Abstract
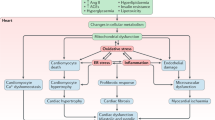
Cardiovascular disease is the most common cause of death in the diabetic population and is currently one of the leading causes of death in the United States and other industrialized countries. The health care expenses associated with cardiovascular disease are staggering, reaching more than US$350 billion in 2003. The risk factors for cardiovascular disease include high fat/cholesterol levels, alcoholism, smoking, genetics, environmental factors and hypertension, which are commonly used to gauge an individual's risk of cardiovascular disease and to track their progress during therapy. Most recently, these factors have become important in the early prevention of cardiovascular diseases. Oxidative stress, the imbalance between reactive oxygen species production and breakdown by endogenous antioxidants, has been implicated in the onset and progression of cardiovascular diseases such as congestive heart failure and diabetes-associated heart dysfunction (diabetic cardiomyopathy). Antioxidant therapy has shown promise in preventing the development of diabetic heart complications. This review focuses on recent advances in oxidative stress theory and antioxidant therapy in diabetic cardiomyopathy, with an emphasis on the stress signaling pathways hypothesized to be involved. Many of these stress signaling pathways lead to activation of reactive oxygen species, major players in the development and progression of diabetic cardiomyopathy.
Similar content being viewed by others
Article PDF
References
Poulter N . Global risk of cardiovascular disease. Heart 2003; 89: 2–5.
Strandberg TE, Salomaa V . Factors related to the development of diabetes during a 20-year follow up. A prospective study in a homogeneous group of middle-aged men. Nutr Metab Cardiovasc Dis 2000; 10: 239–46.
Garcia MJ, McNamara PM, Gordon T, Kannel WB . Morbidity and mortality in diabetics in the Framingham population. Sixteen year follow–up study. Diabetes 1974; 23: 105–11.
Sowers JR . Diabetes mellitus and cardiovascular disease in women. Arch Intern Med 1998; 158: 617–21.
Pradhan AD, Skerrett PJ, Manson JE . Obesity, diabetes, and coronary risk in women. J Cardiovasc Risk 2002; 9: 323–30.
Charpentier G, Genes N, Vaur L, Amar J, Clerson P, Cambou JP, et al. Control of diabetes and cardiovascular risk factors in patients with type 2 diabetes: a nationwide French survey. Diabetes Metab 2003; 29: 152–8.
Li S, Culver B, Ren J . Benefit and risk of exercise on myocardial function in diabetes. Pharmacol Res 2003; 48: 127–32.
Hammes HP, Du X, Edelstein D, Taguchi T, Matsumura T, Ju Q, et al. Benfotiamine blocks three major pathways of hyperglycemic damage and prevents experimental diabetic retinopathy. Nat Med 2003; 93: 294–9.
Feldman EL . Oxidative stress and diabetic neuropathy: a new understanding of an old problem. J Clin Invest 2003; 111: 431–3.
Gilbert RE, Tsalamandris C, Bach LA, Panagiotopoulos S, O'Brien RC, Allen TJ, et al. Long-term glycemic control and the rate of progression of early diabetic kidney disease. Kidney Int 1993; 44: 855–9.
Fein FS, Sonnenblick EH . Diabetic cardiomyopathy. Cardiovasc Drugs Ther 1994; 8: 65–73.
Ren J, Davidoff AJ . Diabetes rapidly induces contractile dysfunctions in isolated ventricular myocytes. Am J Physiol Heart Circ Physiol 1997; 272: H148–58.
Wold LW, Relling DP, Colligan PB, Scott GI, Hintz KK, Ren BH, et al. Characterization of contractile function in diabetic hypertensive cardiomyopathy in adult rat ventricular myocytes. J Mol Cell Cardiol 2001; 33: 1719–26.
Poirier P, Garneau C, Marois L, Bogaty P, Dumesnil JG . Diastolic dysfunction in normotensive men with well-controlled type 2 diabetes: importance of maneuvers in echocardiographic screening for preclinical diabetic cardiomyopathy. Diabetes Care 2001; 24: 5–10.
Buyukgebiz A, Saylam G, Dundar B, Bober E, Unal N, Akcoral A . Dilated cardiomyopathy as the first early complications in a 14 year-old girl with diabetes mellitus type 1. J Pediatr Endocrinol Metab 2000; 13: 1143–6.
Depre C, Young ME, Ying J, Ahuja HS, Han Q, Garza N, et al. Streptozotocin induced changes in cardiac gene expression in the absence of severe contractile dysfunction. J Mol Cell Cardiol 2000; 32: 985–96.
Kang YJ . Molecular and cellular mechanisms of cardiotoxicity. Environ Health Perspect 2001; 109: 27–34.
Ren J, Bode AM . Altered cardiac excitation–contraction coupling in ventricular myocytes from spontaneously diabetic BB rats. Am J Physiol Heart Circ Physiol 2000; 279: H238–44.
Norby FL, Wold LE, Duan J, Hintz KK, Ren J . IGF-I attenuates diabetes-induced cardiac contractile dysfunction in ventricular myocytes. Am J Physiol Endocrinol Metab 2002; 283: E658–66.
Norby FL, Aberle NS, Kajstura J, Anversa P, Ren J . Transgenic overexpression of insulin–like growth factor I prevents streptozotocin–induced cardiac contractile dysfunction and beta–adrenergic response in ventricular myocytes. J Endocrinol 2004; 180: 175–82.
Kajstura J, Fiordaliso F, Andreoli AM, Li B, Chimenti S, Medow MS, et al. IGF-1 overexpression inhibits the development of diabetic cardiomyopathy and angiotensin II-mediated oxidative stress. Diabetes 2001; 50: 1414–24.
Sowers JR, Williams M, Epstein M, Bakris G . Hypertension in patients with diabetes. Strategies for drug therapy to reduce complications. Postgrad Med 2000; 107: 47–54.
Spector KS . Diabetic cardiomyopathy. Clin Cardiol 1998; 21: 885–7.
Rubler S, Dlugash J, Yuceoglu YZ, Kumral T, Branwood AW, Grishman A . New type of cardiomyopathy associated with diabetic glomerulosclerosis. Am J Cardiol 1972; 30: 595–602.
Pierce GN, Russell JC . Regulation of intracellular Ca2+ in the heart during diabetes. Cardiovasc Res 1997; 34: 41–7.
Chatham JC, Forder JR, McNeill JH . The heart in diabetes. Norwell: MA Kluwer Academic Publishers; 1996.
Schaffer SW, Mozafferi M . Abnormal mechanical function in diabetes: relation to myocardial calcium handling. Coron Artery Dis 1996; 7: 109–15.
Dincer UD, Bidasee KR, Guner S, Tay A, Ozcelikay AT, Altan VM . The effect of diabetes on expression of beta1-, beta2-, and beta3-adrenoreceptors in rat hearts. Diabetes 2001; 50: 455–61.
Tamada A, Hattori Y, Houzen H, Yamada Y, Sakuma I, Kitabatake A, et al. Effects of beta-adrenoreceptor stimulation on contractility, [Ca2+]i, and Ca2+ current in diabetic rat cardiomyocytes. Am J Physiol Heart Circ Physiol 1998; 274: H1849–57.
Idris I, Gray S, Donnelly R . Protein kinase C activation: isozymes-specific effects on metabolism and cardiovascular complications in diabetes. Diabetologia 2001; 44: 659–73.
Given MB, Jie O, Zhao X, Giles TD, Greenberg SS . Protein kinase C isozymes in skeletal muscles during the early stage of genetic and streptozotocin diabetes. Proc Soc Exp Biol Med 1998; 218: 382–9.
Devereux RB, Roman MJ, Paranicas M, O'Grady MJ, Lee ET, Welty TK, et al. Impact of diabetes on cardiac structure and function: the strong heart study. Circulation 2000; 101: 2271–6.
Liu JE, Palmieri V, Roman MJ, Bella JN, Fabsitz R, Howard BV, et al. The impact of diabetes on left ventricular filling pattern in normotensive and hypertensive adults: the Strong Heart Study. J Am Coll Cardiol 2001; 37: 1943–9.
Rutter MK, Parise H, Benjamin EJ, Levy D, Larson MG, Meigs JB, et al. Impact of glucose intolerance and insulin resistance on cardiac structure and function: sex–related differences in the Framingham Heart Study. Circulation 2003; 107: 448–54.
Schannwell CM, Schneppenheim M, Perings SM, Zimmermann T, Plehn G, Strauer BE . Alterations of left ventricular function in women with insulin–dependent diabetes mellitus during pregnancy. Diabetologia 2003; 46: 267–75.
Dutta K, Podolin DA, Davidson MB, Davidoff AJ . Cardiomyocyte dysfunction in sucrose–fed rats is associated with insulin resistance. Diabetes 2001; 50: 1186–92.
Zarich SW, Nesto RW . Diabetic cardiomyopathy. Curriculum Cardiol 1989; 118: 1000–12.
Fraser GE, Luk R, Thompson S, Smith H, Carter S, Sharpe N . Comparison of echocardiography variables between Type 1 diabetics and normal control. Am J Cardiol 1995; 75: 141–5.
Raev DC . Which left ventricular function is impaired earlier in the evaluation of diabetic cardiomyopathy. Diabetes Care 1994; 17: 633–9.
Celentano A, Vaccaro O, Tammaro P, Galderisi M, Crivaro M, Oliviero M, et al. Early abnormalities of cardiac function in non-insulin-dependent diabetes mellitus and impaired glucose tolerance. Am J Cardiol 1995; 76: 1173–6.
Grundy SM, Benjamin IJ, Burke GL, Chait A, Eckel RH, Howard BV, et al. Diabetes and cardiovascular disease: a statement for healthcare professionals from the American Heart Association. Circulation 1999; 100: 1134–46.
Brownlee M . Biochemistry and molecular cell biology of diabetic complications. Nature 2001; 414: 813–20.
Cai L, Kang YJ . Oxidative stress and diabetic cardiomyopathy: a brief review. Cardiovasc Toxicol 2001; 1: 181–93.
Ramasamy R, Oates PJ, Schaefer S . Aldose reductase inhibition protects diabetic and nondiabetic rat hearts from ischemic injury. Diabetes 1997; 46: 292–300.
Hwang YC, Bakr S, Ellery CA, Oates PJ, Ramasamy R . Sorbitol dehydrogenase: a novel target for adjunctive protection of ischemic myocardium. FASEB J 2003; 17: 2331–3.
Ramasamy R . Aldose reductase: a novel target for cardioprotective interventions. Curr Drug Targets 2003; 4: 625–32.
Chandra D, Jackson EB, Ramana KV, Kelley R, Srivastava SK, Bhatnagar A . Nitric oxide prevents aldose reductase activation and sorbitol accumulation during diabetes. Diabetes 2002; 51: 3095–101.
Lee AY, Chung SS . Contributions of polyol pathway to oxidative stress in diabetic cataract. FASEB J 1999; 13: 23–30.
Makita Z, Vlassara H, Rayfield E, Cartwright K, Friedman E, Rodby R, et al. Hemoglobin–AGE: a circulating marker of advanced glycosylation. Science 1992; 258: 651–3.
Wolffenbuttel BH, Giordano D, Founds HW, Bucala R . Long–term assessment of glucose control by haemoglobin–AGE measurement. Lancet 1996; 347: 513–5.
Bierhaus A, Illmer T, Kasper M, Luther T, Quehenberger P, Tritschler H, et al. Advanced glycation end product (AGE)-mediated induction of tissue factor in cultured endothelial cells is dependent on RAGE. Circulation 1997; 96: 2262–71.
Bierhaus A, Hofmann MA, Ziegler R, Nawroth PP . AGEs and their interation with AGE-receptors in vascular disease and diabetes mellitus. I. The AGE concept. Cardiovasc Res 1998; 37: 586–600.
Schmidt AM, Stern DM . RAGE: a new target for the prevention and treatment of the vascular and inflammatory complications of diabetes. Trends Endocrinol Metab 2000; 11: 368–75.
Esposito C, Gerlach H, Brett J, Stern D, Vlassara H . Endothelial receptor–mediated binding of glucose–modified albumin is associated with increased monolayer permeability and modulation of cell surface coagulant properties. J Exp Med 1989; 170: 1387–407.
Li J, Schmidt AM . Characterization and functional analysis of the promoter of RAGE, the receptor for advanced glycation end products. J Biol Chem 1997; 272: 16 498–506.
Schmidt AM, Hori O, Brett J, Yan SD, Wautier JL, Stern D . Cellular receptors for advanced glycation end products. Implications for induction of oxidant stress and cellular dysfunction in the pathogenesis of vascular lesions. Arterioscler Thromb 1994; 14: 1521–8.
Ritthaler U, Deng Y, Zhang Y, Greten J, Abel M, Sido B, et al. Expression of receptors for advanced glycation end products in peripheral occlusive vascular disease. Am J Pathol 1995; 146: 688–94.
Yan SD, Stern D, Schmidt AM . What's the RAGE? The receptor for advanced glycation end products (RAGE) and the dark side of glucose. Eur J Clin Invest 1997; 27: 179–81.
Lu M, Kuroki M, Amano S, Tolentino M, Keough K, Kim I, et al. Advanced glycation end products increase retinal vascular endot–helial growth factor expression. J Clin Invest 1998; 101: 1219–24.
Soudis-Liparota T, Cooper M, Papazoglou D, Clarke B, Jerums G . Retardation by aminoguanidine of development of albuminura, mesangial expansion, and tissue fluorescence in streptozotocin-induced diabetic rat. Diabetes 1991; 40: 1328–34.
Nakamura S . Progression of nephropathy in spontaneous diabetic rats is prevented by OPB-9195, a novel inhibitor of advanced glycation. Diabetes 1997; 46: 895–9.
Hammes HP . Aminoguanidine treatment inhibits the development of experimental diabetic retinopathy. Proc Natl Acad Sci USA 1991; 88: 11555–9.
Park L . Suppression of accelerated diabetic atherosclerosis by the soluble receptor for advanced glycation endproducts. Nature Med 1998; 4: 1025–31.
Koya D, King GL . Protein kinase C activation and the development of diabetic complications. Diabetes 1998; 47: 859–66.
Portilla D . Etomoxir-induced PPARalpha-modulated enzymes protect during acute renal failure. Am J Physiol Renal Physiol 2000; 278: F667–75.
Keogh RJ, Dunlop ME, Larkins RG . Effect of inhibition of aldose reductase on glucose flux, diacylglycerol formation, protein kinase C, and phospholipase A2 activation. Metabolism 1997; 46: 41–7.
Stevens MJ, Obrosova I, Feldman EL, Greene DA . The sorbitol-osmotic and sorbitol-redox hypothesis. In: LeRoith D, Taylor SI, Olefsky JM, editors. Diabetes mellitus: a fundamental and clinical text. Philadelphia: Lippincott Williams & Wilkins; 2000. p 972–83.
Lee AY, Chung SK, Chung SS . Demonstration that polyol accumulation is responsible for diabetic cataract by the use of transgenic mice expressing the aldose reductase gene in the lens. Proc Natl Acad Sci USA 1995; 92: 2780–4.
Yamaoka T, Nishimura C, Yamashita K, Itakura M, Yamada T, Fujimoto J, et al. Acute onset of diabetic pathological changes in transgenic mice with human aldose reductase cDNA. Diabetologia 1995; 38: 255–61.
Yagihashi S, Yamagishi S, Wada R, Sugimoto K, Baba M, Wong HG, et al. Galactosemic neuropathy in transgenic mice for human aldose reductase. Diabetes 1996; 45: 56–9.
Singh SB, Malamas MS, Hohman TC, Nilakantan R, Carper DA, Kitchen D . Molecular modeling of the aldose reductase–inhibitor complex based on the X–ray crystal structure and studies with single-site-directed mutants. J Med Chem 2000; 43: 1062–70.
Du XL . Hyperglycemia-induced mitochondrial superoxide overproduction activates the hexosamine pathway and induces plas–minogen activator inhibitor-1 expression by increasing Sp1 glycosylation. Proc Natl Acad Sci USA 2000; 97: 12222–6.
Kakkar R, Kalra J, Mantha SV, Prasad K . Lipid peroxidation and activity of antioxidant enzymes in diabetic rats. Mol Cell Biochem 1995; 151: 113–9.
Ohuwa T, Sato Y, Naoi M . Hydroxyl radical formation in diabetic rats induced by streptozotocin. Life Sci 1995; 56: 1789–98.
Bayes JW, Thorpe SR . Role of oxidative stress in diabetic complications: a new perspective on an old paradigm. Diabetes 1999; 48: 1–9.
Kowluru RA, Engerman RL, Kern TS . Diabetes–induced metabolic abnormalities in myocardium: effect of antioxidant therapy. Free Radical Res 2000; 32: 67–74.
Ustinova EE, Barrett CJ, Sun SY, Schultz HD . Oxidative stress impairs cardiac chemoreflexes in diabetic rats. Am J Physiol 2000; 279: 2176–87.
Rosen P, Du X, Sui GZ . Molecular mechanisms of endothelial dysfunction in the diabetic heart. Adv Exp Med Biol 2001; 498: 75–86.
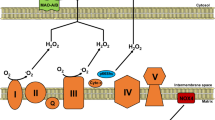
Nishikawa T, Edelstein D, Du XL, Yamagishi S, Matsumura T, Kaneda Y, et al. Normalizing mitochondrial superoxide production blocks three pathways of hyperglycaemic damage. Nature 2000; 404: 787–90.
Mohamed AK, Bierhaus A, Schiekofer S, Tritschler H, Ziegler R, Nawroth PP . The role of oxidative stress and NF-κB activation in late diabetic complications. Biofactors 1999; 10: 157–67.
Mercurio F, Manning AM . NF-κB as a primary regulator of the stress response. Oncogene 1999; 18: 6163–71.
Barnes PJ, Karin M . Nuclear factor-κB: a pivotal transcription factor in chronic inflammatory disease. N Engl J Med 1997; 336: 1066–71.
Baldwin AS Jr . The transcription factor NF-κB and human disease. J Clin Invest 2001; 107: 3–6.
Tak PP, Firestein GS . NF-κB: a key role in inflammatory disease. J Clin Invest 2001; 107: 7–11.
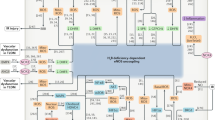
Tibbles LA, Woodgett JR . The stress–activated protein kinase pathways. Cell Mol Life Sci 1999; 55: 1230–54.
Ho FM, Liu SH, Liau CS, Huang PJ, Lin-Shiau SY . High glucose-induced apoptosis in human endothelial cells is mediated by sequential activations of c–Jun NH(2)-terminal kinase and caspase-3. Circulation 2000; 101: 2618–24.
Natarajan R, Scott S, Bai W, Yerneni KKV, Nadler J . Angiotensin II signaling in vascular smooth muscle cells under high glucose conditions. Hypertension 1999; 33: 378–84.
Dunlop ME, Muggli EE . Small heat shock protein alteration provides a mechanism to reduce mesangial cell contractility in diabetes and oxidative stress. Kidney Intl 2000; 57: 464–75.
Frustaci A, Kajstura J, Chimenti C, Jakoniuk I, Leri A, Maseri A, et al. Myocardial cell death in human diabetes. Circ Res 2000; 87: 1123–32.
Stockklauser-Farber K, Ballhausen T, Laufer A, Rosen P . Influence of diabetes on cardiac nitric oxide synthase expression and activity. Biochim Biophys Acta 2000; 1535: 10–20.
Tomita M, Mukae S, Geshi E, Umetsu K, Nakatani M, Katagiri T . Mitochondrial respiratory impairment in streptozotocin–induced diabetic rat heart. Jpn Circ J 1996; 60: 673–82.
Kucharska J, Braunova Z, Ulicna O, Zlatos L, Gvozdjakova A . Deficit of coenzyme Q in heart and liver mitochondria of rats with streptozotocin-induced diabetes. Physiol Res 2000; 49: 411–8.
Doroshow JH, Locker GY, Myers CE . Enzymatic defenses of the mouse heart against reactive oxygen metabolites: alterations produced by doxorubicin. J Clin Invest 1980; 65: 128–35.
Chen Y, Saari JT, Kang YJ . Weak antioxidant defenses make the heart a target for damage in copper-deficient rats. Free Radical Biol Med 1994; 17: 529–36.
Kersten JR, Schmeling TJ, Orth KG, Pagel PS, Warltier DC . Acute hyperglycemia abolishes ischemic preconditioning in vivo. Am J Physiol 1998; 275: H721–5.
Joyeux M, Faure P, Godin-Ribuot D, Halimi S, Patel A, Yellon DM, et al. Heat stress fails to protect myocardium of streptozotocin-induced diabetic rats against infarction. Cardiovasc Res 1999; 43: 939–46.
Elangovan V, Shohami E, Gati I, Kohen R . Increased hepatic lipid soluble antioxidant capacity as compared to other organs of streptozotocin-induced diabetic rats: a cyclic voltametry study. Free Radical Res 2000; 32: 125–34.
Ye G, Metreveli NS, Ren J, Epstein PN . Metallothionein prevents diabetes-induced deficits in cardiomyocytes by inhibiting reactive oxygen species production. Diabetes 2003; 52: 777–83.
Ye G, Metreveli NS, Donthi RV, Xia S, Xu M, Carlson EC, et al. Catalase protects cardiomyocyte function in models of type 1 and type 2 diabetes. Diabetes 2004; 53: 1336–43.
Alici B, Gumustas MK, Ozkara H, Akkus E, Demirel G, Yencilek F, et al. Apoptosis in the erectile tissues of diabetic and healthy rats. BJU Int 2000; 85: 326–9.
Cai L, Chen S, Evans T, Deng DX, Mukherjee K, Chakrabarti S . Apoptotic germ-cell death and testicular damage in experimental diabetes: prevention by endothelin antagonism. Urol Res 2000; 28: 342–7.
Srinivasan S, Stevens M, Wiley JW . Diabetic peripheral neuropathy: evidence for apoptosis and associated mitochondrial dysfunction. Diabetes 2000; 49: 1932–8.
Fiordaliso F, Li B, Latini R, Sonnenblick EH, Anversa P, Leri A, et al. Myocyte death in streptozotocin-induced diabetes in rats is angiotensin II-dependent. Lab Invest 2000; 80: 531–7.
Privratsky JR, Wold LE, Sowers JR, Quinn MR, Ren J . AT1 blockade prevents glucose-induced cardiac dysfunction in ventricular myocytes: role of the AT1 receptor and NADPH oxidase. Hypertension 2003; 42: 206–12.
Dzau VJ . Theodore Cooper Lecture: tissue angiotensin and pathobiology of vascular disease: a unifying hypothesis. Hypertension 2001; 37: 1047–52.
Nickenig G, Harrison DG . The AT1type angiotensin receptor in oxidative stress and atherogenesis, part I: oxidative stress and atherogenesis. Circulation 2002; 105: 393–6.
Sowers JR . Hypertension, angiotensin II, and oxidative stress. N Engl J Med 2002; 346: 1999–2001.
Mascareno E, Siddiqui MA . The role of Jak/STAT signaling in heart tissue renin-angiotensin system. Mol Cell Biochem 2000; 212: 171–5.
Wang HD, Xu S, Johns DG, Du Y, Quinn MT, Cayatte AJ, et al. Role of NADPH oxidase in the vascular hypertrophic and oxidative stress response to angiotensin II in mice. Circ Res 2001; 88: 947–53.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wold, L., Ceylan-isik, A. & Ren, J. Oxidative stress and stress signaling: menace of diabetic cardiomyopathy. Acta Pharmacol Sin 26, 908–917 (2005). https://doi.org/10.1111/j.1745-7254.2005.00146.x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1111/j.1745-7254.2005.00146.x
Keywords
This article is cited by
-
Engineered cardiac tissues: a novel in vitro model to investigate the pathophysiology of mouse diabetic cardiomyopathy
Acta Pharmacologica Sinica (2021)
-
Caffeic Acid Protects against Iron-Induced Cardiotoxicity by Suppressing Angiotensin-Converting Enzyme Activity and Modulating Lipid Spectrum, Gluconeogenesis and Nucleotide Hydrolyzing Enzyme Activities
Biological Trace Element Research (2021)
-
Mitochondrial Ca2+ regulation in the etiology of heart failure: physiological and pathophysiological implications
Acta Pharmacologica Sinica (2020)
-
Biochemical characterization and 1H NMR based metabolomics revealed Melicope lunu-ankenda leaf extract a potent anti-diabetic agent in rats
BMC Complementary and Alternative Medicine (2017)
-
Reactive oxygen species up-regulates SOCS-3 in 3T3-L1 adipocytes
International Journal of Diabetes in Developing Countries (2015)