Abstract
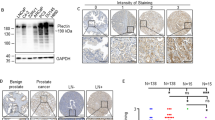
Membrane receptors coupled to heterotrimeric G-proteins play an essential role in the transmission of signals from the extracellular environment to the cytoplasm of the cell. A wide variety of external stimuli, including neurotransmitters, hormones, phospholipids, photons, odorants, taste ligands, and growth factors, can activate specific members of the G-protein coupled receptors (GPCRs). Besides essential functions in fully differentiated cells and tissues, GPCRs are also involved in embryogenesis, tissue regeneration, cell growth stimulation, and cell proliferation. In this study, we identified a novel prostate-specific G-protein coupled receptor that interacts with Gα12 in our yeast two-hybrid assays. The expression of the receptor protein is highly restricted to human prostate tissues using multiple-tissue Northern blot analysis, and tissue expression array. Furthermore, the expression of prostate-specific receptor is increased significantly in prostate tumors in comparison with the matched normal prostate tissues using PCR and Southern blot analysis, suggesting a potential role of this tissue-specific G-protein coupled receptor in prostate cancer development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Abe K, Kusakabe Y, Tanemura K, Emori Y, Arai S . 1993 J. Biol. Chem. 268: 12033–12039
Allen LF, Lefkowitz RJ, Caron MG, Cotecchia S . 1991 Proc. Natl. Acad. Sci. USA 88: 11354–11358
Chan AM, Fleming TP, McGovern ES, Chedid M, Miki T, Aaronson SA . 1993 Mol. Cell. Biol. 13: 762–768
Collins LR, Ricketts WA, Olefsky JM, Brown JH . 1997 Oncogene 15: 595–600
Crespo P, Xu N, Simonds WF, Gutkind JS . 1994 Nature 369: 418–420
Dermott JM, Reddy MR, Onesime D, Reddy EP, Dhanasekaran N . 1999 Oncogene 18: 7185–7189
Dhanasekaran N, Dermott JM . 1996 Cell Signal 8: 235–245
Dhanasekaran N, Heasley LE, Johnson GL . 1995 Endocr. Rev. 16: 259–270
Edwards SW, Tan CM, Limbird LE . 2000 Trends Pharmacol. Sci. 21: 304–308
Gohla A, Offermanns S, Wilkie TM, Schultz G . 1999 J. Biol. Chem. 274: 17901–17907
Gutkind JS . 1998a Oncogene 17: 1331–1342
Gutkind JS . 1998b J. Biol. Chem. 273: 1839–1842
Gutkind JS, Novotny EA, Brann MR, Robbins KC . 1991 Proc. Natl. Acad. Sci. USA 88: 4703–4707
Hart MJ, Jiang X, Kozasa T, Roscoe W, Singer WD, Gilman AG, Sternweis PC, Bollag G . 1998 Science 280: 2112–2114
Jiang H, Wu D, Simon MI . 1993 FEBS Lett. 330: 319–322
Jiang Y, Ma W, Wan Y, Kozasa T, Hattori S, Huang XY . 1998 Nature 395: 808–813
Jones TL, Gutkind JS . 1998 Biochemistry 37: 3196–3202
Julius DLT, Jessell TM, Axel R . 1989 Science 244: 1057–1062
Kozasa T, Jiang X, Hart MJ, Sternweis PM, Singer WD, Gilman AG, Bollag G, Sternweis PC . 1998 Science 280: 2109–2111
Landis CA, Masters SB, Spada A, Pace AM, Bourne HR, Vallar L . 1989 Nature 340: 692–696
Lin X, Voyno-Yasenetskaya TA, Hooley R, Lin CY, Orlowski J, Barber DL . 1996 J. Biol. Chem. 271: 22604–22610
Lopez-Ilasaca M, Crespo P, Pellici PG, Gutkind JS, Wetzker R . 1997 Science 275: 394–397
Lyons JLC, Harsh G, Vallar L, Grunewald K, Feichtinger H, Duh QY, Clark OH, Kawasaki E, Bourne HR, et al . 1990 Science 249: 655–659
Marinissen MJ, Chiariello M, Pallante M, Gutkind JS . 1999 Mol. Cell. Biol. 19: 4289–4301
Radhika V, Dhanasekaran N . 2001 Oncogene 20: 1607–1614
Rhee SG, Choi KD . 1992 Adv. Sec. Mess. Phosphopro. Res. 26: 35–61
Simon MI, Strathmann MP, Gautam N . 1991 Science 252: 802–808
Voyno-Yasenetskaya TA, Faure MP, Ahn NG, Bourne HR . 1996 J. Biol. Chem. 271: 21081–21087
Wess J . 1997 FASEB J. 11: 346–354
Whitehead IP, Zohn IE, Der CJ . 2001 Oncogene 20: 1547–1555
Xu LL, Stackhouse BG, Florence K, Zhang W, Shanmugam N, Sesterhenn IA, Zou Z, Srikantan V, Augustus M, Roschke V, Carter K, McLeod DG, Moul JW, Soppett D, Srivastava S . 2000 Cancer Res. 60: 6568–6572
Xu N, Bradley L, Ambdukar I, Gutkind JS . 1993 Proc. Natl. Acad. Sci. USA 90: 6741–6745
Xu N, Voyno-Yasenetskaya T, Gutkind JS . 1994 Biochem. Biophys. Res. Commun. 201: 603–609
Young D, Waitches G, Birchmeier C, Fasano O, Wigler M . 1986 Cell 45: 711–719
Acknowledgements
This work was supported in part by a Scientist Development Grant from American Heart Association National and the Basil O'Connor Starter Scholar Award from March of Dimes Foundation (to M Liu).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xia, C., Ma, W., Wang, F. et al. Identification of a prostate-specific G-protein coupled receptor in prostate cancer. Oncogene 20, 5903–5907 (2001). https://doi.org/10.1038/sj.onc.1204803
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204803
Keywords
This article is cited by
-
Therapeutic potential of ectopic olfactory and taste receptors
Nature Reviews Drug Discovery (2019)
-
Medium-chain fatty acids modulate myocardial function via a cardiac odorant receptor
Basic Research in Cardiology (2017)
-
Prostate-specific G-protein-coupled receptor collaborates with loss of PTEN to promote prostate cancer progression
Oncogene (2016)
-
Prostate specific G protein coupled receptor is associated with prostate cancer prognosis and affects cancer cell proliferation and invasion
BMC Cancer (2015)
-
PSGR promotes prostatic intraepithelial neoplasia and prostate cancer xenograft growth through NF-κB
Oncogenesis (2014)