Abstract
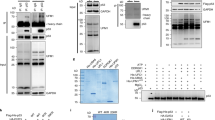
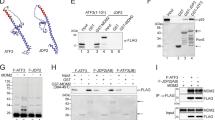
Mdm2 has been shown to promote its own ubiquitination and the ubiquitination of the p53 tumour suppressor by virtue of its E3 ubiquitin ligase activity. This modification targets Mdm2 and p53 for degradation by the proteasome. The p14ARF tumour suppressor has been shown to inhibit degradation of p53 mediated by Mdm2. Several models have been proposed to explain this effect of p14ARF. Here we have compared the effects of p14ARF overexpression on the in vivo ubiquitination of p53 and Mdm2. We report that the inhibition of the Mdm2-mediated degradation of p53 by p14ARF is associated with a decrease in the proportion of ubiquitinated p53. The levels of polyubiquitinated p53 decreased preferentially compared to monoubiquitinated species. p14ARF overexpression increased the levels of Mdm2 but it did not reduce the overall levels of ubiquitinated Mdm2 in vivo. This is unexpected because p14ARF has been reported to inhibit the ubiquitination of Mdm2 in vitro. In addition we show that like p14ARF, the proteasome inhibitor MG132 can promote the accumulation of Mdm2 in the nucleolus and that this can occur in the absence of p14ARF expression. We also show that the mutation of the nucleolar localization signal of Mdm2 does not impair the overall ubiquitination of Mdm2 but is necessary for the effective polyubiquitination of p53. These studies reveal important differences in the regulation of the stability of p53 and of Mdm2.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
An WG, Chuman Y, Fojo T, Blagosklonny MV . 1998 Exp. Cell Res. 244: 54–60
Bottger A, Bottger V, Sparks A, Liu WL, Howard SF, Lane DP . 1997 Curr. Biol. 7: 860–869
Boyd SD, Tsai KY, Jacks T . 2000 Nature Cell Biol. 2: 563–568
Chen J, Marechal V, Levine AJ . 1993 Mol. Cell Biol. 13: 4107–4114
Ciechanover A . 1998 EMBO J. 17: 7151–7160
Fang S, Jensen JP, Ludwig RL, Vousden KH, Weissman AM . 2000 J. Biol. Chem. 275: 8945–8951
Geyer RK, Zhong KY, Maki CG . 2000 Nature Cell Biol. 2: 569–573
Haupt Y, Maya R, Kazaz A, Oren M . 1997 Nature 387: 296–299
Honda R, Tanaka H, Yasuda H . 1997 FEBS Lett. 420: 25–27
Honda R, Yasuda H . 1999 EMBO J. 18: 22–27
Honda R, Yasuda H . 2000 Oncogene 19: 1473–1476
Ko LJ, Prives C . 1996 Genes Dev. 10: 1054–1072
Kubbutat MHG, Jones SN, Vousden KH . 1997 Nature 387: 299–303
Laín S, Midgley C, Sparks A, Lane EB, Lane DP . 1999 Exp. Cell Res. 248: 457–472
Lane DP, Hall PA . 1997 Trends. Biochem. Sci. 22: 372–374
Levine AJ . 1997 Cell 88: 323–331
Lohrum MAE, Ashcroft M, Kubbutat MHG, Vousden KH . 2000 Nature Cell Biol. 2: 179–181
Maltzman W, Czyzyk L . 1984 Mol. Cell. Biol. 4: 1689–1694
Midgley CA, Desterro JMP, Saville MK, Howard S, Sparks A, Hay RT, Lane DP . 2000 Oncogene 19: 2312–2323
Midgley CA, Fisher CJ, Bartek J, Vojtesek B, Lane D, Barnes DM . 1992 J. Cell Sci. 101: 183–189
Midgley CA, Owens B, Briscoe CV, Thomas DB, Lane DP, Hall PA . 1995 J. Cell. Sci. 108: 1843–1848
Oren M . 1999 J. Biol. Chem. 274: 36031–36034
Pochampally R, Fodera B, Chen L, Shao W, Levine EA, Chen J . 1998 Oncogene 17: 2629–2636
Pomerantz J, SchreiberAgus N, Liegeois NJ, Silverman A, Alland L, Chin L, Potes J, Chen K, Orlow I, Lee HW, CordonCardo C, DePinho RA . 1998 Cell 92: 713–723
Rodriguez MS, Desterro JMP, Laín S, Midgley CA, Lane DP, Hay RT . 1999 EMBO J. 18: 6455–6461
Rodriguez MS, Desterro JM, Laín S, Lane DP, Hay RT . 2000 Mol. Cell. Biol. 20: 8458–8467
Stephen CW, Helminen P, Lane DP . 1995 J. Mol. Biol. 248: 58–78
Stott FJ, Bates S, James MC, McConnell BB, Starborg M, Brookes S, Palmero I, Ryan K, Hara E, Vousden KH, Peters G . 1998 EMBO J. 17: 5001–5014
Tao WK, Levine AJ . 1999 Proc. Natl. Acad. Sci., USA 96: 6937–6941
Thrower JS, Hoffman L, Rechsteiner M, Pickart CM . 1999 EMBO J. 19: 94–102
Treier M, Staszewski LM, Bohmann D . 1994 Cell 78: 787–798
Vousden KH . 2000 Cell 103: 691–694
Wang Y, Blandino G, Oren M, Givol D . 1998 Oncogene 17: 1923–1930
Weber JD, Kuo M-L, Bothner B, DiGiammarino EL, Kriwacki RW, Roussel MF, Sherr CJ . 2000 Mol. Cell. Biol. 20: 2517–2528
Weber JD, Taylor LJ, Roussel MF, Sherr CJ, BarSagi D . 1999 Nat. Cell. Biol. 1: 20–26
Zhang YP, Xiong Y . 1999 Mol. Cell 3: 579–591
Zhang YP, Xiong Y, Yarbrough WG . 1998 Cell 92: 725–734
Acknowledgements
We are grateful to Drs M Oren, K Vousden, C Sherr, A Levine and S Mittnacht for providing us with reagents. We also thank our colleagues in Dundee, E Warbrick and C Stephen for comments and critical review of the manuscript and C Midgley for providing the vector expressing the Mdm2 C464A mutant. D Xirodimas, S Laín and M Saville are recipients of postdoctoral fellowships from Cancer Research Campaign (UK) and Tenovus (Scotland). DP Lane is a Gibb fellow of the Cancer Research Campaign.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xirodimas, D., Saville, M., Edling, C. et al. Different effects of p14ARF on the levels of ubiquitinated p53 and Mdm2 in vivo. Oncogene 20, 4972–4983 (2001). https://doi.org/10.1038/sj.onc.1204656
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204656
Keywords
This article is cited by
-
The E3 ubiquitin-protein ligase MDM2 is a novel interactor of the von Hippel–Lindau tumor suppressor
Scientific Reports (2020)
-
Genome-wide screening identifies novel genes implicated in cellular sensitivity to BRAFV600E expression
Oncogene (2020)
-
Influenza A viruses alter the stability and antiviral contribution of host E3-ubiquitin ligase Mdm2 during the time-course of infection
Scientific Reports (2018)
-
NEDDylation promotes nuclear protein aggregation and protects the Ubiquitin Proteasome System upon proteotoxic stress
Nature Communications (2018)
-
The NEDD8 inhibitor MLN4924 increases the size of the nucleolus and activates p53 through the ribosomal-Mdm2 pathway
Oncogene (2016)