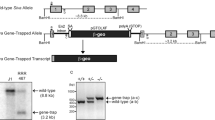
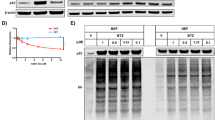
Abstract
The p53 tumor suppressor gene product is negatively regulated by the product of its downstream target, mdm2. The deletion of mdm2 in the mouse results in embryonic lethality at 5.5 days post coitum (d.p.c.) which can be overcome by simultaneous loss of the p53 tumor suppressor, substantiating the importance of the negative regulatory function of MDM2 on p53 function in vivo. Hence, the loss of MDM2 allowed the unregulated p53 protein to continuously exert its growth-suppressing activity, which either led to a complete G1 arrest or induced the p53-dependent apoptotic pathway, resulting in the death of the mdm2−/− embryos. To determine which of these possibilities is occurring, mouse embryo fibroblasts (MEFs) from p53 null and p53/mdm2 double null embryos were transfected with a retroviral vector carrying a temperature-sensitive p53 (tsp53) cDNA. Shifting of single-cell clonal populations to the permissive temperature caused the p53−/−mdm2−/− fibroblasts expressing tsp53 to undergo apoptosis in a dose-dependent manner. This phenotype was not observed in the tsp53 expressing p53−/− clones nor the parental cell lines. Thus, our data indicate that the simple loss of mdm2 can induce the p53-dependent apoptotic pathway in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Baker SJ, Markowitz S, Fearon E, Willson JKV and Vogelstein B . 1990 Science 249: 912–915
Barak Y and Oren M . 1992 EMBO J 11: 2115–2121
Barak Y, Juven T, Haffner R and Oren M . 1993 EMBO J 12: 461–468
Buckbinder L, Talbott R, Velasco-Miguel S, Takenaka I, Faha B, Seizinger BR and Kley N . 1995 Nature 377: 646–649
Caron de Fromentel C and Soussi T . 1992 Genes Chrom Cancer 4: 1–15
Chen PL, Chen Y, Bookstein R and Lee WH . 1990 Science 250: 1576–1580
Chen X, Ko L, Jayaraman L and Prives C . 1996 Genes Dev 10: 2438–2451
Clarke AR, Purdie CA, Harrison DJ, Morris RG, Bird CC, Hooper ML and Wyllie AH . 1993 Nature 362: 849–852
Clarke AR, Gledhill S, Hooper ML, Bird CC and Wyllie AH . 1994 Oncogene 9: 1767–1773
Cordon-Cardo C, Latres E, Drobnjak M, Oliva MR, Pollack D, Woodruff JM, Marechal V, Chen J, Brennan MF and Levine AJ . 1994 Cancer Res 54: 794–799
Deffie A, Hao M, Montes de Oca Luna R, Hulboy DL and Lozano G . 1995 Mol Cell Biol 15: 3926–3933
Diller L, Kassel J, Nelson CE, Gryka MA, Litwak G, Gebhardt M, Bressac B, Ozturk M, Baker SJ, Vogelstein B and Friend SH . 1990 Mol Cell Biol 10: 5772–5781
Donehower LA, Harvey M, Slagle B, McArthur MJ, Montgomery CA, Butel J and Bradley A . 1992 Nature 356: 215–221
Dulic V, Kaufmann WK, Wilson S, Tlsty T, Lees E, Harper JW, Elledge S and Reed S . 1994 Cell 76: 1013–1023
Fields S and Jang SK . 1990 Science 249: 1046–1049
Friedlander P, Haupt Y, Prives C and Oren M . 1996 Mol Cell Biol 16: 4961–4971
Gannon JV and Lane DP . 1991 Nature 349: 802–806
Ginsberg D, Michalovitz D, Ginsberg D and Oren M . 1991 Mol Cell Biol 11: 582–585
Gottlieb TM and Oren M . 1996 Biochim Biophys Acta 1287: 77–102
Gottlieb TM and Oren M . 1998 Seminars in Cancer Biology 8: 359–368
Gu Y, Turck CW and Morgan DO . 1993 Nature 366: 707–710
Harper JW, Adami GR, Wei N, Keyomarsi K and Elledge SJ . 1993 Cell 75: 805–816
Haupt Y, Maya R, Kazaz A and Oren M . 1997 Nature 387: 296–299
Hollstein M, Sidransky D, Vogelstein B and Harris CC . 1991 Science 253: 49–53
Itoh N, Yonehara S, Ishii A, Yonehara M, Mizushima SI, Sameshima M, Hase A, Seto Y and Nagata S . 1991 Cell 66: 233–243
Jacks T, Remington L, Williams BO, Schmitt E, Halachmi S, Bronson RT and Weinberg RA . 1994 Curr Biol 4: 1–7
Johnson P, Gray D, Mowat M and Benchimol S . 1991 Mol Cell Biol 11: 1–11
Jones SN, Roe AE, Donehower LA and Bradley A . 1995 Nature 378: 206–208
Kastan MB, Radin AI, Kuerbitz SJ, Onyekwere O, Wolkow CA, Civin CI, Stone KD, Woo T, Ravindranath Y and Craig RW . 1991a Cancer Res 51: 4279–4286
Kastan MB, Onyekwere O, Sidransky D, Vogelstein B and Craig RW . 1991b Cancer Res 51: 6304–6311
Kastan MB, Zhan Q, El-Deiry WS, Carrier F, Jacks T, Walsh WV, Plunkett BS, Vogelstein B and Fornace Jr AJ . 1992 Cell 71: 587–597
Kern SE, Kinzler KW, Bruskin A, Jarosz D, Friedman P, Prives C and Vogelstein B . 1991 Science 252: 1708–1711
Ko LJ and Prives C . 1996 Genes Dev 10: 1054–1072
Kubbutat M, Jones SN and Vousden KH . 1997 Nature 387: 299–303
Kuerbitz SJ, Plunkett BS, Walsh WV and Kastan MB . 1992 Proc Natl Acad Sci USA 89: 7491–7495
Lin D, Shields MT, Ullrich SJ, Appella E and Mercer WE . 1992 Proc Natl Acad Sci USA 89: 9210–9214
Lotem J and Sachs L . 1993 Blood 82: 1092–1096
Lowe SW, Schmitt EM, Smith SW, Osborne BA and Jacks T . 1993 Nature 362: 847–849
Lozano G and Montes de Oca Luna R . 1998 Biochim Biophys Acta 1377: M55–M59
Ludwig RL, Bates S and Vousden KH . 1996 Mol Cell Biol 16: 4952–4960
Marshall CJ . 1991 Cell 64: 313–326
Martinez J, Georgoff I, Martinez J and Levine AJ . 1991 Genes Dev 5: 151–159
Mayo LD, Turchi JJ and Berberich SJ . 1997 Cancer Res 57: 5013–5016
McMasters KM, Montes de Oca Luna R, Pena J and Lozano G . 1996 Oncogene 13: 1731–1736
Mercer WE, Shields MT, Amin M, Sauve GJ, Appella E, Romano JW and Ullrich SJ . 1990 Proc Natl Acad Sci USA 87: 6166–6170
Michalovitz D, Halevy O and Oren M . 1990 Cell 62: 671–680
Miyashita T, Krajewski S, Krajewska M, Wang HG, Lin HK, Hoffman B, Lieberman DA and Reed JC . 1994 Oncogene 9: 1799–1805
Miyashita T and Reed JC . 1995 Cell 80: 293–299
Momand J, Zambetti GP, Olson DC, George D and Levine AJ . 1992 Cell 69: 1237–1245
Momand J, Jung D, Wilezynski S and Niland J . 1998 Nuc Acids Res 26: 3453–3459
Montes de Oca Luna R, Wagner DS and Lozano G . 1995 Nature 378: 203–206
Muller AJ, Young JC, Pendergast A, Pondel M, Landau NR, Littman DR and Witte ON . 1991 Mol Cell Biol 11: 1785–1792
Muller M, Wilder S, Bannasch D, Israeli D, Lehlbach K, Li-Weber M, Friedman SL, Galle PR, Stremmel W, Oren M and Krammer PH . 1998 J Exp Med 188: 2033–2045
Okamoto K and Beach D . 1994 EMBO J 13: 4816–4822
Oliner JD, Kinzler KW, Meltzer PS, George DL and Vogelstein B . 1992 Nature 358: 80–83
Oliner JD, Pietenpol JA, Thiagalingam S, Guyris J, Kinzler K and Vogelstein B . 1993 Nature 362: 857–860
Owen-Schaub L, Zhang W, Cusack JC, Angelo LS, Santee S, Fujiwara T, Roth Deisseroth A, Zhang W, Kruzel E and Radinsky R . 1995 Mol Cell Biol 15: 3032–3040
Pear WS, Nolan GP, Scott ML and Baltimore D . 1993 Proc Natl Acad Sci USA 90: 8392–8396
Piette J, Neel H and Marechal V . 1997 Oncogene 15: 1001–1010
Raycroft L, Wu H and Lozano G . 1990 Science 249: 1049–1051
Raycroft L, Schmidt JR, Yoas K, Hao M and Lozano G . 1991 Mol Cell Biol 11: 6067–6074
Ryan JJ, Danish R, Gottlieb CA and Clarke MF . 1993 Mol Cell Biol 13: 711–719
Shieh SY, Ikeda M, Taya Y and Prives C . 1997 Cell 91: 325–334
Sheikh MS, Shao Z, Hussain A and Fontana J . 1993 Cancer Res 53: 3226–3228
Tishler RB, Calderwood SK, Coleman CN and Price BD . 1993 Cancer Res 53: 2212–2216
Tamura T, Aoyama N, Saya H, Haga H, Futami S, Miyamoto M, Koh T, Ariyasu T, Tachi M, Kasuga M and Takahashi R . 1995 Oncogene 11: 1939–1946
Wu X, Bayle JH, Olson D and Levine AJ . 1993 Genes Dev 7: 1126–1132
Xiong Y, Hannon GJ, Zhang H, Casso D, Kobayashi R and Beach D . 1993 Nature 366: 701–704
Yonish-Rouach E, Resnitzky D, Lotem J, Sachs L, Kimchi A and Oren M . 1991 Nature 352: 345–347
Yonish-Rouach E, Grunwald D, Wilder S, Kimchi A, May E, Lawrence JJ, May P and Oren M . 1993 Mol Cell Biol 13: 1415–1423
Zauberman A, Lupo A and Oren M . 1995 Oncogene 10: 2361–2366
Acknowledgements
We thank Sylvie Wilder for expert technical assistance and Susan Evans and Mini Kapoor for constructive criticism and advice on the manuscript. This work was supported by grants from the NCI (CA47296) to G Lozano and (CA40099) to M Oren and in part from funds for The Faculty Achievement Award in Basic Research to G Lozano.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Rozieres, S., Maya, R., Oren, M. et al. The loss of mdm2 induces p53 mediated apoptosis. Oncogene 19, 1691–1697 (2000). https://doi.org/10.1038/sj.onc.1203468
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1203468
Keywords
This article is cited by
-
Activating transcription factor 3 mediates apoptotic functions through a p53-independent pathway in human papillomavirus 18 infected HeLa cells
Virus Genes (2022)
-
USP7 inhibitor P22077 inhibits neuroblastoma growth via inducing p53-mediated apoptosis
Cell Death & Disease (2013)
-
Molecular mechanisms of melatonin’s inhibitory actions on breast cancers
Cellular and Molecular Life Sciences (2013)
-
Spinal Muscular Atrophy and the Antiapoptotic Role of Survival of Motor Neuron (SMN) Protein
Molecular Neurobiology (2013)
-
Novel targeted therapeutics: inhibitors of MDM2, ALK and PARP
Journal of Hematology & Oncology (2011)