Abstract
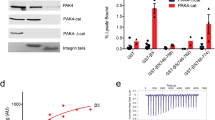
The SH2-SH3 adaptor protein Crkl has been implicated in the signal transduction pathways of several membrane-bound receptors. Tyrosine phosphorylation of proteins associated with such signalling complexes can generate binding sites for the Crkl SH2-domain and can recruit proteins constitutively bound to Crkl via the Crkl SH3 domain into such complexes. In the current study we show that Crkl, but only a minor amount of the related Crk, form constitutive complexes in vivo with guanine nucleotide exchange factor C3G in 3T3 fibroblasts. Adhesion of both normal and transformed cells to fibronectin or other extracellular matrix proteins consistently induces the tyrosine-phosphorylation of C3G. Adhesion-induced tyrosine phosphorylation of C3G is dependent on an intact cytoskeleton and peaks at 5–10 min after attachment. In contrast, 3T3 cells stably transfected with Bcr/Abl P210 show a prominent reduction in the amount of C3G complexed to Crkl and do not exhibit tyrosine-phosphorylation of C3G upon spreading and attachment. These data establish that integrin-mediated cell adhesion results in Crkl-mediated tyrosine phosphorylation of C3G, a pathway which can be disrupted by Bcr/Abl.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
de Jong, R., van Wijk, A., Heisterkamp, N. et al. C3G is tyrosine-phosphorylated after integrin-mediated cell adhesion in normal but not in Bcr/Abl expressing cells. Oncogene 17, 2805–2810 (1998). https://doi.org/10.1038/sj.onc.1202207
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202207
Keywords
This article is cited by
-
C3G forms complexes with Bcr-Abl and p38α MAPK at the focal adhesions in chronic myeloid leukemia cells: implication in the regulation of leukemic cell adhesion
Cell Communication and Signaling (2013)
-
Anaplastic lymphoma kinase activates the small GTPase Rap1 via the Rap1-specific GEF C3G in both neuroblastoma and PC12 cells
Oncogene (2010)
-
Phospholipase Cγ1 regulates the Rap GEF1-Rap1 signalling axis in the control of human prostate carcinoma cell adhesion
Oncogene (2008)
-
C3G-mediated suppression of oncogene-induced focus formation in fibroblasts involves inhibition of ERK activation, cyclin A expression and alterations of anchorage-independent growth
Oncogene (2004)
-
Crk family adaptors–signalling complex formation and biological roles
Oncogene (2001)