Abstract
Background: The objective of this study was to analyse the relationship of polymorphisms of the angiotensinogen (AGT) gene with the changes in microalbuminuria during 3 years of antihypertensive treatment in a group of young adults with essential hypertension.
Methods: Essential hypertensives, less than 50 years old, never previously treated with antihypertensive drugs and in the absence of diabetes mellitus were included. After the initial evaluation, patients were treated using only nonpharmacological measures (n=23), only β-blockers (n=26), only angiotensin-converting enzyme inhibitors (ACEi) (n=57) or a combination of treatments (n=25). The office blood pressure, biochemical profile and urinary albumin excretion (UAE) were measured at the beginning and then yearly. The polymorphism A-6G of the AGT gene located in the promoter region was analysed.
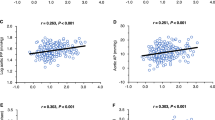
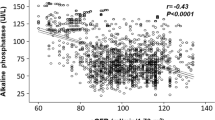
Results: In total, 131 patients, 35 (27%) microalbuminurics, were included. Although no significant differences in systolic blood pressure (SBP), diastolic blood pressure (DBP), fasting glucose and UAE were observed among genotypes at the initial examination, during the 3 years of antihypertensive treatment the slope values for the DBP, fasting glucose and UAE differed significantly despite no differences in the distribution of treatments being present. The subjects carrying the AA-6 genotype had the largest DBP decrease, but the lowest UAE reduction and the highest slope of glucose. Out of 35 initially microalbuminuric patients, 24 became normoalbuminuric and the lowest reduction rates were observed in subjects who carried the allele A-6. No interaction between the type of treatment and genotype was observed on the changes in UAE, BP or glucose values. In the subset of 57 patients treated with ACEi, the changes in UAE, BP and glucose had the same trend as was observed in the total population.
Conclusions: Subjects carrying the AA genotype of the A-6G AGT gene polymorphism are resistant to a reduction of microalbuminuria. Whether this can be attributed to a predisposition to glucose metabolic disturbance or not needs to be confirmed in further studies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Bigazzi R, Bianchi S, Baldari D, Campese VM . Microalbuminuria predicts cardiovascular events and renal insufficiency in patients with essential hypertension. J Hypertens 1998; 16: 1325–1333.
Jensen JS et al. Arterial hypertension, microalbuminuria and risk of ischemic heart diseases. Hypertension 2000; 35: 898–903.
Redon J et al. Factors related to the presence of microalbuminuria in essential hypertension. Am J Hypertens 1994; 7: 801–807.
Redon J et al. Circadian variability and microalbuminuria in essential hypertension. J Hypertens 1994; 12: 947–954.
Bianchi S et al. Elevated serum insulin levels in patients with essential hypertension and microalbuminuria. Hypertension 1994; 23 (Part 1): 681–687.
Redon J et al. Hyperinsulinemia as a determinant of microalbuminuria in essential hypertension. J Hypertens 1997; 15: 79–86.
Redon J et al. Influence of the I/D polymorphism of the angiotensin converting enzyme gene on the outcome of microalbuminuria in essential hypertension. Hypertension 2000; 35: 490–495.
Chaves FJ et al. Angiotensin II AT1 receptor gene polymorphisms and microalbuminuria in essential hypertension. Am J Hypertens 2001; 14: 364–370.
Jeunemaitre X et al. Molecular basis of human hypertension role of angiotensinogen. Cell 1992; 71: 169–180.
Johnson AG et al. M235 to T polymorphism of the angiotensinogen gene predicts hypertension in the elderly. J Hypertens 1996; 14: 1061–1065.
Sato N et al. association of variants in critical core promoter element of angiotensinogen gene with increased risk of essential hypertension in Japanese. Hypertension 1997; 30: 321–325.
Ishigami T et al. Essential hypertension and 5′ upstream core promoter region of human angiotensinogen gene. Hypertension 1997; 30: 1325–1330.
Inoue I et al. A nucleotide substitution in the promoter of human angiotensinogen is associated with essential hypertension and affects basal transcription in vitro. J Clin Invest 1997; 99: 1786–1797.
Ishigami T et al. Angiotensinogen gene polymorphism near transcription start site and blood pressure: role of a T-to-C transition at intron 1. Hypertension 1999; 34: 430–434.
Kunz R et al. Association between the angiotensinogen 235T variant and essential hypertension in whites. A systematic review and methodological appraisal. Hypertension 1997; 30: 1331–1337.
Provience MA et al. Lack of association of the angiotensinogen −6 polymorphism with blood pressure levels in the comprehensive NHLBI Family Blood Pressure Programme. J Hypertens 2000; 18: 867–876.
Pontremoli R et al. The deletion/insertion polymorphism of the angiotensin I-converting enzyme gene is associated with target organ damage in essential hypertension. J Am Soc Nephrol 1996; 7: 2550–2558.
Yudkin JS et al. The angiotensin-converting enzyme gene and the angiotensin II type I receptor gene as a candidate genes for microalbuminuria. A study in non-diabetic and non-insulin dependent diabetic subjects. Arterioscl Thromb Vasc Biol 1997; 17: 2188–2191.
Fernandez-Llama P et al. Angiotensin converting enzyme I/D polymorphism in essential hypertension and nephroangiosclerosis. Kidney Int 1998; 53: 1743–1747.
Wang W et al. Exclusion of angiotensinogen gene in molecular bases of human hypertension. Am J Med Genet 1999; 87: 53–60.
Onipinla A et al. Relationships between M235T and G(−6)A polymorphism of the angiotensinogen gene. J Hum Hypertens 1999; 13: 865–866.
Lander ES, Schork NJ . Genetic dissection of complex traits. Science 1994; 265: 2037–2048.
Sharma AM, Jeunemaitre X . The future of genetic association studies in hypertension: improving the signal-to-noise ratio. J Hypertens 2000; 18: 811–814.
Gould AB, Green D . Kinetics of the human renin and human substrate reaction. Cardiovasc Res 1971; 5: 86–89.
Redon J . Renal protection by antihypertensive drugs: Insights from microalbuminuria studies. J Hypertens 1998; 16: 2091–2100.
Richey JM, Ader M, Moore D, Bergman RN . Angiotensin II induces insulin resistance independent of changes in interstitial insulin. Am J Physiol 1999; 277 (5 Part 1): E920–926.
Folli F et al. Crosstalk between insulin and angiotensin II signalling systems. Exp Clin Endocrinol Diabetes 1999; 107: 133–139.
Acknowledgements
This study was supported by Grant (018/98) of the Fondo de Investigaciones Sanitarias (FIS), of the Spanish Health Ministry. Pablo Marín was a recipient of a Grant, FPI98-vs-23-008, from the Department of Culture, Education and Science, Generalitat Valenciana.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marin, P., Julve, R., Chaves, F. et al. Polymorphisms of the angiotensinogen gene and the outcome of microalbuminuria in essential hypertension: a 3-year follow-up study. J Hum Hypertens 18, 25–31 (2004). https://doi.org/10.1038/sj.jhh.1001630
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jhh.1001630
Keywords
This article is cited by
-
Development of microalbuminuria in essential hypertension
Current Hypertension Reports (2006)