Host: Benjamin Thompson
Welcome back to the Nature Podcast. This week, we’ll be hearing how performance arts could help researchers perform science.
Host: Shamini Bundell
And we’ll be shining a light on chiral chemistry. I’m Shamini Bundell.
Host: Benjamin Thompson
And I’m Benjamin Thompson.
[Jingle]
Interviewer: Benjamin Thompson
These days, it’s hard to escape the word ‘interdisciplinary’. While most modern researchers don’t have quite the variety of interests as certain famous Victorian polymaths, there’s certainly more collaboration happening between different scientific subject areas. But it’s not just collaborations between scientific disciplines that are being explored. This week in Nature, we’ve got an essay in our Books and Arts section looking at what researchers can learn from artists and performers, and vice versa. One of the essay’s co-authors is Roger Kneebone, a surgeon and general practitioner by training, now professor of Surgical Education and Engagement Science at Imperial College London. Roger dropped by the studio along with Will Houstoun, a magician who has a PhD in Victorian Magic Literature. The two of them chatted about what the worlds of medicine and science can learn from performance, magic and even cooking. Roger began by talking about the performance of a medical consultation.
Interviewee: Roger Kneebone
I’ve been in the world of medicine ever since I qualified in 1977 but as I became more experienced, I came to see the consultation itself as a kind of performance, and that’s where, when I met some magicians a while ago, a penny dropped with me that there was an awful lot of stuff that I could learn from other expert performers who deal, in this case, with a very small audience if I thought of myself as a performer as a doctor, and Will I know that you do a lot of work with very small audiences.
Interviewee: Will Houstoun
Yeah, absolutely. One of the most common places for someone to perform magic I suppose is in a close-up setting, working with two or three people at a time, showing them card tricks, coin tricks and that sort of thing. And in those settings, people don’t necessarily know what you’re doing – are you going to do something that’s fun for them, are you some sort of con man, are you somebody who’s going to make fun of them? They don’t really know, so those same sort of interpersonal dynamics I guess come into play a little.
Interviewee: Roger Kneebone
Because you get those in the consulting room as well. I mean people come and see you and they’ve never met and they’re often very anxious. The skill is, I think, making a connection with somebody and then moving the conversation forward. One of the things that struck me when I first saw you work is the way that you are very skilful in establishing a connection while making it seem completely natural and effortless, but I know from my own experience that making things seem natural and effortless takes a lot of effort.
Interviewee: Will Houstoun
Yeah, absolutely. So, you don’t want somebody to feel, I think, that they’re in an incredibly contrived and peculiar situation or that you are there sort of just blindly going through something you’ve practiced loads of times, and so you have to make sure you’re working with the person and aware of what they’re doing and how they’re acting. And you certainly have an idea of where you’re trying to go with something, what the end of the trick is, what point you’re hoping to reach, but you never know quite what’s going to happen along the way.
Interviewee: Roger Kneebone
So, the work that we’ve done about looking at the consultation as performance really made me think that there are all kind of insights that people in my profession – in my case, medicine – can gain from people in completely different areas of practice. A year or so ago I know you took part in a symposium I organised at Imperial College London where we brought together about 65 people from very, very different backgrounds to explore with one another what they actually do. And there we had scientists and clinicians and artists and performers, and an awful lot of people found that they had unexpected areas of similarity, whether they were restoring statues in the V&A Museum, or whether they were manipulating tiny insects under the microscope or doing things in a chemistry laboratory. And I know that you do things that require great skill and delicacy and manipulation, but I know that you’ve also got a science background as well yourself, haven’t you, an engineering background.
Interviewee: Will Houstoun
Yes.
Interviewee: Roger Kneebone
And I wonder if that made sense to you as another point of sort of bridging.
Interviewee: Roger Kneebone
I did a Mechanical Engineering degree to start with, and very much liked the world of magic and did magic tricks, but never really thought the two could crossover. And then it was only really afterwards when I started getting more interested in the magic world, did a PhD based on Victorian Magic Literature, and then started to find actually there’s lots of interesting stuff where the two cross. I rather wish there had been somebody saying, ‘You know what, these things can inform each other.’
Interviewee: Roger Kneebone
Although we talk about performing operations, we talk about performing experiments, I think, you know, people don’t always follow that thought through. But when I started exploring whether these ideas I’d noticed in my world of medicine might also play out in laboratory science, I discovered that there were all sorts of similarities there that I hadn’t thought of, so people in chemistry laboratories having to work with very precise measurement and exact temperatures and mixtures of things and close observation, all those skills seem to me to be mirrored very closely by what happens in high-end chefs, kitchens. And interestingly, the symposium I mentioned has now led onto a collaboration and we’re going to be getting undergraduate chemistry students to spend time learning culinary techniques in a kitchen, and getting culinary students to spend time in a chemistry laboratory to see whether each can learn from the other.
Interviewee: Will HoustounAnd of course, the wonderful thing about that I suppose is that if I’m doing something which is about magic, I have a feeling of obligation to be good and sort of know something about it and be able to do it to a certain extent, but if I’m spending time with somebody else – I’ve been working with a puppeteer, for example, recently – I have no expectation of being good at puppetry, and perhaps I can learn more because I’m not worried about proving myself in the same way.
Interviewee: Roger Kneebone
And that’s exactly the case with the Chemical Kitchen, as we’re calling this project, which is being funded by Imperial College London as a way of helping chemistry students particularly to learn these techniques that, these days, they may not have learnt much or indeed at all at school. And this is a way of making sure that they gain those skills, exactly as you say, without them feeling embarrassed in front of a chemistry professor if they can’t do a chemical experiment because no one expects them to be able to make a good soufflé or vice versa. And I think that particularly as the school curriculum is becoming scooped out and opportunities to perform and to do things with your hands are being systematically eliminated, I think it’s getting more and more important that we give people the opportunities to make these connections with people they might not otherwise think of meeting. And so, the collaboration that we’ve developed between magic and medicine seems to me to be an example of something that has a much wider set of implications.
Interviewee: Will Houstoun
And a particularly exciting one for me because my PhD was looking at a man who wrote a book about how to do magic in the Victorian period, not because everyone should be magicians but rather because he thought that magic would teach people useful skills for the rest of their lives, and that’s something that sort of took off a little bit, then faded away in the intervening 100-120 years, and it’s rather exciting to think that maybe the idea of using magic to help look at other things and examine other areas is there, and also of course that magic can become much richer by interacting with other areas, by finding out more about other things, and by using some of that in the way it develops.
Interviewer: Benjamin ThompsonThat was Will Houstoun chatting with Roger Kneebone. You can read Roger’s essay over at nature.com/news. If you’d like to see a longer discussion between the two on the lessons that medical consultations could learn from performance magic, you can find one over on Gresham College’s website where Roger has been giving a series of talks, and I’ll put a link to that on our show page over at nature.com/nature/podcast.
Host: Shamini BundellLater in the show, Holly Else will be joining us with an update from an AI conference – that’s coming up in the News Chat. Now though, Anna Nagle is here with this week’s Research Highlights.
[Jingle]
Interviewer: Anna NagleI’d imagine that dissecting a mosquito takes some pretty fine motor skills, but how about dissecting half a million mosquitoes? That’s what a team from the University of California did in a bid to uncover new compounds to treat malaria. When an infected mosquito bites someone, Plasmodium parasites enter into the person’s bloodstream, and first move to the liver where they mature. The California team wanted to target the parasite at this early stage in its life cycle, which meant painstakingly removing Plasmodium parasites from nearly half a million mosquitoes. That then mixed the parasites with human liver cells and pitted them against various chemical compounds to see how they fared. Out of more than 500,000 compounds, almost 6,000 quashed the pathogen without damaging the liver cells, and could form the basis for new malaria prevention drugs. Read more on that research in Science.
[Jingle]
Interviewer: Anna NagleDid you know that geckos can race across the surface of water at impressive speeds? Well, now scientists have used high-speed cameras to work out exactly how they manage it. Many species can travel along water’s surface. Lightweight creatures like insects make use of surface tension, while some larger animals like basilisk lizards use vigorous water slapping techniques. Geckos, it turns out, use a mix of approaches. Scientists at Rockefeller University in New York found that geckos use all four limbs to slap and stroke the water’s surface, creating underwater air pockets that propel them forwards. They also wiggle their semi-submerged bodies from side to side to generate thrust and get a boost from the water’s surface tension. This combination of techniques means that geckos can speed across a body of water at around 61 centimetres per second. That’s faster than a young alligator can swim. You can find that research in Current Biology.
[Jingle]
Interviewer: Shamini Bundell
Now, one of the nice things about working at Nature is you get to hang around with a lot of people who are very excited about the goings on in their particular field, and they’re usually pretty happy to tell you about it too. One such person is Bryden Le Bailly, a senior editor for the Nature journal. I got him down to the studio last week to fill us in on some cool new research from the field of organic chemistry. So, you have a new paper that you’ve accepted for publication in Nature, and it’s one that you were quite excited about. What have they actually done in this paper?
Interviewee: Bryden Le Bailly
So, they’ve done something that people couldn’t do before and didn’t know was possible before, so what they’ve done is they’ve managed to take a mixture of enantiomers, which is basically mirror image versions of the same compound, the same molecule, and they’ve managed to convert one to the other.
Interviewer: Shamini Bundell
And this idea of having different enantiomers, let’s explain that a little bit. What is an enantiomer?
Interviewee: Bryden Le Bailly
Sure, so I think the easiest way of explaining it and the one they usually tell people at school or at university is it’s like having two hands, right. So, you have a left land and a right hand, and they are mirror images of each other. So, molecules are exactly the same, they can exist as two hands of each other and they’re called enantiomers.
Interviewer: Shamini Bundell
So, one of the important things is that if you take your left hand, you can’t just rotate it and get the exact same pattern as your right hand.
Interviewee: Bryden Le Bailly
Exactly, yeah.
Interviewer: Shamini Bundell
Because your thumb is sticking out and your palm is in the wrong direction.
Interviewee: Bryden Le Bailly
Yeah.
Interviewer: Shamini Bundell
So, there are molecules like that as well and they’re called chiral molecules if they have these.
Interviewee: Bryden Le Bailly
Exactly, yeah, so a molecule is chiral if it has another form that’s a mirror image, and those two forms are called enantiomers.
Interviewer: Shamini Bundell
And why is that so important?
Interviewee: Bryden Le Bailly
So, biology is inherently chiral. So, for example, if you think about your DNA, that’s the classic right-handed helix of DNA, that’s determined by the fact that the nucleic acids that make up DNA, they induce it to form a right-handed helix. Likewise, your proteins are made up of amino acids, and they also form helices, single helices, and they form left-handed helices.
Interviewer: Shamini Bundell
So, if I had the wrong enantiomer in my DNA or in my proteins, the chemistry would be different.
Interviewee: Bryden Le Bailly
Exactly, it wouldn’t fold properly. It wouldn’t be able to perform its function the same way.
Interviewer: Shamini Bundell
Why is this a problem for chemists?
Interviewee: Bryden Le Bailly
Because biology is inherently chiral, this means that if we make a drug, any kind of chemical to interact with biology, if we make the wrong enantiomer, the wrong hand, it won’t have the same effects. So, one really obvious example of this is limonene, which is a compound that’s found in citrus fruits. So we have one enantiomer of limonene that smells like oranges, smells like lemons, that’s the citrus smell, but if you have the other version of limonene, the other hand in this, that smells like industrial solvent, and that’s what it’s actually used for.
Interviewer: Shamini Bundell
No way.
Interviewee: Bryden Le Bailly
Yeah, so these molecules look almost exactly the same, but they’re mirror images of each other and they have very different physical properties and biological properties.
Interviewer: Shamini Bundell
So, when biology is making all these molecules, it’s natural processes end up with all of one kind, but in chemistry you’d usually end up with 50/50 of each enantiomer, so how do people get around that currently?
Interviewee: Bryden Le Bailly
So, normally when you make a chiral molecule, you start from starting material, another molecule, and you transform it and impart the chirality on it, okay, and you form one enantiomer, and that’s done normally using quite expensive, metal-based catalysts. And if you don’t have the catalyst set up right, you won’t get one enantiomer, you’ll just get the mixture and then you might have bad side effects, and there are several examples in the literature where the other enantiomer has bad side effects.
Interviewer: Shamini Bundell
So, what have these guys done now that’s different?
Interviewee: Bryden Le Bailly
In this paper, what they do is they take this mixture of enantiomers, of a molecule called an allene, and they effectively can resolve that mixture to just one enantiomer, but of the exact same thing, and this is something that people didn’t know was possible before. So, this means that if you get to the end of your synthesis of making a drug that’s chiral, you could potentially go all the way through with your mixture, and then just at the end convert it into the right enantiomer that you want to work as a drug.
Interviewer: Shamini Bundell
So, with normal metal catalysts that people use to create one-handed chiral molecules, you’re starting off with non-chiral molecules and you’re adding this chirality and you’re adding the one-handedness, one version, of that.
Interviewee: Bryden Le Bailly
Exactly.
Interviewer: Shamini Bundell
Whereas in this one you’re starting with already chiral molecules and just flipping half of them over.
Interviewee: Bryden Le Bailly
Absolutely, yeah. So, the importance is that it doesn’t matter that you have this mixture, because then you can go on to convert it later.
Interviewer: Shamini Bundell
And the mixtures are actually quite easy to create.
Interviewee: Bryden Le Bailly
Exactly.
Interviewer: Shamini Bundell
And how have this team achieved it if normal metal catalysts can’t do it?
Interviewee: Bryden Le Bailly
So, the importance here is that they’ve used light, so the catalyst is activated by light, so what happens is the catalyst absorbs light, and then it will transfer that light energy to the molecule, and that gives the molecule enough energy to covert one enantiomer to the other.
Interviewer: Shamini Bundell
So, this is going to be for sort of pharmaceutical, industry and drug development and that kind of thing. This is going to be potentially a new way of making existing drugs, or maybe a more efficient way of making them.
Interviewee: Bryden Le Bailly
Sure.
Interviewer: Shamini Bundell
And maybe we’ll be able to make compounds that we’ve so far just not really been able to make.
Interviewee: Bryden Le Bailly
Exactly.
Interviewer: Shamini Bundell
And is it just drugs? Are there any other elements of the world in which chirality is important?
Interviewee: Bryden Le Bailly
Obviously, the whole world is chemical, right, so anything to do with materials or any other processes where you’re doing chemical manufacturing. So for example, in polymer chemistry which obviously you use in plastics and things like that, the chirality of those polymers can be used to affect what their strength is, how bendable they are.
Interviewer: Shamini Bundell
And this is just the first demonstration of this new technique. How far does it have to go before industry and people can actually make use of it?
Interviewee: Bryden Le Bailly
So, one of the limitations for industry is the use of lights. So, light is actually because of its penetration that makes it difficult to use on a very big scale. So, if you have a really big reactor, you know, tons and tons of chemicals being made, to try and get light into there is a problem, so this is something that we also have to overcome.
Interviewer: Shamini Bundell
So maybe we’ll have vats full of little LEDs.
Interviewee: Bryden Le Bailly
Yeah, absolutely. I mean at the moment, light-based chemistry is a very hot topic. The last ten years or so have seen an explosion in what we call photocatalysis because what it’s allowed us to do, as in this paper, is explore completely new kinds of reactivity that were just not really accessible to us before.
Interviewer: Shamini Bundell
That was Bryden Le Bailly, journal editor here at Nature, talking about a paper which you can find at nature.com/nature, along with a News and Views to explain some more of that lovely chiral chemistry.
Interviewer: Benjamin ThompsonRight then listeners, it’s that time of the show. It’s actually the penultimate News Chat of 2018, and I’m joined here in the studio by Holly Else, one of the reporters here at Nature. Hi Holly.
Interviewee: Holly ElseHi.
Interviewer: Benjamin Thompson
A couple of stories today, and the first one is about AI and a recent conference you were at.
Interviewee: Holly ElseYes, so I travelled to Montreal last week to attend the NeurIPS conference which is one of the biggest AI conferences in the world.
Interviewer: Benjamin Thompson
And there were two big announcements as I understand it. What were they?
Interviewee: Holly ElseYeah, one was a big announcement from researchers in Europe who want to keep AI going on the continent because they fear they’re losing a lot of people to North America and China.
Interviewer: Benjamin Thompson
And what are they doing to kind of prevent this flow of researchers to other parts of the world?
Interviewee: Holly Else
So, they’ve set up a group called ELLIS which stands for the European Laboratory for Learning and Intelligent Systems, and this is kind of like an organisation that’s going to be based on EMBO (the European Molecular Biology Organisation). And what they’re going to do is coordinate better AI research in Europe.
Interviewer: Benjamin Thompson
So, is this something that needs better coordination then? Is it not in such a good state as we stand at the moment?
Interviewee: Holly ElseWell, there are still some leading labs in Europe, but the danger is that a lot of the industry and PhD programmes, for example, actually the top ones are in America now. And what this group fear is that they’re actually sort of draining the resources out of Europe, so taking the students, taking the money, and developing the technology outside of Europe.
Interviewer: Benjamin Thompson
Well, quite a broad ambition it sounds like. What sort of things will this include specifically, Holly?
Interviewee: Holly ElseSo they’re going to set up a few research centres. They weren’t able to tell us exactly where they would be yet, but there’s definitely interest in Germany and Austria. They’re also planning to create a pan-European PhD programme and they’ll also create some kind of focal point for industry. A lot of European companies might need AI for the future and perhaps aren’t so developed in it as their counterparts in the States or China.
Interviewer: Benjamin Thompson
Right, and I guess an important question is who’s going to pay for it?
Interviewee: Holly ElseWell, we don’t know yet, that’s the big question. So, the announcement at NeurIPS was basically just to say that they’re forming this professional association in order to do these things, but they weren’t able to tell us where they’re going to get the money from yet.
Interviewer: Benjamin Thompson
Well, I guess wait and see on that one then, but I know the other thing was related to ethics, and it’s something that we’ve talked about on the podcast before, you know, the ethics of machine learning and AI and what have you. What was announced at the conference?
Interviewee: Holly ElseSo the University of Montreal and the Quebec Research Fund came together a year ago and decided to write some principles that can help guide researchers to do AI ethically, and what they did over the past year is consult the public and also different groups like policymakers, sociologists, public organisations, about AI and what the future will hold. And together they came up with these ten principles known as the Montreal Declaration, and a lot of it is about fairness, equality, responsible AI development. Really the main gist is that AI is a technology that should benefit everybody in society, not just some few.
Interviewer: Benjamin Thompson
I mean is this the first time this has been attempted? It seems like trying to get these core principles is an important thing to have done when we started off working with AI.
Interviewee: Holly ElseYeah, so there are other guidelines, but these have really been devised by the researchers themselves, so this is the first time the scientists have actually gone into the community and asked the people, who really are the ones who are going to be affected by AI in the long term, what they think and how they think that technology should be developed.
Interviewer: Benjamin Thompson
I guess the key with these things is it’s all very well to sort of develop these guidelines, but putting them into practice I guess can be the trick. How are these going to be implemented and what are researchers thinking about them?
Interviewee: Holly ElseSo at the moment about 500 people have signed up to these guidelines – most of them are Canadian or French researchers. So, there’s obviously some way to go in terms of publicising this to the wider community.
Interviewer: Benjamin Thompson
Well Holly, let’s move on to our second story this week, and it’s also from a conference but a very different conference, and one that’s going on right now.
Interviewee: Holly ElseYeah, so this is the conference of the American Geophysical Union, and some research that was presented there suggests that East Antarctica is melting much faster than people originally thought.
Interviewer: Benjamin Thompson
I mean it seems rare that we get some good news from Antarctica, and this doesn't sound like good news. What specifically has been going on?
Interviewee: Holly ElseWell, people thought the east side of Antarctica was relatively stable, but since the early 2010s, the height of the glaciers is falling by about half a metre a year.
Interviewer: Benjamin Thompson
Crikes. Okay, well I mean a couple of questions from me here. One, we talked about East Antarctica which seems kind of broad, where specifically are we looking? And two, how does one go about measuring the height of a glacier? Presumably you’re not just there with a metre ruler or something like that.
Interviewee: Holly ElseYeah, so to answer your first question, the researchers specifically were looking at an area called Vincennes Bay, which has four glaciers in it. And how did they do it? Well, they used data from two satellites – one run by NASA and another one by the European Space Agency – and they used this data to create a 3D view of what the surface of each glacier looked like and how they thickened and thinned over time. They found that in the 1990s, the glaciers shrank while they shed some ice, but then they bulked up again in the 2000s, and then from 2010 onwards they started to thin again.
Interviewer: Benjamin Thompson
And this is the 0.5 metres a year that we’re currently seeing.
Interviewee: Holly ElseYes.
Interviewer: Benjamin Thompson
I mean it does seem like there’s been a little bit of variation there, some up and down, but where do we think this is going next?
Interviewee: Holly ElseWell, a lot of this comes down to the temperature of the water around East Antarctica, and that changes every year and can be based on factors like wind speed and direction or whether there’s sea ice in the vicinity. But in general, climate scientists think that the warmer waters are going to make their way closer to East Antarctica more regularly in the future, and that doesn’t bode too well for the glaciers.
Interviewer: Benjamin Thompson
No, indeed not. Well Holly, thank you for joining me, and listeners if you’d like more of the latest science news, head over to nature.com/news.
Host: Shamini BundellAnd that’s it for this week’s show, but you have to tune in next week because we have a holiday show and I’m very much looking forward to that.
Host: Benjamin ThompsonYeah, me too. But listeners, while you wait for that, if you’d like to see magician Will Houstoun performing a bit of magic here in our studio, head over to our Twitter feed (@NaturePodcast) where you’ll find him in GIF form.
Host: Shamini BundellYeah, I’ve seen those GIFs – they’re crazy. I’m sure you must have manipulated the video somehow Ben.
Host: Benjamin Thompson
No, it’s all magic and it’s all amazing. I’m Benjamin Thompson.
Host: Shamini BundellAnd I’m Shamini Bundell.

 Science in hand: how art and craft can boost reproducibility
Science in hand: how art and craft can boost reproducibility
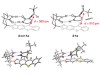 Read the paper: Catalytic deracemization of chiral allenes by sensitized excitation with visible light
Read the paper: Catalytic deracemization of chiral allenes by sensitized excitation with visible light
 Five years of record warmth intensify Arctic's transformation
Five years of record warmth intensify Arctic's transformation