89: 219–227
Han S, Semba S, Abe T, Makino N, Furukawa T, Fukushige S, Takahashi H, Sakurada A, Sato M, Shiiba K, Matsuno S, Nimura Y, Nakagawara A, Horii A . 1999 Eur. J. Surg. Oncol. 25: 194–198
Hanahan D, Folkman J . 1996 Cell 86: 353–364
Harris CC . 1996 Br. J. Cancer 73: 261–269
Hollstein M, Soussi T, Thomas G, von Brevern MC, Bartsch H . 1997 Recent Results Cancer Res. 143: 369–389
Holmgren L, Jackson G, Arbiser J . 1998 Oncogene 17: 819–824
Hsu SC, Volpert OV, Steck PA, Mikkelsen T, Polverini PJ, Rao S, Chou P, Bouck NP . 1996 Cancer Res. 56: 5684–5691
Kaelin Jr WG . 1999 J. Natl. Cancer Inst. 91: 594–598
Kaghad M, Bonnet H, Yang A, Creancier L, Biscan JC, Valent A, Minty A, Chalon P, Lelias JM, Dumont X, Ferrara P, McKeon F, Caput D . 1997 Cell 90: 809–819
Levrero M, De Laurenzi V, Costanzo A, Gong J, Wang JY, Melino G . 2000 J. Cell. Sci. 113: 1661–1670
Mettouchi A, Cabon F, Montreau N, Vernier P, Mercier G, Blangy D, Tricoire H, Vigier P, Binetruy B . 1994 EMBO J. 13: 5668–5678
Mukhopadhyay D, Tsiokas L, Sukhatme VP . 1995 Cancer Res. 55: 6161–6165
Nagy JA, Masse EM, Herzberg KT, Meyers MS, Yeo KT, Yeo TK, Sioussat TM, Dvorak HF . 1995 Cancer Res. 55: 360–368
Ng SW, Yiu GK, Liu Y, Huang LW, Palnati M, Jun SH, Berkowitz RS, Mok SC . 2000 Oncogene 19: 1885–1890
Nishimori H, Shiratsuchi T, Urano T, Kimura Y, Kiyono K, Tatsumi K, Yoshida S, Ono M, Kuwano M, Nakamura Y, Tokino T . 1997 Oncogene 15: 2145–2150
Nomoto S, Haruki N, Kondo M, Konishi H, Takahashi T . 1998 Cancer Res. 58: 1380–1383
Rak J, Mitsuhashi Y, Bayko L, Filmus J, Shirasawa S, Sasazuki T, Kerbel RS . 1995 Cancer Res. 55: 4575–4580
Saez E, Rutberg SE, Mueller E, Oppenheim H, Smoluk J, Yuspa SH, Spiegelman BM . 1995 Cell 82: 721–732
Salimath B, Marme D, Finkenzeller G . 2000 Oncogene 19: 3470–3476
Salnikow K, Cosentino S, Klein C, Costa M . 1994 Mol. Cell. Biol. 14: 851–858
Sheibani N, Frazier WA . 1995 Proc. Natl. Acad. Sci. USA 92: 6788–6792
Slack JL, Bornstein P . 1994 Cell Growth Differ 5: 1373–1380
Takahashi H, Ichimiya S, Nimura Y, Watanabe M, Furusato M, Wakui S, Yatani R, Aizawa S, Nakagawara A . 1998 Cancer Res. 58: 2076–2077
Taraboletti G, Roberts D, Liotta LA, Giavazzi R . 1990 J. Cell. Biol. 111: 765–772
Taraboletti G, Sonzogni L, Vergani V, Hosseini G, Ceruti R, Ghilardi C, Bastone A, Toschi E, Borsotti P, Scanziani E, Giavazzi R, Pepper MS, Stetler-Stevenson WG, Bani MR . 2000 Exp. Cell. Res. 258: 384–394
Tikhonenko AT, Black DJ, Linial ML . 1996 J. Biol. Chem. 271: 30741–30747
Tokuchi Y, Hashimoto T, Kobayashi Y, Hayashi M, Nishida K, Hayashi S, Imai K, Nakachi K, Ishikawa Y, Nakagawa K, Kawakami Y, Tsuchiya E . 1999 Br. J. Cancer 80: 1623–1629
Vecchi A, Garlanda C, Lampugnani MG, Resnati M, Matteucci C, Stopacciaro A, Schnurch H, Risua W, Ruco L, Mantovani A, Dejana E . 1994 Eur. J. Cell. Biol. 63: 247–254
Vikhanskaya F, D'Incalci M, Broggini M . 2000 Nucleic Acids Res. 28: 513–519
Vikhanskaya F, Marchini S, Marabese M, Galliera E, Broggini M . 2001 Cancer Research 61: 935–938
Vogelstein B, Lane D, Levine AJ . 2000 Nature 408: 307–310
Volpert OV, Dameron KM, Bouck N . 1997 Oncogene 14: 1497–1502
Wen S, Stolarov J, Myers MP, Su JD, Wigler MH, Tonks NK, Durden DL . 2001 Proc. Natl. Acad. Sci. USA 98: 4622–4627
Yang A, Walker N, Bronson R, Kaghad M, Oosterwegel M, Bonnin J, Vagner C, Bonnet H, Dikkes P, Sharpe A, McKeon F, Caput D . 2000 Nature 404: 99–103
Yokomizo A, Mai M, Tindall DJ, Cheng L, Bostwick DG, Naito S, Smith DI, Liu W . 1999 Oncogene 18: 1629–1633
Zabrenetzky VS, Harris CC, Steeg PS, Roberts DD . 1994 Int. J. Cancer 59: 191–195
Zwahlen D, Tschan MP, Grob TJ, Peters UR, Fink D, Haenggi W, Altermatt HJ, Cajot JF, Tobler A, Fey MF, Aebi S . 2000 Int. J. Cancer 88: 66–70
Acknowledgements
We would like to thank GL Semenza (Baltimore, MD, USA) and P Bornstein (Seattle, WA, USA) for the VEGF and TSP-1 promoter constructs, B Vogelstein (Baltimore, MD) for HCT116+/+ and HCT116 −/−, A Vecchi (Milan, Italy) for the anti-CD31 antibody, M Martinelli for in vivo experiments, and E Scanziani (Milan, Italy) for helpful comments on the histochemical analysis of tumors. This work was partially supported by grants from the Italian Association for Cancer Research (AIRC), and the Italian Foundation for Cancer Research (FIRC). F Vikhanskaya is a visiting scientist from the Institute of Cytology (Russian Accademy of Sciences), St. Petersburg, Russia.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vikhanskaya, F., Bani, M., Borsotti, P. et al. p73 overexpression increases VEGF and reduces thrombospondin-1 production: implications for tumor angiogenesis. Oncogene 20, 7293–7300 (2001). https://doi.org/10.1038/sj.onc.1204896
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204896
Keywords
This article is cited by
-
Prediction of chemo-response in serous ovarian cancer
Molecular Cancer (2016)
-
Novel endogenous angiogenesis inhibitors and their therapeutic potential
Acta Pharmacologica Sinica (2015)
-
Hypoxia-inducible TAp73 supports tumorigenesis by regulating the angiogenic transcriptome
Nature Cell Biology (2015)
-
p73 is required for endothelial cell differentiation, migration and the formation of vascular networks regulating VEGF and TGFβ signaling
Cell Death & Differentiation (2015)
-
Expression of human papillomavirus type 16 E6 and E7 oncoproteins in primary foreskin keratinocytes is sufficient to alter the expression of angiogenic factors
Oncogene (2004)